| |
Chronic Hepatitis B & HIV/HBV Coinfection
|
| |
| |
Stephanie D. Straley, PA-C, MS and Norah A. Terrault, MD, MPH
University of California, San Francisco, S357, 513 Parnassus Avenue, San Francisco, CA, 94143-0538, USA.
Current Treatment Options in Gastroenterology 2004, 7:477-489
CONTENTS
Introduction
• NATURAL HISTORY OF CHRONIC HEPATITIS B INFECTION
• ASSESSING DISEASE ACTIVITY AND SEVERITY
Treatment
-Prevention and surveillance
-Diet and lifestyle
-Pharmacologic therapy
-Treatment for special populations
• Patients coinfected with HIV/HBV
• Patients with decompensated liver disease
Emerging pharmacologic therapies
References and Recommended Reading
HIGHLIGHTS OF THIS REPORT:
--It is estimated that 6% to 10% of patients infected with HIV are HBsAg positive. Testing patients for coinfection is even more crucial when considering treatment of HIV or HBV, as drug resistance is a problem in both diseases.
--In the United States, sexual contact and injection drug use are the most common routes of transmission
--Patients with a prolonged phase of active viral replication are at greater risk of liver-related complications than patients with shorter duration of active viral replication
--Cirrhosis is estimated to occur in about 20% of chronically-infected individuals. The risk of cirrhosis and other liver complications increases with the duration of disease and is higher in those with high alcohol intake, and in those who are coinfected with HCV or HIV
--For individuals who are found to be HBsAg-positive, additional testing is needed to determine the level of viral replication, severity of histologic disease, and presence of complications of chronic liver disease (see Table 1). Measurement of ALT and AST levels, total bilirubin; albumin; and prothrombin time plus HBeAg, anti-HBe, HBV DNA levels, and anti-HDV are recommended for the initial assessment of a patient with chronic HBV. In patients with elevated AST and ALT, additional testing to rule out other causes of chronic liver disease is indicated
--The primary goals of anti-HBV treatment are to ameliorate ongoing liver injury and prevent future complications of chronic infection such as decompensated cirrhosis and HCC
--Current therapies available are targeted to patients with persistently elevated liver enzymes (AST and ALT); HBV DNA levels greater than 105 copies/mL [100,000] (if HBeAg positive) or greater than 104 copies/mL [10,000] (if HBeAg negative); or evidence of "active" hepatitis on liver biopsy.
--Drugs available approved for treatment of HBV in the patient infected with HIV include interferon alfa, lamivudine, and adefovir. Tenofovir and emtricitabine are approved only for treatment of HIV infection but have shown activity against HBV. Interferon alfa would be difficult to use in an HIV infected patient, given the frequency of side effects and risk of infection. Interferon alfa is not contraindicated, and it may be an option in patients with well-controlled HIV disease. Interferon is used in the treatment of HCV infection in patients coinfected with HIV
--New drugs: entecavir is in phase III studies & pending FDA approval; FTC. Other nucleoside/nucleotide analogs under investigation include telbivudine and valtorcitabine, clevudine, coviracil and amdoxovir, remofovir, and racivir
ARTICLE TEXT
Opinion Statement
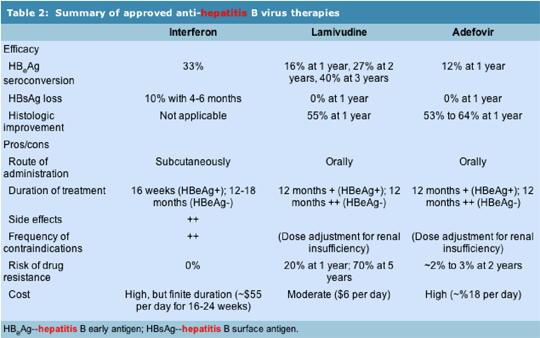
Interferon alpha, lamivudine, and adefovir are the three drugs currently approved for the treatment of chronic hepatitis B virus (HBV). There are pros and cons associated with the use of each drug. Individualization of therapy, based upon factors such as patient comorbidities, response to prior therapies, and stage of disease, is recommended. Patients with abnormal liver enzymes, indices of active viral replication (HBV DNA positive hepatitis B early antigen [HBeAg] positive) or evidence of necroinflammatory activity on liver biopsy, and compensated liver disease are potential candidates for treatment with interferon, lamivudine, or adefovir. Patients with abnormal liver enzymes, indices of active viral replication (HBV DNA positive HBeAg positive), and decompensated liver disease are candidates for treatment with lamivudine or adefovir. Consideration of liver transplantation should occur concurrently. Interferon alpha treatment results in hepatitis B surface antigen [HBeAg] loss and sustained suppression of HBV DNA replication in 30% to 40% of treated patients. Loss of HBsAg occurs in nearly 10% of patients and a higher than expected frequency of HBsAg loss occurs long-term. The main limitation of therapy is the side effects and the need for parenteral administration. Additionally, interferon therapy is not applicable to all patient groups. Lamivudine achieves HBeAg seroconversion in 15% to 20% of patients treated for 12 months, but (HBsAg) loss is rare. Reduction in HBV DNA to undetectable levels (by hybridization assay) during treatment is nearly universal, and histologic improvement is seen in about 55% of patients. The main limitation of lamivudine therapy is the development of drug resistance, which occurs in 20% of patients after 12 months and increases with duration of therapy (55% at 3 years). Adefovir achieves HBeAg seroconversion in 12% of patients treated for 12 months, but HBsAg loss is rare. (note from Jules Levin: 3 years of data on adefovir were reported at AASLD Nov 2004. HBeAg seroconversion was 12% after 1 yr ADV therapy, 29% after 2 yrs, & 43% after 3 yrs. <http://www.natap.org/2004/AASLD/aasld_23.htm>). An average 3.5 log reduction in HBV DNA levels is and histologic improvement occurs in 50% to 60% of patients. It is effective against both wild-type and lamivudine-resistant HBV. The risk of drug resistance is low and estimated to be approximately 2% to 3% after 2 years of treatment. Several new antiviral agents are currently under evaluation in clinical trials. In addition, there are two drugs (tenofovir and emtricitabine) that have been approved for HIV infection and that have anti-HBV activity. In the future, combination therapy for chronic HBV infection can be anticipated. Utilization of two or more anti-HBV drugs would be predicted to enhance efficacy and reduce the likelihood of emergence of drug resistance.
INTRODUCTION
Hepatitis B virus (HBV) is a parenterally transmitted virus that is acquired from exposure to infected blood or body secretions. In the United States, sexual contact and injection drug use are the most common routes of transmission, thus the majority of new cases of hepatitis B occur in adolescents and adults. Perinatal and early childhood infections are much less frequent in the United States, but in countries where HBV is endemic this is an important mode of transmission. The incidence of hepatitis B is declining due to the implementation of HBV vaccination programs and prevention measures aimed at reducing transmission of both HIV and HBV infections. However, a large reservoir of chronically infected individuals still remain. Chronic hepatitis B affects 1.25 million Americans and 350 million individuals worldwide [1]. In the United States, hepatitis B surface antigen [HBsAg] carrier rates are higher in Asian-Americans and African-Americans than in whites, and the highest rates reported are among individuals who have immigrated to the United States from areas of high HBV endemnicity, such as Southeast Asia, Africa, and the Mediterranean area [1, 2].
NATURAL HISTORY OF CHRONIC HEPATITIS B INFECTION
Following an acute infection, the risk of developing chronic HBV varies with the age of the individual at the time of infection. In exposed infants, the risk of chronic infection is 90% or higher, whereas in immunocompetent adults the risk is only 2% to 5%. In terms of risk of future complications, patients with chronic HBV infection can generally be divided into two broad categories, those with "active" liver disease (abnormal serum aminotransferase levels, HBV DNA levels 3 105-6 copies/mL, evidence of chronic necroinflammation on biopsy and presence of hepatitis B early antigen [HBeAg]) and those with "inactive" liver disease (normal serum aminotransferase levels, HBV DNA levels < 105-6 copies/mL, no or minimal necroinflammation on liver biopsy, and HBeAg negative). Patients with "active" disease have the greatest risk of liver-related complications and are the target group for treatment [3].
Seroconversion from HBeAg to anti-HBe, either spontaneously or with treatment, is typically accompanied by a decline in HBV DNA levels (< 105 copies/mL), normalization of liver enzymes (aspartate aminotransferase [AST]/ alanine aminotranferease [ALT]), and resolution of necroinflammatory activity on liver histology. The rate of spontaneous resolution of active replication and seroconversion from HBeAg to anti-HBe is 5% to 20% per year [4, 5]. The rate of HBsAg seroconversion is much less, about 1% per year [5]. There is a subgroup of HBV-infected individuals who are HBeAg-negative but have high levels of HBV DNA, abnormal liver enzymes, and evidence of active histologic disease on biopsy. These patients have a HBV variant virus that does not produce HBeAg, due to the presence of one or more mutations in the precore or core promoter regions of the HBV genome [6, 7]. This variant form of HBV infection is more common in Southern Europe and Southeast Asia than it is in the United States, but HBeAg-negative variants are also found in the United States. In a recent nationwide survey, precore and core promoter variant viruses were detected in 27% and 44% of patients respectively, highlighting the prevalence of HBeAg-negative "active" HBV disease in the United States population [7]. Such patients are at risk for progressive liver disease and are candidates for antiviral therapy despite the absence of HBeAg. Treatment of this subgroup of patients differs from that of HBeAg-positive patients because the risk of relapse following discontinuation of treatment is higher, so consequently longer treatment periods are required.
The outcome of chronic HBV infection is largely dependent upon the severity and duration of active HBV disease. Patients with a prolonged phase of active viral replication are at greater risk of liver-related complications than patients with shorter duration of active viral replication [8, 9]. Cirrhosis is estimated to occur in about 20% of chronically-infected individuals. The risk of cirrhosis and other liver complications increases with the duration of disease and is higher in men than in women, in those with high alcohol intake, and in those who are coinfected with HCV or HIV. In case-control studies, the risk of hepatocellular carcinoma (HCC) is increased five to 15-fold in HBV-infected persons compared with the general population [9]. Patients with cirrhosis are at higher risk of cancer than those without cirrhosis; 70% to 90% of cases of HBV-associated cancers occur in persons with cirrhosis [10].
|
|
| |
| |
 |
|
| |
| |
*Surveillance tests for hepatocellular carcinoma include ultrasonography (or other liver imaging study) AFP. †Measured if changes in liver enzymes occur, suggesting spontaneous seroconversion or increased disease activity. Several quantitative tests to measure HBV DNA are available, but none are standardized, thus the results from one assay are not comparable with another. When following an individual patient, the use of the same HBV DNA quantitative tests is most informative. ¶Ultrasonography is the most common form of liver imaging study used. AFP--a-fetoprotein; ALT--alanine aminotranferase; AST--aspartate aminotranferase; HBeAg--hepatitis B early antigen; PT--prothrombin time.
ASSESSING DISEASE ACTIVITY AND SEVERITY
For individuals who are found to be HBsAg-positive, additional testing is needed to determine the level of viral replication, severity of histologic disease, and presence of complications of chronic liver disease (Table 1). Measurement of ALT and AST levels, total bilirubin; albumin; and prothrombin time plus HBeAg, anti-HBe, HBV DNA levels, and anti-HDV are recommended for the initial assessment of a patient with chronic HBV. In patients with elevated AST and ALT, additional testing to rule out other causes of chronic liver disease is indicated. An ultrasound of the abdomen is used to assess for radiologic signs of cirrhosis and portal hypertension. A liver biopsy provides the most accurate means of assessing the severity of disease and can be used to determine the need for antiviral therapy and to rule out concomitant diseases, such as nonalcoholic fatty liver disease [11]. However, a liver biopsy is not required before initiating antiviral therapy.
Patients with abnormal AST and ALT levels or evidence of necroinflammatory activity on liver biopsy are potential candidates for antiviral therapy [3, 12]. Patients with persistently normal liver enzymes are not recommended for treatment with the agents that are currently available. Because disease activity can fluctuate over time, patients with normal liver enzymes should be monitored on a regular basis (every 6 months at a minimum) for changes in disease activity. If liver enzymes become abnormal for a period of at least 3 months, patients should be considered for instituting antiviral treatment.
Patients with decompensated liver disease are potential candidates for treatment with lamivudine or adefovir but not interferon. Liver transplantation referral should be considered concurrently.
Treatment
Prevention and surveillance
• Patients need to be counseled regarding the prevention risk of transmitting HBV to others, especially sexual partners and household contacts. All sexual partners and members of the patient's household should be tested for HBV and vaccinated if they do not have evidence of prior HBV exposure.
• The Centers for Disease Control recommends vaccination against hepatitis A virus for all individuals with chronic liver disease.
• The risk of HCC is increased among patients with chronic HBV infection, especially those with cirrhosis. Older age, male gender and a family history of HCC predict higher risk of cancer. Most clinicians recommend surveillance for HCC using ultrasound with or without alpha-fetoprotein every 6 months for patients with cirrhosis, patients older than 40 years of age, and those with a family history of HCC [11]. Whether surveillance programs reduce the mortality rates from HCC is controversial [13, 14]. Prospective evaluation of currently alternative screening strategies is needed and ongoing [13, 14].
• Hepatitis B early antigen status and high HBV DNA levels have been correlated with higher risk of HCC [9]. However, no prospective study has determined that treatment for patients who are HBeAg positive or have high levels of HBV DNA levels (3 105-6) with normal liver enzymes and minimal histologic disease reduces their risk of HCC. Thus, treatment of patients with "inactive disease" but with HBeAg or elevated HBV DNA levels is not recommended for the prevention of HCC at this time.
|
|
| |
| |
 |
|
| |
| |
Diet and lifestyle
• Heavy alcohol use (typically defined as more than 5 to 8 units/d for at least 5 years) is associated with a high frequency of cirrhosis and liver cancer [15, 16]. The effect of lesser amounts of alcohol has not been carefully studied, but abstinence or minimal alcohol intake is recommended.
• Smoking has been associated with an increased risk of HCC in some, but not all, studies of patients with chronic HBV disease. The effects of smoking and alcohol may be synergistic [17].
• No dietary factors have been linked specifically to HBVdisease activity or severity. However, excess iron is associated with reduced responsiveness to interferon therapy and it has been linked to increased risk of HCC [18]. Thus, iron supplements should be avoided unless iron deficiency is present. Vitamin A in excessive amounts can be directly hepatotoxic and use of supplementation is not recommended unless deficiency is documented.
• Herbal supplements should be used with caution, if at all. Many of these preparations can be hepatotoxic and can cause severe liver injury. Examples of hepatotoxic herbal products associated with acute or chronic hepatitis include kava kava, chaparral, comfrey, germander, and jin bu huan. Due to a lack of regulation of supplements, formulations and doses can vary widely.
• Patients who have coexistent nonalcoholic fatty liver disease should be simultaneously counseled on optimizing weight, achieving and maintaining normal triglyceride levels, and controlling diabetes mellitus.
(Drug) Pharmacologic therapy
• The primary goals of anti-HBV treatment are to ameliorate ongoing liver injury and prevent future complications of chronic infection such as decompensated cirrhosis and HCC. Treatment guidelines have been recently published [3, 19, 20*, 21*] (Table 2). Eradication of infection is also an important goal but is achieved much less frequently with therapies that are currently available. Responses to treatment are typically defined by one or more of the following endpoints: reduction in inflammatory activity and fibrosis (histologic response); normalization of aminotransferase levels (biochemical response); sustained suppression of HBV DNA replication (virologic response); or seroconversion of HBeAg to anti-HBe.
• Current therapies available are targeted to patients with persistently elevated liver enzymes (AST and ALT); HBV DNA levels greater than 105 copies/mL [100,000] (if HBeAg positive) or greater than 104 copies/mL [10,000] (if HBeAg negative); or evidence of "active" hepatitis on liver biopsy.
• Although a liver biopsy is not essential for the initiation of treatment of patients with abnormal ALT levels, the severity of disease on liver biopsy may influence the choice and duration of therapy.
Interferon alpha
(FDA & European regulatory approval for Pegasys, pegylated interferon, is pending. Peginterferon will likely replace standard interferon as the preferred choice of interferon therapy.) At AASLD Nov 2004, researchers reported study data on Pegasys for HBV: http://www.natap.org/2004/AASLD/aasld_11.htm
Interferon alfa (IFN-alfa) has both immunomodulatory and antiviral effects. In HBeAg-positive patients, IFN-alfa increases the rate of sustained loss of HBeAg by 20% when compared with untreated subjects. Treated patients also have a greater sustained loss suppression of HBV DNA (37% vs 17%) and loss of HBsAg (8% vs 1.8%) compared with untreated patients [5]. The two factors most consistently associated with IFN responsiveness are low baseline HBV DNA levels and high baseline aminotransferase activity (³5 times upper limit of normal). Long-term follow-up of interferon-treated patients indicates that remission is maintained in the majority, and late HBsAg seroconversion occurs at a higher rate than untreated patients. Histologic progression of disease is slowed in responders and rates of HCC and liver-related death appear to be reduced. In patients with HBeAg-negative infection (HBV precore variant), a longer duration of treatment is needed, as relapse following discontinuation of treatment is high. The rates of sustained virologic response with 12 months of IFN-alfa treatment is 22% versus 11% in those treated for only 6 months [22]. In a recent uncontrolled study treating HBeAg- negative patients with active disease for 24 months, a sustained virologic response was achieved in 30%, and among responders liver disease did not progress [23].
Interferon alpha requires subcutaneous administration. Side effects are common, but discontinuation of treatment due to side effects is infrequent [24]. These are the main disadvantages of interferon therapy. Advantages include the finite period of treatment (16 or 32 weeks in HBeAg-positive patients) and the ability to achieve HBsAg seroconversion. Additionally, treatment is not associated with any known risk of resistance mutations.
Standard dosage
(note: pegylated interferon is a once weekly subcutaneous injection and will likely be the preferred choice of treatment replacing standard interferon. Pegasys dose is pre-fixed and will likely)
For HBeAg-positive patients, HBV DNA levels greater than or equal to 105 copies/mL (100,000) in adults, the dose is 10 million units (MU) subcutaneous (SC) three times weekly or 5 MU daily for 16 weeks. An additional 16 weeks of IFN treatment is recommended for those patients who remain HBeAg positive but have low HBV DNA titers (< 10 pg/mL) at the end of the initial 16 weeks of IFN therapy. For HBeAg-negative patients, HBV DNA levels ³104 copies/mL (10,000) adults (HBeAg-negative variants) the dose is 10 million units (MU) SC three times weekly or 5 MU daily for 48 weeks. An additional 48 weeks of IFN therapy increases the response rates by approximately 10%. For HBeAg-poitive patients, HBV DNA-positive children 2 years of age or older, 6 MU/m2 SC three times weekly for 16 weeks.
Contraindications
Absolute: decompensated liver disease (elevated bilirubin or prothrombin time, low albumin, history of ascites, encephalopathy, or variceal bleeding); uncontrolled psychiatric disease; pregnancy (category C) or breastfeeding, significant leukopenia or thrombocytopenia significant comorbid diseases such as unstable coronary artery disease, uncontrolled seizure disorder, diabetes, or hypertension. Relative: autoimmune diseases (psoriasis, rheumatoid arthritis); prior history of depression or psychiatric illness.
Main drug interactions
Caution is needed in patients on other myelosuppressive drugs. IFN reduces the clearance of theophylline.
Main side effects
Flu-like symptoms including fatigue, headache, myalgias, anorexia, nausea, and fever are frequent in first week of treatment but decrease in severity with continued administration. Alopecia, leukopenia, and thrombocytopenia are common. Less common but more serious side effects are neuropsychiatric disturbances, thyroid dysfunction, and bacterial infections (related to leukopenia). Rare side effects include retinopathy, pulmonary fibrosis, and renal dysfunction. Dose reductions due to adverse effects occur in approximately 20% of patients but serious adverse effects requiring drug discontinuation occur in less than 10% [24].
Special points
Administration of IFN-a at bedtime may reduce the frequency of the flu-like symptoms. Anti-pyretics (eg, acetaminophen, 325 to 650 mg preinjection) reduces fever, myalgias, and headaches. Baseline testing: complete blood count (CBC) with platelets, AST, ALT, total bilirubin, glucose, urea, creatinine, thyroid-stimulating hormone (TSH), antinuclear antibodies, and rheumatoid factor. Electrocardiogram if over 50 years of age and no prior infection. Monitoring during treatment: CBC with differential weekly for first 2 to 4 weeks, then monthly; liver and renal function tests monthly; TSH at week 12 every 3 months.
Cost effectiveness
Treatment of persons with chronic HBV carriers (HBsAg and HBeAg positive) with chronic hepatitis (without cirrhosis) using interferon alpha (16-week course of 10 MU three times weekly) is associated with increased life expectancy and quality-adjusted life-expectancy, as well as reduced total health care costs compared with the standard management of chronic liver disease without interferon treatment [25, 26]. Cost-effectiveness analyses comparing lamivudine versus interferon with adefovir for the treatment of chronic HBV have not been performed.
Lamivudine
Lamivudine, a cytosine analogue, is orally administered and well tolerated, even in patients with advanced liver disease. With a treatment period of 12 months, seroconversion from HBeAg to anti-HBe occurs in 16% to 18%, and treatment response correlates with reduced inflammatory indices on liver biopsy and decreased rates of fibrosis progression [27]. The duration of lamivudine treatment may be important in determining the durability of the response when treatment is stopped. The most important predictor of response is high baseline aminotransferase levels (³ 5 times upper limits of normal) [28]. The rate of HBeAg seroconversion increases with duration of therapy, with rates of 27% and 40% after 2 and 3 years of lamivudine treatment. HBeAg seroconversion was more frequent in those achieving HBV DNA levels below 104 copies/mL. Reemergence of HBV DNA while on drug therapy (lamivudine resistance) occurs in approximately 15% to 20% of treated patients after 12 months of treatment. Treatment breakthrough among compliant patients is associated with mutations in the YMDD locus of domain C of the polymerase and decreased sensitivity to lamivudine in vitro. The risk of treatment breakthrough increases with time, reaching 55% after 3 years of lamivudine therapy [29]. Clinically, treatment breakthrough is characterized by increasing HBV DNA levels and abnormal AST and ALT. Deterioration in liver function and worsening clinical status are rare with treatment breakthrough, unless the patient has underlying cirrhosis or advanced liver disease [30]. Factors associated with emergence of lamivudine resistance are high baseline HBV DNA levels, persistent HBV DNA levels over 200 copies/mL during treatment, and prolonged treatment duration [29, 31].
As with IFN, sustained response rates differ in HBeAg-positive and HBeAg-negative patients. The rate of relapse after discontinuation of lamivudine at 12 months of treatment is much higher in HBeAg-negative patients (90%) compared with HBeAg-positive patients (20% to 30%) [32, 33]. Continued treatment beyond 12 months is limited by an increasing rate of HBV resistance and virologic breakthrough. Thus, the optimal duration of treatment for patients with the HBeAg-negative variant form of chronic active HBV infection has not been defined.
The advantages of lamivudine include an oral route of administration and excellent tolerability. The disadvantages of lamivudine are lower rates of HBeAg and HBsAg seroconversion (with 12 months treatment) than with interferon, the need for long-term therapy to increase seroconversion rates, and the risk of viral resistance that accompanies prolonged therapy. Although there may be few serious clinical consequences associated with the development of resistant HBV infection in the short-term, the long-term consequences are not fully known.
Standard dosage
100 mg orally once daily. The optimal duration of treatment has not been defined, but the usual treatment period is 12 months or longer.
Contraindications
Pregnancy category C. Dose reduction for renal insufficiency (creatinine clearance (CrCl) < 50 mL/min). If CrCl is 30 to 49 mL/min, first dose should be 100 mg, then 50 mg daily; if CrCl is 15 to 29 mL/min, first dose should be 100 mg, then 25 mg daily. If CrCl is 5 to14 mL/min, first dose should be 35 mg, then 15 mg daily, and if CrCl is less than 10 mL/min, first dose should be 35 mg, then 10 mg daily. A liquid formulation is available.
Main drug interactions
Zalcitabine (ddC) can inhibit intracellular phosphorylation of lamivudine, therefore, concurrent use is not recommended. Trimethoprim/sulfamethoxazole has been shown to increase levels of lamivudine. However, dose adjustments are not recommended for either drug.
Main side effects
Fatigue, nausea, and headache are most commonly reported, although infrequent. Elevated serum aminotransferase levels are seen in 25% of patients during treatment or in the 6 months following discontinuation of therapy. This "flare" in disease activity may be indicative of seroconversion from HBeAg to anti-HBe. Asymptomatic elevations of serum amylase levels are seen, but pancreatitis is exceedingly rare.
Special points
Patients should undergo HIV testing pretreatment to avoid the inadvertent induction of lamivudine-resistant HIV. Patients coinfected with HIV/HBV should be on antiretroviral therapy prior to or have antiretroviral therapy started concurrently with initiation of lamivudine treatment for HBV. A higher dose of lamivudine (150 mg twice daily) is used in patients coinfected with HIV/HBV.
Cost effectiveness
Cost of lamivudine for 12 months is less than IFN for 16 weeks but there is lower rate of seroconversion (16% vs 33% after 12 months therapy) and the durability of the response appears to be less. Formal cost-effective analyses have not been conducted. Lamivudine, 100 mg daily for 12 months is less expensive than adefovir, 10 mg daily for 12 months, but rates of drug resistance are higher for lamivudine than adefovir.
Adefovir dipivoxil
Adefovir dipivoxil, an adenosine analogue, is orally administered and well tolerated in patients with compensated and decompensated disease. Adefovir is approved for the treatment of patients with wild-type and lamivudine-resistant chronic HBV infection. Adefovir, 10 mg daily for 12 months, results in histologic improvement in 53% to 64% of patients compared with 25% to 35% of placebo-treated patients and normalization of liver enzymes in 48% to 72% versus 16% to 29% of placebo-treated patients. HBeAg seroconversion is seen in 12% of patients after 12 months of treatment (note from Jules Levin: as mentioned above, 3 years data was reported at AASLD Nov 2004 showing 43% rate of HBeAg seroconversion). As with lamivudine, pretreatment ALT level is the best predictor of treatment response. Patients with high ALT levels and low HBV DNA levels are most likely to respond [34*, 35*]. Additionally, a rapid rate of decline of HBV DNA during treatment has been associated with higher rates of seroconversion. Adefovir is nephrotoxic at higher doses (60 and 120 mg) as demonstrated in prior HIV clinical trials. However, at the approved dose of 10 mg used in HBV treatment, no significant changes in serum creatinine have been observed in up to 109 weeks of treatment [36]. A novel mutation in the HBV polymerase conferring resistance to adefovir dipivoxil has been identified [37]; this mutant virus appears to be sensitive to lamivudine. The incidence of adefovir resistance is low, with 2% developing the mutation and clinical resistance after 2 years of treatment [38*].
Standard dosage
10 mg orally once daily. The optimal duration of therapy is at least 12 months.
Contraindications
Pregnancy category C. Dose reduction recommended for patients with a CrCl less than 50 mL/min. If CrCl is 20 to 49 mL/min the adefovir dosage is 10 mg every 48 hours; if CrCl is 10 to19 mL/min the dosage is 10 mg every 72 hours. If patient is on hemodialysis the dose is 10 mg every 7 days following dialysis.
Main drug interactions
Ibuprofen, 800 mg three times daily and increased adefovir exposure by approximately 23%. The clinical significance of this increase is not known.
Main side effects
Asthenia, headache, and abdominal pain are the most common side effects reported. Hypophosphatemia and a decrease in free L-carnitine levels can occur, and supplementation should be provided if deficiencies are identified.
Special points
Whenever possible, HIV/HBV coinfected patients should be on antiretroviral therapy prior to initiation of anti-HBV therapy with adefovir. Because adefovir has anti-HIV activity, treatment with adefovir may theoretically lead to emergence of HIV resistance in unrecognized or untreated HIV infection. Although preliminary data on the risk of adefovir-resistant HIV in patients receiving adefovir for HBV infection suggests that the dose of adefovir used in HBV treatment (10 mg daily) is too low to induce HIV resistance, the available data are limited and caution is urged.
Cost effectiveness
Cost of adefovir for 12 months is less than IFN but there is lower rate of seroconversion (12% vs 33% after 12 months of therapy), and the impact on risk of cirrhosis and HCC has not been established. Adefovir is more costly than lamivudine. Formal cost-effective analyses have not been conducted.
Adefovir + FTC
At the AASLD Nov 2004 conference researchers reported initial findings from studying FTC+adefovir combination therapy. Here is link to report finding added reduction in HBV DNA:
http://www.natap.org/2004/AASLD/aasld_26.htm
Treatment for special populations
-Patients coinfected with HIV/HBV
• Treatment of patients infected with HBV in HIV has become more important with the emergence of highly active anti-retroviral therapy (HAART). Death from opportunistic infections in patients with HIV has declined whereas mortality from liver disease (primary HBV and HCV) has increased dramatically. Because HBV and HIV share common modes of transmission, all patients with HIV should be screened for HBV and vice versa. It is estimated that 6% to 10% of patients infected with HIV are HBsAg positive. Testing patients for coinfection is even more crucial when considering treatment of HIV or HBV, as drug resistance is a problem in both diseases.
Note from Jules Levin: tenofovir is undergoing study for HBV treatment. Initial studies find tenofovir may reduce HBV DNA more than adefovir.
• Drugs available approved for treatment of HBV in the patient infected with HIV include interferon alfa, lamivudine, and adefovir. Tenofovir and emtricitabine are approved only for treatment of HIV infection but have shown activity against HBV. Interferon alfa would be difficult to use in an HIV infected patient, given the frequency of side effects and risk of infection. Interferon alfa is not contraindicated, and it may be an option in patients with well-controlled HIV disease. Interferon is used in the treatment of HCV infection in patients coinfected with HIV. The most efficient way to treat HBV in patients coinfected with HIV/HBV is with the inclusion of a nucleoside analogue with activity against HBV in the HAART regimen. Lamivudine was the first nucleoside analogue available with activity against HBV and HIV. Dosing for the HIV patient is 150 mg twice daily, versus 100 mg daily for the patient without HIV. Lamivudine resistance is more common in coinfected patients, up to 90% at 4 years. Thus, any patient with HIV/HBV on lamivudine as part of the HAART regimen should be monitored closely for the development of resistance by measuring AST and ALT every 3 months. If liver enzymes increase, HBV DNA levels should be checked. An increasing HBV viral load in association with elevated AST/ALT levels in a compliant patient is indicative of lamivudine resistance. Once resistance is documented, a nucleoside analogue effective against lamivudine-resistant infection should be added.
• Adefovir and tenofovir are currently available and are effective against lamivudine-resistant infection. Adefovir at the 10-mg dose is not effective against HIV. A small study in HIV/HBV coinfected patients with wild-type and lamivudine-resistant HBV found a 4.7 to 5.3 log drop in HBV DNA levels after 48 weeks of treatment, with no difference in response between patients with lamivudine-resistant versus wild-type HBV infection [39]. The drug was well tolerated. Limited data in patients with lamivudine-naïve and lamivudine-resistant HBV infection without HIV coinfection indicate tenofovir may result in reductions in HBV DNA and ALT levels similar to that seen with adefovir [40]. Emtricitabine is not effective against lamivudine-resistant infection, but is useful in patients with wild-type HBV and it may have higher rates of HBeAg seroconversion rate and lower resistance rates of resistance than lamivudine [41]. It would be reasonable to consider use of emtricitabine in a patient with HIV/HBV who is lamivudine-naïve. There are no data available on the effect of combination tenofovir-emtricitabine on HBV replication in patients coinfected with HIV.
• Hepatologists should work closely with the provider who is managing the patient's HIV disease to determine the best HAART regimen and monitoring plan for the patient coinfected with HIV/HBV.
-Patients with decompensated liver disease
• A decline in liver synthetic function (elevated bilirubin or prothrombin time, low albumin) or symptoms of decompensation such as fluid retention, encephalopathy, or variceal bleeding should prompt consideration of liver transplantation in patients with chronic HBV infection. In patients with elevated liver enzymes and elevated HBV DNA levels (usually ³105 copies/mL [100,000]), treatment with lamivudine or adefovir is indicated. Studies have shown that lamivudine or adefovir are safe in patients with decompensated liver disease and treatment is frequently associated with significant clinical improvement. Studies have shown that the time for transplantation can be delayed with lamivudine and adefovir treatment, but 6 months or more are usually needed to obtain the desired clinical benefits. [30, 42, 43, 44]. Patients with decompensated liver disease require long-term, possibly indefinite, treatment, as virologic relapse after discontinuation of therapy can be associated with a rapid clinical deterioration. However, prolonged treatment is associated with an increased rate of drug resistance, and treatment breakthrough can led to worsening liver function and decompensation in some patients [30]. Thus, close monitoring for drug resistance is recommended. For patients with treatment breakthrough on lamivudine, adefovir results in inhibition of viral replication and improvement in synthetic function in most cases [43, 44]. Thus, adefovir is the treatment of choice for patients with decompensated liver disease and lamivudine-resistant HBV infection. Interferon is contraindicated in patients with decompensated liver disease, as serious bacterial infections and deterioration in liver disease have occurred during treatment [45].
• Liver transplantation is the treatment of choice for patients with liver failure due to chronic HBV infection, although the early experience with liver transplantation in patients infected with hepatitis B was discouraging due to graft loss resulting from recurrent HBV cirrhosis. In the past decade, effective therapies to prevent and treat HBV infection in liver transplant recipients have been identified [46]. Therapies such as hepatitis B immunoglobulin (HBIg), lamivudine, and adefovir have resulted in reduced rates of recurrent HBV infection and prevented disease progression in those with recurrent HBV disease posttransplantation.
Emerging (drug) pharmacologic therapies
• Published studies have not found a consistent advantage of combination therapy with lamivudine and interferon (standard or pegylated) over interferon monotherapy but the combination does appear to be superior to lamivudine monotherapy [47, 48, 49, 50, 51]. Only one study compared combination therapy with both interferon and lamivudine monotherapy [47, 51], and the per protocol results suggested an advantage of combination therapy, primarily in those with moderately elevated liver enzymes (ALT 2 to 5 times upper limit of normal). Because differences in outcomes between these studies may relate to the differences in treatment protocols (timing and duration of lamivudine and interferon treatment) or the patient populations studied, the specific place for utilization of interferon and lamivudine combination therapy is still evolving.
(note from Jules Levin:
REMAINDER OF ARTICLE RESUMES FOLLOWING THIS ABSTRACT ON COMBINATION THERAPY. In the other study [51] no benefit for combination therapy was found but the study was conducted in HBeAg-negative patients.
[47] Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: a randomised trial.
Gut • 2000 Apr;46(4):562-8
Abstract
BACKGROUND, AIM, AND METHODS: Alpha interferon is the generally approved therapy for HBe antigen positive patients with chronic hepatitis B, but its efficacy is limited. Lamivudine is a new oral nucleoside analogue which potently inhibits hepatitis B virus (HBV) DNA replication. To investigate the possibility of an additive effect of interferon-lamivudine combination therapy compared with interferon or lamivudine monotherapy, we conducted a randomised controlled trial in 230 predominantly Caucasian patients with hepatitis B e antigen (HBeAg) and HBV DNA positive chronic hepatitis B. Previously untreated patients were randomised to receive: combination therapy of lamivudine 100 mg daily with alpha interferon 10 million units three times weekly for 16 weeks after pretreatment with lamivudine for eight weeks (n=75); alpha interferon 10 million units three times weekly for 16 weeks (n=69); or lamivudine 100 mg daily for 52 weeks (n=82). The primary efficacy end point was the HBeAg seroconversion rate at week 52 (loss of HBeAg, development of antibodies to HBeAg and undetectable HBV DNA). RESULTS: The HBeAg seroconversion rate at week 52 was 29% for the combination therapy, 19% for interferon monotherapy, and 18% for lamivudine monotherapy (p=0.12 and p=0.10, respectively, for comparison of the combination therapy with interferon or lamivudine monotherapy). The HBeAg seroconversion rates at week 52 for the combination therapy and lamivudine monotherapy were significantly different in the per protocol analysis (36% (20/56) v 19% (13/70), respectively; p=0.02). The effect of combining lamivudine and interferon appeared to be most useful in patients with moderately elevated alanine aminotransferase levels at baseline. Adverse events with the combination therapy were similar to interferon monotherapy; patients receiving lamivudine monotherapy had significantly fewer adverse events. CONCLUSIONS: HBeAg seroconversion rates at one year were similar for lamivudine monotherapy (52 weeks) and standard alpha interferon therapy (16 weeks). The combination of lamivudine and interferon appeared to increase the HBeAg seroconversion rate, particularly in patients with moderately elevated baseline aminotransferase levels. The potential benefit of combining lamivudine and interferon should be investigated further in studies with different regimens of combination therapy.)
• Combining two or more nucleoside analogs would be predicted to increase efficacy and decrease viral resistance. However, studies of lamivudine plus adefovir have not been shown to be more effective than treatment with adefovir alone, although study treatment periods beyond 48 weeks have not been studied [52*].
At the AASLD Nov 2004, several entecavir studies were reported including phase III studies in 3TC-experienced patients:http://www.natap.org/2004/AASLD/aasld_09.htmHBeAG+ treatment-naïve patients:http://www.natap.org/2004/AASLD/aasld_02.htmHBeAg- treatment-naïve patients:http://www.natap.org/2004/AASLD/aasld_05.htm
• Entecavir, an orally administered guanine nucleoside analogue, produces a 2 to 3 log reduction of HBV DNA titers with short-term treatment. In a study of 169 patients, undetectable HBV DNA levels (by Quantiplex bDNA assay) were achieved in 84% of entecavir (0.5 mg) treated patients versus 58% of lamivudine-treated patients [53]. HBeAg seroconversion was rare. Reported side effects included headache, dizziness, and photophobia, suggesting mild neurotoxicity. Entecavir has been found to be effective in patients with lamivudine-resistant HBV and has a safety profile similar to lamivudine. No resistance has been seen in patients treated for 1 year with the 1-mg dose [54, 55]. The results of completed phase III trials are anticipated in 2004.
• (note from Jules Levin: as referred to above, regulatory approval in the USA & Europe is pending for Pegasys use in treating HBV; there is a link above to the study). Pegylated interferons are modified interferon alfa molecules covalently bound to polyethylene glycol molecules that produce more sustained absorption and reduced clearance compared with unmodified IFN-alfa. The drug's half-life is increased to allow for once weekly dosing. Although the efficacy of pegylated interferons is well established for chronic HCV, they are still under study for chronic HBV. One study comparing interferon alfa-2a (4.5 MU three times weekly) with varying doses of pegylated interferon alfa-2a (Pegasys) (90, 180, and 270 mg) for 24 weeks found that pegylated interferon (all groups combined) was more effective than traditional interferon (at the doses used) in reducing HBV DNA levels and achieving HBeAg seroconversion [56*].
• The use of pegylated interferon rather than standard interferon is increasing in in clinical practice, but optimal doses of pegylated interferon for HBV infection need to be defined.Tenofovir disoproxil fumarate is a nucleotide analog of adenosine and is approved by the US Food and Drug Administration for the treatment of HIV; it has in vitro activity against lamivudine-resistant HBV [57]. Small clinical studies of HIV-HBV-coinfected and HBV-monoinfected patients with lamivudine resistant HBV infection found that tenofovir resulted in a 4 to 5 log drop in HBV DNA levels after 24 to 48 weeks [57, 58, 59]. Long-term data on the HBeAg seroconversion rates, histologic benefits, and safety and risk of resistance with tenofovir have not been reported.
At the AASLD Nov 2004 conference researchers reported study results of FTC in chronoc HBV+ patients:http://www.natap.org/2004/AASLD/aasld_14.htm
• Emtricitabine (FTC), the 5-fluorinated derivative of lamivudine, produces a greater than 3 log reduction in HBV DNA with short-term treatment [50]. Although not approved for HBV treatment, FTC is currently available for HIV infection. In a study of 98 nucleoside analogue naïve patients with chronic HBV infection, 29% achieved HBeAg seroconversion rate after 2 years of therapy, with resistance developing in 19% during this same period of treatment, indicating FTC may be more effective than lamivudine at inducing HBeAg seroconversion with a lower rate of resistance [41]; the drug was well tolerated. Cross-resistance between emtricitabine and lamivudine has been documented and, therefore, FTC would not be effective in treating lamivudine-resistant HBV infection.
• Other nucleoside/nucleotide analogs under investigation include telbivudine and valtorcitabine (Idenix Pharmaceuticals, Cambridge, MA), clevudine (Triangle Pharmaceuticals, Durham, NC), coviracil and amdoxovir (Gilead Pharmaceutials, Foster City, CA), remofovir (Valeant Pharmaceuticals, Costa Mesta, CA), and racivir (Pharmasset, Tucker, GA). Other novel therapies being studied include monoclonal antibodies (XTL Biopharmaceuticals, Rehovot, Israel), ribozymes (Ribozyme Pharmaceuticals, Boulder, CO), and immune stimulants (SciClone, San Mateo, CA).
References and Recommended Reading
Recently published papers of particular interest have been highlighted as:
* Of importance
** Of major importance
1.McQuillan G, et al.: Prevalence of hepatitis B virus infection in the United States: the National Health and Nutrition Examination Surveys, 1976 through 1994. Am J Public Health 1999, 89:14-18.
2.Tong MJ: Hepatitis B virus infection in Asian Americans. Gastroenterology Clinics of North America 1994, 23:569-579.
3.Lok A: Chronic Hepatitis B--AASLD Practice Guidelines. Hepatology 2001, 34:1225-1241.
4.Hoofnagle J, et al.: Seroconversion from hepatitis B e antigen to antibody in chronic type B hepatitis. Ann Intern Med 1981, 94:744-748.
5.Wong D, et al.: Effect of alpha-interferon treatment in patients with hepatitis B e antigen-positive chronic hepatitis B. A meta-analysis. Ann Intern Med 1993, 119:312-323.
6.Brunetto M, et al.: A new hepatitis B virus strain in patients with severe anti-HBe positive chronic hepatitis B. J Hepatol 1990, 10:258-261.
7.Chu C, et al.: Prevalence of HBV precore/core promoter variants in the United States. Hepatology 2003, 38:619-628.
8.De Jongh FE, et al.: Survival and prognostic indicators in hepatitis B surface antigen-positive cirrhosis of the liver. Gastroenterology 1992, 103:1630-1635.
9.Yang HI LS, et al.: Hepatitis B e antigen and the risk of hepatocellular carcinoma. N Eng J Med 2002, 347:168-174.
10.Beasley R: Hepatitis B. The major etiology of hepatocellular carcinoma. Cancer 1988, 61:1942-1956.
11.Lok A: Management of hepatitis B: 2000 - Summary of a Workshop. Gastroenterology 2001, 120:1828-1853.
12.Canadian Association for the Study of the Liver: Management of Viral Hepatitis: Recommended Guidelines for Physicians. Canadian Association for the Study of the Liver 2000, 1999. Ottawa, Ontario.
13.Sherman M: Screening for hepatocellular carcinoma in chronic carriers of hepatitis B virus: Incidence and prevalence of hepatocellular carcinoma in a North American urban population. Hepatology 1995, 22:432-438.
14.McMahon B, et al.: Screening for hepatocellular carcinoma in Alaska natives infected with chronic hepatitis B: a 16-year population-based study. Hepatology 2000, 32:842-846.
15.Frieden T, et al.: Chronic liver disease in central Harlem: the role of alcohol and viral hepatitis. Hepatology 1999, 29:883-888.
16.Donato F, et al.: Hepatitis B and C virus infection, alcohol drinking, and hepatocellular carcinoma: a case-control study in Italy. Brescia HCC Study. Hepatology 1997, 26:579-584.
17.Yu M, et al.: Prospective study of hepatocellular carcinoma and liver cirrhosis in asymptomatic chronic hepatitis B virus carriers. Am J Epidemiol 1997, 145:1039-1047.
18.Mandishona E, et al.: Dietary iron overload as a risk factor for hepatocellular carcinoma in Black Africans. Hepatology 1998, 27:1563-1566.
19.Lok ASK: Chronic hepatitis B. AASLD Practice Guidelines, Hepatology, 2001, 1225-1241.
20.* Keeffe E, et al.: A treatment algorithm for the management of chronic hepatitis B virus infection in the U.S. Clin Gastroenterol Hepatol 2004, 2:87-106.
Current treatment algorithms from a panel of hepatology experts are provided in this comprehensive review article.
21.* Lok AS: Practice Guidelines Committee, American Association for the Study of Liver Diseases (AASLD). Chronic hepatitis B: update of recommendations. Hepatology 2004, 39:857-861.
Treatment practice guidelines were initially published by the AASLD in 2001 and were updated in 2004 to include new therapies and information on management of drug-resistant HBV.
22.Manesis E: Interferon alpha treatment and retreatment of hepatitis B e antigen-negative chronic hepatitis B. Gastroenterology 2001, 121:101-109.
23.Lampertico P, et al.: Long-term suppression of hepatitis B e antigen-negative chronic hepatitis B by 24-month interferon therapy. Hepatology 2003, 37:756-763.
24.Renault P: Side effects of alpha interferon. Sem Liver Dis 1989, 9:273-277.
25.Wong JB, et al.: Cost-effectiveness of interferon-alpha 2b treatment for hepatitis B e antigen-positive hepatitis B. Ann Intern Med 1995, 122:664-675.
26.Dusheiko G: Treatment of chronic type B and C hepatitis with interferon alfa: an economic appraisal. Hepatology 1995, 22:1863-1873.
27.Dienstag J, et al.: Lamivudine as initial treatment for chronic hepatitis B in the United States. N Engl J Med 1999, 341:1256-1263.
28.Perrillo R, et al.: Predictors of HBeAg loss after lamivudine treatment for chronic hepatitis B. Hepatology 2002, 36:186-194.
29.Yuen MF, et al.: Factors associated with hepatitis B virus DNA breakthrough in patients receiving prolonged lamivudine therapy. Hepatology 2001, 34:785-791.
30.Perrillo R, et al.: A multicenter United States-Canadian trial to assess lamivudine monotherapy before and after liver transplantation for chronic hepatitis B. Hepatology 2001, 33:424-432.
31.Allen M, et al.: Identification and characterization of mutations in hepatitis B virus resistant to lamivudine. Hepatology 1998, 27:1670-1677.
32.Santantonio T, et al.: Long-term follow-up of patients with anti-HBe/HBV DNA-positive chronic hepatitis B treated for 12 months with lamivudine. Hepatology 2000, 32:300-306.
33.Hadziyannis S, et al.: Efficacy of long-term lamivudine monotherapy in patients with hepatitis B e antigen-negative chronic hepatitis B. Hepatology 2000, 32:847-851.
34.* Marcellin P, et al.: Adefovir dipivoxil for the treatment of hepatitis B e antigen positive chronic hepatitis B. New England Journal of Medicine 2003, 348:808-816.
Clinical trial that established the treatment of 48 weeks of adefovir in patients with HBeAg-positive and HBeAg negative chronic HBV infection. Adefovir was shown to be safe in doses of 10 mg daily and effective in suppressing HBV DNA levels in the majority of treated patients.
35.* Hadziyannis S, et al.: Adefovir dipivoxil for the treatment of hepatitis B e antigen negative chronic hepatitis B. N Engl J Med 2003, 348:800-807.
Landmark clinical trial that established the efficacy of 48 weeks of adefovir in patients with HBeAg-positive and HBeAg negative chronic HBV infection, respectively. The drug was shown to be safe in doses of 10 mg daily and effective in suppressing HBV DNA levels in the majority of treated patients.
36.Tong M, et al.: Long-term safety beyond 48 weeks of adefovir dipivoxil 10 mg once daily for chronic hepatitis B: an integrated analysis of two phase III studies. Digestive Disease Week 2003, A-71.
37.Angus P, et al.: Resistance to adefovir dipivoxil therapy associated with the selection of a novel mutation in the HBV polymerase. Gastroenterology 2003, 125:292-297.
38.* Locarnini S, et al.: Resistance surveillance of HBeAg-chronic hepatitis B patients treated for two years with adefovir dipivoxil. 11th International Symposium on Viral Hepatitis and Liver Disease. Sydney, Australia, April 2003.
Overview of viral mutations associated with drug resistance, the methods of diagnosis, clinical consequences and implications for future therapy.
39.Dore GJ, et al.: Efficacy of tenofovir disoproxil fumarate in antiretroviral therapy-naïve and -experienced patients coinfected with HIV-1 and hepatitis B virus. J Infect Dis 2004, 189:1185-1192.
40.Nelson M, et al.: An open-label study of tenofovir in HIV-1 and Hepatitis B virus co-infected individuals. AIDS 2003, 17:F7-F10.
41.Gish R, et al.: Antiviral Activity, Safety, and Incidence of Resistance in chronically Infected Hepatitis B Patients Given Once Daily Emtricitabine for 2 Years. Hepatology 2002, 36:372A.
42.Yao F, et al.: Lamivudine treatment is beneficial in patients with severely decompensated cirrhosis and actively replicating hepatitis B infection awaiting liver transplantation: A comparative study using a matched, untreated cohort. Hepatology 2001, 34:411-416.
43.Mutimer D, et al.: Significant Clinical Improvement Following the Addition of Adefovir Dipivoxil to Lamivudine in Decompensated Patients with YMD Variant HBV and a Reduced Response to Lamivudine -- 1 Year Results. Hepatology 2002, 36:625A.
44.Hoofnagle J, et al.: Interferon alfa for patients with clinically apparent cirrhosis due to chronic hepatitis B. Gastroenterology 1993, 104:1116-1121.
45.Schiff E, et al.: Adefovir Dipivoxil for the Treatment of Chronic Hepatitis B in Patients Pre- and Post-Liver Transplantation with Lamivudine Resistant Hepatitis B Virus Patients. Hepatology 2002, 36:371A.
46.Terrault N: Hepatitis B and Liver Transplantation. Clin Liver Dis 1999, 3:389-416.
47.Schalm S, et al.: Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: a randomised trial. Gut 2000, 46:562-568.
48.Schiff E, et al.: Lamivudine and 24 weeks of lamivudine/interferon combination therapy for hepatitis B e antigen-positive chronic hepatitis B in interferon nonresponders. J Hepatol 2003, 38:818-26.
49.Barbarini G, et al.: Long-Term Efficacy of Interferon Alpha-2b and Lamivudine in Combination Compared to Lamivudine Monotherapy in Patients With Chronic Hepatitis B An Italian Multicenter Randomized Trial. Hepatology 2001, 34:318A.
50.Torresi J: Antiviral chemotherapy for the treatment of hepatitis B virus infections. Gastroenterology 2000, 118(Suppl 1):S83-S103.
51.Marcellin P, et al.: Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. N Engl J Med 2004, 351:1206-1217.
52.* Peters MG, et al.: Adefovir dipivoxil alone or in combination with lamivudine in patients with lamivudine-resistant chronic hepatitis B. Gastroenterology 2004, 1126:91-101.
One of the first studies to address the issue of whether combination therapy is superior to monotherapy. This study evaluated the effects of combination lamivudine and adefovir versus adefovir alone in patients with lamivudine-resistant HBV infection. No difference in outcomes was apparent between the combination and monotherapy groups after 48 weeks treatment.
53.Lai C, et al.: Entecavir is superior to lamivudine in reducing hepatitis B virus DNA in patients with chronic hepatitis B infection. Gastroenterology 2002, 123:1831-1838.
54.Colonno R: Genotypic Analysis Following 48 Week Entecavir Treatment of Chronic HBV Subjects Failing Lamivudine Suggests Sustained Susceptibility and Lack of Resistance Development. Hepatology 2002, 36:644A.
55.Tassopoulos N, et al.: Entecavir is Effective in Treating Patients With Chronic Hepatitis B Who Have Failed Lamivudine Therapy. Hepatology 2001, 34:340A.
56.* Cooksley W, et al.: Peginterferon Alfa-2a (40KDa): an advance in the treatment of hepatitis B e antigen-positive chronic hepatitis B. J Viral Hepatitis 2003, 10:298-305.
First published clinical trial of pegylated interferon for the treatment of HBV. In a comparison of standard interferon (4.5 MU three times weekly) versus 90, 180 and 270 mg pegylated interferon alfa 2a, pegylated interferons achieved superior results.
57.Park J, et al.: Co-Infection With HIV and HBV: The Effect of Tenofovir Disoproxil Fumarate in Lamivudine and Famciclovir Experienced Patients. Hepatology 2002, 36:641A.
58.Cecil B: Tenofovir 300 mg Daily is Effective in Patients With Chronic HBV Who Fail Lamivudine. Hepatology 2002, 36:630A.
59.Nunez M, et al.: Therapy With Tenofovir in HBV/HIV Co-Infected Patients With Resistance or Partial Response to Lamivudine. Hepatology 2002, 36:636A.
|
|
| |
| |
|
|
|