|
HAART-Multiregimen Failure
The 20 Faces of HIV
European Resistance Workshop–Part 5. Multiregimen Failure
Mark Mascolini ( markmascolini@earthlink.net)
Knowing when to switch all or part of an incompletely suppressive regimen rests partly on science and partly on art. At the Third European HIV Drug Resistance Workshop, Jens Lundgren (Hvidore University Hospital, Copenhagen) outlined the art involved in a review talk [1] (Table 5.1).

For people with advanced HIV disease and few antiretroviral options, the choice is easier because the science is stronger. Numerous studies cited by Lundgren show that CD4 counts remain stable or even rise when an only partially effective regimen continues [2-4]. So it makes sense to continue therapy while hoping a suppressive regimen can be patched together later.
The workshop added some science for clinicians to consult when making tough switch-or-stay calls.
Testing a salvage strategy called continuously alternating therapy (CAT), clinicians in Berlin uncovered an interesting phenomenon. CAT switches between regimens consisting only of protease inhibitors (PIs) and regimens consisting only of nucleoside reverse transcriptase inhibitors (NRTIs), aiming with each flip to select drug-sensitive virus that the following regimen can suppress [5]. Although only a few CAT participants have seen their virus revert to a largely sensitive population, no major mutations emerge during the switches and viral loads do drop after a new regimen starts.
To figure out what’s going on, Monika Tschochner (Institute for Clinical and Molecular Virology, Erlangen) inspected 127 viral clones derived from samples collected from three people who had at least a 3-fold jump in viral load with a new regimen, two who had more than a 3-fold drop, and four who maintained a stable load [6].
Tschochner’s exercise turned up 56 mutations at 31 protease positions that conventional sequencing had missed, and 189 missed mutations at 106 reverse transcriptase positions. The number of covert mutations proved higher in clones derived from people with rising viral loads, decreased HIV susceptibility to antiretrovirals, and changes in viral replicative capacity (or "fitness").
This higher number of minority mutants, Tschochner surmised, may indicate "recruitment of different viral variants with optimized viral replication during stable nonsuppressive therapy." In other words, even when major mutations do not arise while a shaky regimen continues, HIV continues to mutate in subtle ways that can make the virus meaner.
Andrea De Luca (Catholic University, Rome) took a closer look at one interaction explored by Tschochner–the impact of resistance mutations on viral fitness in people starting a rescue regimen [7]. His study involved 139 people from the ARGENTA trial, which rated the prowess of genotyping in picking a new regimen [8].
One regimen had failed in half of the ARGENTA group, two regimens in 25%, and three or more regimens in the other 25%. They had tried a median of two potent combinations for a median 18 months (interquartile range [IQR] 12 to 24 months) and began ARGENTA with a median HIV load of 4.28 logs (IQR 3.78 to 4.69 logs) and a median CD4 count of 264 cells/µL (IQR 146 to 389 cells/µL).
De Luca measured fitness with the ViroLogic assay, which reckons the replicative capacity of mutant virus compared with nonmutant or "wild-type" virus. A replication capacity of 50%, for example, means the mutant virus multiplies half as well as nonmutant virus.
Linear regression analysis picked out six mutations in protease and five in reverse transcriptase that correlated with worse replication capacity (Table 5.2).
Table 5.2. Decreased viral fitness tied to specific resistance mutations
| |
r* |
P |
|
Protease |
|
|
|
L10I |
-0.18 |
0.03 |
|
M46I |
-0.31 |
0.0001 |
|
I54V |
-0.23 |
0.004 |
|
V82A |
-0.22 |
0.003 |
|
I84V |
-0.25 |
0.006 |
|
L90M |
-0.10 |
0.03 |
|
Reverse transcriptase |
|
|
|
Y75I |
-0.23 |
0.01 |
|
F116Y |
-0.14 |
0.09 |
|
Y118I |
-0.16 |
0.05 |
|
M184V |
-0.15 |
0.06 |
|
T215Y |
-0.15 |
0.07 |
*The r value measures the slope of the correlation. A higher number means a tighter correlation.
In a multivariate analysis two mutations predicted a replication capacity less than the average minus 1 standard deviation:
- L90M: odds ratio 3.2, P < 0.01
- Y118I: odds ratio 2.7, P = 0.05
Notably, no nonnucleoside-related mutations affected viral fitness. Because of the ARGENTA group’s relatively modest size, De Luca cautioned that the specific mutations that arose in this group partly dictate the links he found between mutations and fitness.
Cumulative mutations also correlated with reduced replication capacity–up to a point. Fitness waned more with one mutation than with none, more with two than with one, and more with three than with two (P = 0.003 for zero versus three mutations and for two versus three). But a fourth mutation did not further impair fitness.
Substitutions at six sites correlated with improved fitness–59 and 64 in protease and 90, 95, 100, and 172 in reverse transcriptase.
Analysis of 632 people genotyped during antiretroviral failure from June 1999 to June 2003 found that about one quarter of the population had virus resistant to multiple NRTIs or nonnucleosides (NNRTIs), while only 13% had multi-PI resistant virus [9]. Rates of multi-NNRTI-resistant virus hit 60% in the first year of NNRTI regimen failure–not a surprise since a single mutation can knock out the whole class. Rates of multi-NRTI and multi-PI resistant virus started lower but rose inexorably with each year of failed therapy. This cohort study adds to the understanding of multidrug resistance by correlating it with virologic response after a new regimen begins.
Defining multidrug resistance by International AIDS Society (IAS)-USA criteria, Valerio Tozzi (National Institute for Infectious Diseases Lazzaro Spallanzani, Rome) rated resistance patterns in 632 people in whom an average 3.8 regimens had failed (+2.2 standard deviation [SD]). The group viral load averaged 4.5 logs (+1.3 SD) and the CD4 count 298 cells/µL (+214 SD).
Tozzi counted 169 people (28.7%) with multi-NRTI-resistant virus, 156 of them with four or more thymidine analog mutations (TAMs) and only a handful with multiple resistance via the Q151M resistance complex or position 69 insertions. The rate of Q151M-inspired multiresistance peaked at around 5% after 2 years of NRTI therapy, then dropped. But TAM-induced resistance climbed significantly year after year (P < 0.001).
The cohort included 164 people (25.9%) with multi-NNRTI resistance, 130 of them with the class-killing K103N mutation and most of the rest with Y188L. Only 32 people (13.0%) had virus resistant to multiple PIs, a finding reflecting slower emergence of resistance to this class.
Multivariate analysis pinpointed several factors that independently boosted the risk of multi-NRTI or multi-PI-resistant virus (Table 5.3).
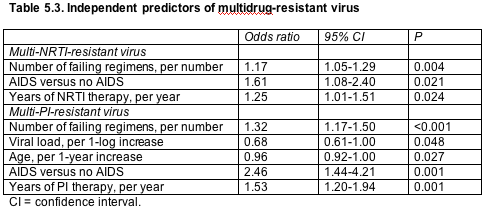
CI = confidence interval.
None of the variables analyzed independently predicted multi-NNRTI-resistant virus. Viral load, CD4 count at genotyping, age, and gender did not correlate with multi-NRTI resistance; and CD4 count at genotyping did not predict multi-PI resistance.
Another multifactor analysis showed that multi-NRTI and multi-NNRTI resistance each lowered the chance of getting the viral load under 500 copies/mL by about 25%. Multi-PI resistance trimmed the chance of responding about 35%. And triple-class resistance lowered the chance of responding 78%.
Randomized trials of genotyping after virologic failure show that this resistance test does better than drug history alone in picking a new combination. But follow-up in these studies rarely extends beyond a year. To find out whether genotyping really helps over the long term in a single clinic, Mark Oette (University Clinic of Düsseldorf, Germany) took a retrospective look at 117 people genotyped, retreated, and tracked for 96 weeks [10]. By intent-to-treat analysis about 40% got their viral load under 50 copies/mL and kept it there throughout follow-up.
This is a predominantly male (86%) and gay (60%) cohort with a median viral load of 7710 copies/mL (range 166 to 500,001 copies/mL) and a median CD4 count of 291 cells/µL (range 1 to 931 cells/µL). They had taken antiretrovirals for a median 252 weeks (range 22 to 843 weeks) and had tried a median of eight antiretrovirals (range two to 15). About 37% had a second antiretroviral switch during follow-up and about 20% a third.
Oette found that about half of the cohort regained complete control of viral replication within 24 weeks of switching, and that rate did not slip through 96 weeks of follow-up (Table 5.4).

But once a person in this cohort had to switch regimens, they generally needed later switches to maintain consistent viral control. People who changed regimens only once during follow-up did worse than people who changed therapy more often. Although the on-treatment sub-50-copy rate among single switchers hit 50% in 24 weeks and stayed there, the intent-to-treat rate crested at 40% in 24 weeks then skidded steadily to about 20% at week 96.
The median CD4 count rose more than 100 cells/µL in the whole group through 96 weeks but only about 50 cells/µL among single switchers.
Mark Mascolini writes about HIV infection ( markmascolini@earthlink.net).
References
(To view slides and posters from the Third European HIV Drug Resistance Workshop, go to http://www.hivpresentation.com.)
1 . Lundgren J. How to read viral load in patients with virus variants that you cannot suppress. Third European HIV Drug Resistance Workshop. March 30-April 1, 2005. Athens.
2. Kaufmann D, Pantaleo G, Sudre P, et al. CD4-cell count in HIV-1-infected individuals remaining viraemic with highly active antiretroviral therapy (HAART): Swiss HIV Cohort Study. Lancet 1998;351:723-724.
3. Deeks SG, Wrin T, Liegler T, et al. Virologic and immunologic consequences of discontinuing combination antiretroviral-drug therapy in HIV-infected patients with detectable viremia. N Engl J Med 2001;344:472-480.
4. Ledergerber B, Lundgren JD, Walker AS, et al. Predictors of trend in CD4-positive T-cell count and mortality among HIV-1-infected individuals with virological failure to all three antiretroviral-drug classes. Lancet 2004;364:51-62.
5. Stocker H, Weber C, Masuhr A, et al. Continuously alternating therapy (CAT) can lead to resensitization to antiretroviral drugs in patients with multi-drug resistant HIV-1. XV International AIDS Conference. July 11-16, 2004. Bangkok. Abstract TuPeB4556.
6. Tschochner M, Weber C, Stocker H, et al. Small increases in HIV viral load under non-suppressive antiretroviral treatment are associated with increasing numbers of mutations in viral minority populations. Third European HIV Drug Resistance Workshop. March 30-April 1, 2005. Athens. Abstract 38. Poster 7.2
7. De Luca A, Cingolani A, Bates M, et al. Relationship between replicative capacity and amino acid substitutions in HIV-1 reverse transcriptase and protease from HAART-failing patients. Third European HIV Drug Resistance Workshop. March 30-April 1, 2005. Athens. Abstract 39. Poster 7.3.
8. Cingolani A, Antinori A, Rizzo MG, et al. Usefulness of monitoring HIV drug resistance and adherence in individuals failing highly active antiretroviral therapy: a randomized study (ARGENTA). AIDS 2002;16:369-379.
9. Tozzi V, Zaccarelli M, Forbici F, et al. Prevalence and risk factors for multi-nucleoside, -non-nucleoside, and —protease resistance in patients failing antiretroviral therapy. Third European HIV Drug Resistance Workshop. March 30-April 1, 2005. Athens. Abstract 72. Poster 9.17.
10. Oette M, Daeumer M, Kaiser R, et al. Long-term efficacy of HIV salvage therapy guided by genotypic resistance testing. Third European HIV Drug Resistance Workshop. March 30-April 1, 2005. Athens. Abstract 71. Poster 9.16.
|