| |
Disease Status & Therapeutic End Points in Hepatitis B
|
| |
| |
Hari S. Conjeevaram, MD, MS
Division of Gastroenterology, University of Michigan Medical Center, 3912
Taubman Center, Ann Arbor, MI
Current Hepatitis Reports 2005, 4:5-13
"...The primary end points of treatment in HBeAg-positive patients are HBeAg seroconversion along with sustained suppression of HBV DNA and normalization of serum ALT...
...patients with advanced disease who develop resistance can have further disease progression and decompensation, raise concerns over the continued use of lamivudine (3TC) after development of (HBV) drug resistance and the long-term clinical impact of lamivudine-resistant mutants.....
...HBV genotypes may be related to disease outcomes (ie, HBeAg seroconversion, disease progression) and response to antiviral therapy, (see discussion below)....
...The natural course of HBeAg-positive chronic hepatitis B is punctuated by spontaneous flares of disease activity. Recurrent episodes of necroinflammation and regeneration may increase the risk of fibrosis and cirrhosis as well as carcinogenesis. Therefore, all patients with chronic hepatitis B infection independent of their serologic status require long-term clinical, laboratory, and radiologic assessment for disease progression and potential complications.....
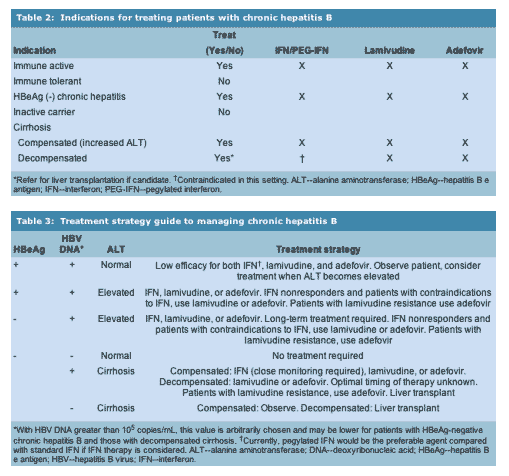
........ Significant advances have been made in the management of chronic hepatitis B, both with the diagnosis and understanding of disease stages as well as the availability of multiple treatment options. An important part of disease management is determining what phase of the natural history in chronic hepatitis B the patient is in and deciding whether treatment is warranted and what the end points of therapy should be (Table 1). Selection of antiviral agent is based upon several factors, such as patients' age, comorbid conditions, HBV replication status, severity of liver disease, and safety and side-effect profiles of the various agents (Tables 2 and 3). The role of continued long-term treatment with some of these agents and the role of combination therapies is not clear at this time. Thus, careful balance of the disease status, likelihood of response, and potential adverse events and complications is needed before treatment is recommended......"
Chronic hepatitis B virus (HBV) infection is the leading cause of cirrhosis and hepatocellular carcinoma. The availability of universal vaccination has made a significant impact in reducing the incidence of these complications. Several developments have been made in the treatment of chronic HBV infection. Three drugs (interferon alpha, lamivudine, and adefovir dipivoxil) are currently approved for use in HBV infection in the United States; these treatments have been shown to reduce morbidity and mortality for some individuals with chronic hepatitis B. However, these therapies are not universally successful and have limited long-term efficacy. In addition, the development of drug-resistant HBV mutants also impacts on the long-term benefit of some of these agents. Several new agents are currently undergoing study and will add to the list of available agents for the treatment of chronic HBV infection. In addition, the role of combination therapy using two or more agents has not been well defined in the management of hepatitis B.
TOPICS
Introduction
Natural History of Chronic Hepatitis B
Role of HBV DNA and HBV Genotypes
Management of Patients with HBeAg-positive and HBeAg-negative Chronic HBV Infection
--Standard interferons
HBeAg-negative chronic hepatitis
Pegylated interferon in chronic hepatitis B
--Lamivudine
HBeAg-negative chronic hepatitis B
--Adefovir dipivoxil
HBeAg-negative chronic hepatitis B
Use of adefovir in lamivudine-resistant hepatitis B
Management of Patients with Chronic Hepatitis B and Advanced Liver Disease and Post-liver Transplantation
--Interferon
--Lamivudine
--Adefovir
Drug Resistance in Antiviral Therapy of Chronic Hepatitis B
--Lamivudine-resistant HBV
--Adefovir-resistant HBV
Newer Therapies on the Horizon
Conclusions
References and Recommended Reading
INTRODUCTION
Hepatitis B virus (HBV) infection is a major global public health issue [1] because it is the leading cause of cirrhosis and hepatocellular carcinoma (HCC) worldwide. It is estimated there are at least 400 million HBV carriers in the world and that up to one million die annually due to hepatitis B-associated liver disease. Efforts to prevent and treat chronic hepatitis B infection are important toward the goal of global eradication of hepatitis B. Currently available treatments have been shown to reduce morbidity for some individuals with chronic hepatitis B; however, these therapies are not universally successful and have limited long-term efficacy. As a result, a complete understanding of the natural history of the disease along with factors influencing disease progression are important, as well as the indications and associated risks and benefits of available therapies.
NATURAL HISTORY OF CHRONIC HEPATITIS B
The natural course of chronic HBV infection can be broadly classified into four phases [2**]. The first two phases are characterized by the presence of hepatitis B e antigen (HBeAg) and high serum levels of HBV DNA (HBeAg-positive chronic hepatitis B). With this form of chronic hepatitis B, the serum alanine aminotransferase (ALT) levels may be elevated (immune active phase) or they may be normal in patients with perinatally acquired infection (immune tolerant phase). Whereas spontaneous and treatment-related HBeAg seroconversion is most common in the immune active phase of HBeAg-positive chronic hepatitis B, this rate is very low in the immune tolerant phase with or without treatment. Transition from the replicative phase to an inactive phase (inactive carrier-state) is characterized by spontaneous HBeAg clearance (HBeAg-), along with development of hepatitis B e antibody (anti-HBe+), also known as HBeAg seroconversion (HBeAg-, anti-HBe+), low or sometimes undetectable serum levels of HBV DNA, and normal ALT levels. Patients in the inactive carrier state do not require antiviral treatment but do require disease monitoring and surveillance for liver cancer. Some patients ultimately lose hepatitis B surface antigen (HBsAg) and this final phase is referred to as "resolution" of infection; loss of HBsAg is usually but not always accompanied by the development of antibody to HBsAg (anti-HBs), also known as HBsAg seroconversion (HBsAg-, anti-HBs+).
Another form of chronic hepatitis B infection referred to as "HBeAg-negative chronic hepatitis" is characterized by high levels of HBV DNA (> 100,000 copies/mL), elevated ALT, negative HBeAg, and presence of anti-HBe. This serologic profile usually arises due to a mutation that the virus undergoes at the time of or after HBeAg seroconversion. Patients with HBeAg-negative chronic hepatitis B may have residual populations of wild-type HBV or HBV variants that prevent the production of HBeAg (precore and core-promoter variants). This form of chronic hepatitis is marked by a fluctuating course and spontaneous remissions are rare. Thus, antiviral treatment may be indicated.
The natural course of HBeAg-positive chronic hepatitis B is also punctuated by spontaneous flares of disease activity [3]. Recurrent episodes of necroinflammation and regeneration may increase the risk of fibrosis and cirrhosis as well as carcinogenesis. Therefore, all patients with chronic hepatitis B infection independent of their serologic status require long-term clinical, laboratory, and radiologic assessment for disease progression and potential complications.
Role of HBV DNA and HBV Genotypes
Several assays to measure HBV-DNA levels, HBV genotypes, and HBV mutants and variants are currently available. These molecular diagnostic assays have improved our understanding of the clinical manifestations and natural history of HBV infection and facilitated the monitoring of response to treatment [2**]. Whereas hybridization and signal amplification assays have sensitivity limits of 105 to 106 copies/mL for HBV DNA, target amplification assays such as polymerase chain reaction (PCR) assays are capable of detecting very low levels (ie, 10 to 100 copies/mL) of HBV DNA.
In 2001, the National Institutes of Health (NIH) workshop on Management of Hepatitis B recommended that antiviral treatment be considered in patients with HBeAg-positive or HBeAg-negative chronic hepatitis and HBV DNA greater than 105 copies/mL2. This value of HBV DNA was arbitrarily chosen to include patients with detectable HBV DNA by nonamplified assays, although the HBV-DNA level associated with progressive liver disease has not been clearly determined. A virologic response associated with treatment-induced suppression of HBV DNA was defined as levels of less than 105 copies/mL. A low pretreatment HBV-DNA level has also been shown to be an important predictor of treatment response especially with the use of interferon (IFN), although the correlation between pretreatment serum HBV-DNA level and response to nucleos(t)ide analogues is not as strong.
There are at least eight different genotypes of HBV (A-H) based on an intergroup divergence in the complete nucleotide sequence [2**, 3, 4, 5]. The geographical distribution of HBV genotypes is varied, with genotype A being more common in the United States (U.S.). Although the use of HBV genotypes has not yet become part of routine patient management, HBV genotypes may be related to disease outcomes (ie, HBeAg seroconversion, disease progression) and response to antiviral therapy. The most common mutation (precore mutation G1896A) that blocks the production of HBeAg is predominantly found in association with HBV genotypes B, C, and D; as a result, HBeAg-negative chronic hepatitis B is more common in Asia and Southern Europe where these genotypes are more prevalent compared with the U.S. Patients with genotype C infection have been found to have a lower rate of spontaneous HBeAg seroconversion and higher rates of cirrhosis compared to those with genotype B [4, 6, 7]. Among IFN-treated patients, genotypes A and B have been reported to be associated with higher rates of antiviral response than genotypes C and D, respectively [8, 9].
Management of Patients with HBeAg-positive and HBeAg-negative Chronic HBV Infection
The primary end points of treatment in HBeAg-positive patients are HBeAg seroconversion along with sustained suppression of HBV DNA and normalization of serum ALT. In patients with HBeAg chronic HBV, sustained suppression of HBV DNA and normalization of serum ALT are the primary end points of therapy. A recent study found that whereas the relative risk of HCC among men with HBsAg alone was 9.6 compared to those without HBsAg, the risk increased to 60.2 in patients with detectable HBsAg and HBeAg [10]. There is also data to suggest that survival among cirrhotic patients is lower among those who are HBeAg positive [11]. These observations highlight the central role of virus replication in disease course and outcome and the need to induce sustained suppression of HBV replication.
Currently, three treatments are approved for use in the U.S. for the treatment of chronic hepatitis B: standard IFN alpha, lamivudine, and adefovir dipivoxil. Several other antiviral and immunomodulatory therapies are being evaluated and may play a key role in the future management of chronic HBV infection. A response to therapy is defined as undetectable HBV DNA in serum (using nonamplified assays), loss of HBeAg with or without detection of anti-HBe (HBeAg seroconversion), and improvement in liver disease (characterized by normalization of ALT levels and decrease in necroinflammation or prevention of fibrosis progression in liver biopsies). The NIH workshop on Management of Hepatitis B recommended that the definition and criteria of response to antiviral therapy in chronic hepatitis B be standardized [2**]. Response should be defined as biochemical, virologic, or histologic during treatment (at the end of treatment or maintained response during long-term therapy) or sustained 6 to 12 months off-therapy (Table 1).
TABLE 1. Definition of Response to Antiviral Therapy of Chronic Hepatitis B
Category of Response
--biochemical: decrease of serum ALT to within the normal range
--virologic: decrease of serum HBV DNA to undetectable levels in nonamplified assays (<10 to the 5th, 100,000 c/ml), and loss of HBeAg in patients who were initiatlly HBeAg positive
--histologic: decrease in histology activity index by at least 2 points compared with pretreatment liver biopsy
--complete: fulfill criteria of biochemical and virologic response, as well as HBsAg loss
Timing of Response
--on-treatment: response during treatment
--sustained: response that is maintained up to 6 to 12 months after cessation of treatment
STANDARD INTERFERONS
Ed note from Jules Levin, regarding Interferon in coinfection: the use of interferon for HBV therapy in HIV-positive individuals was studied before HAART. Results did not show much benefit. It is thought now that peginterferon may be a useful and effective therapy in coinfected patients but the studies have not been conducted in the post-HAART era. The thinking is that before HAART immune deficiency associated with HIV may have played a role in non-response to interferon, but in the post-HAART era the reconstitution of the immunity with increased CD4 counts may play a role in successful therapy of peginterferon. For individuals triply infected with HBV+HCV+HIV it makes sense to consider peginterferon+ribavirin.
HBeAg-positive chronic hepatitis
The effects of IFN in inducing virologic and biochemical response have been well established. Meta-analyses have shown that response (defined as normalization of serum ALT, loss of HBV DNA and HBeAg) is achieved in 25% to 40% of HBeAg-positive patients within 12 months of initiating treatment [2**, 12, 13]. This is in contrast to a rate of 10% to 15% spontaneous loss of HBeAg among untreated control subjects. The majority of responders maintain a virologic response long term and reactivation is seen in only 10% to 15%, mostly within the first year of stopping therapy. These patients continue to have low levels of HBV replication as determined by PCR assays. However, some long-term follow-up studies have shown that 5-year cumulative rates of HBeAg clearance are similar or even higher in untreated patients; therefore, the main role of IFN appears to be to hasten viral clearance, thereby reducing the duration of active liver disease [14, 15]. Experience in children has shown that IFN has a similar response with HBeAg clearance rates of approximately 30%, compared with 10% in untreated control subjects [2**, 14, 16].
Another important aspect of IFN therapy has been that treatment leads to higher rates of HBsAg loss compared to no treatment. Loss of HBsAg with IFN treatment occurs in 5% to 10% of patients within 1 year of the initiation of treatment and this increases with time among sustained virologic responders, with 5-year cumulative rates of 11% to 20% in European studies [14, 17] and up to 71% at 11 years in one U.S. study [18]. However, delayed HBsAg loss after a treatment response has not been observed in Asian studies [19, 20]. Although histologic assessment has not been studied in detail with IFN, studies have found that responders tend to have lower rates of hepatic decompensation and liver-related deaths when compared with control subjects; the effect of IFN on HCC development is less clear at this time [15, 17, 18].
With regard to predicting response to IFN, high pretreatment ALT and low HBV-DNA levels are the best predictors in HBeAg-positive patients and these factors are also associated with a higher rate of spontaneous HBeAg seroconversion [2**, 12, 15, 21, 22]. Patients who have "immune tolerant chronic hepatitis B" have a poor response to IFN (< 10% HBeAg loss) [12] and, therefore, treatment is generally not recommended in these patients.
The recommended dose of standard IFN for adults is 5 MU/d or 10 MU three times a week, and for children 6 MU/m2 three times a week with a maximum of 10 MU [22, 23**]. The recommended duration of treatment for patients with HBeAg-positive chronic hepatitis B is 16 to 24 weeks. One study reported that among patients who have not cleared HBeAg after 16 weeks of IFN, those randomized to continue treatment until week 32 had significantly higher rates of HBeAg clearance compared with those who stopped treatment [24].
HBeAg-negative chronic hepatitis
In this group, response to therapy is defined as undetectable serum HBV DNA by hybridization assays along with normalization of ALT level. A recent analysis showed that virologic response in HBeAg-negative patients ranged from 38% to 90% on IFN therapy, compared with 0% to 37% of control subjects [2**]. During follow-up at 12 months, sustained response rates varied from 10% to 47% (average 24%) among the treated patients and 0% in the control subjects. These patients appear to require longer duration of therapy compared with HBeAg-positive patients and response is more durable in patients who received treatment for 12 months or longer. HBsAg clearance has been noted in up to 15% to 30% of sustained responders during long-term follow-up [2**, 21, 25]. In addition, a recent study of 164 (103 treated) consecutive patients with HBeAg-negative chronic hepatitis showed that IFN slowed histologic disease progression, and sustained responders had lower rates of death and HCC [25].
PEGYLATED INTERFERON IN CHRONIC HEPATITIS B
In a recent report of 194 HBeAg-positive patients with chronic hepatitis B, treatment with pegylated IFN alfa-2a (90, 180, or 270 mg/wk) for 24 weeks resulted in more rapid and greater decline in HBV DNA compared with standard IFN (4.5 MU three times a week) [26]. HBeAg seroconversion was observed in 37%, 33%, and 27% versus 25% in the pegylated IFN and standard IFN groups, respectively.
In a recent phase III study of HBeAg-positive chronic hepatitis B, 266 patients were randomized to receive pegylated IFN alfa-2b alone or in combination with lamivudine for 12 months [27]. At the end of 6-month follow-up after completion of therapy, HBeAg loss was seen in 36% and 35%, respectively. Loss of HBsAg was seen in approximately 5% in both groups.
Recently, the results of a randomized, placebo- controlled study of 48 weeks of pegylated IFN alfa-2a 180 mg once weekly (PEG-IFN) plus lamivudine (100 mg/d) versus placebo or lamivudine monotherapy in patients with HBeAg-negative chronic hepatitis B were reported (n: 177, 179, and 181 patients, respectively) [28*]. Sustained normalization of serum ALT or undetectable HBV DNA (levels to below 20,000 copies/mL) was significantly higher with PEG-IFN monotherapy (59% and 43%, respectively) and PEG-IFN plus lamivudine (60% and 44%), compared with lamivudine monotherapy (44%, P = 0.004 and P = 0.003, respectively; and 29%, P = 0.007 and P = 0.003, respectively). Rates of sustained suppression of HBV DNA to below 400 copies/mL were 19% with PEG-IFN monotherapy, 20% with combination therapy, and 7% with lamivudine alone (P < 0.001 for PEG-IFN plus placebo vs with lamivudine alone). HBsAg loss was seen in 12 patients (3%) who received PEG-IFN and none of the patients who received lamivudine alone. As in patients with HBeAg- positive hepatitis, the addition of lamivudine to PEG-IFN does not appear to increase virologic response rates.
LAMIVUDINE (3TC)
Lamivudine (Epivir-HBV; GlaxoSmithKline, Research Triangle Park, NC) is the (-) enantiomer of 2',3'-dideoxy-3' -thiacytidine, which is phosphorylated to the triphosphate and competes with deoxycytidine triphosphate for incorporation into growing DNA chains causing chain termination.
HBeAg-positive chronic hepatitis B
Three large randomized clinical trials involving 731 patients from the U.S, Europe, and Asia have shown that lamivudine therapy for 1 year leads to HBeAg seroconversion in 16% to 18% of patients, compared with 4% to 6% of untreated control subjects [29, 30, 31, 32, 33]. Histologic improvement (defined as reduction in necroinflammatory score greater than 2 points) occurred in 49% to 56% of treated patients and in 23% to 25% of control subjects. In the Asian study, HBeAg seroconversion rates increased with duration of treatment, ranging from 17% at 1 year to 27%, 40%, 47%, and 50% at 2, 3, 4, and 5 years, respectively [34].
In a recent large multicenter controlled trial involving 286 patients aged 2 to 17 years with elevated ALT, patients were randomized to receive lamivudine (3 mg/kg/d up to 100 mg/d) or placebo for 52 weeks [35]. At the end of treatment, 23% of children treated with lamivudine lost HBeAg, compared with 13% on placebo. Drug-resistant HBV mutants were detected in 18% of treated children.
As with IFN, pretreatment serum ALT is the strongest predictor of response in patients with HBeAg-positive chronic hepatitis [36, 37]. In a recent analysis of four phase III trials (where a total of 406 patients received lamivudine for 1 year) [37], HBeAg seroconversion occurred in 2%, 9%, 21%, and 47% of patients with pretreatment ALT levels within normal, one to two times normal, two to five times normal, and more than 5 times normal; whereas the corresponding figures for 196 patients in the placebo group were 0%, 5%, 9%, and 15%, respectively. In the large multicenter placebo-controlled study of lamivudine in children, HBeAg seroconversion rate was higher in those with pretreatment ALT greater than two times normal (34% vs 16%) [35].
HBeAg-negative chronic hepatitis B
In patients with HBeAg-negative chronic hepatitis B [38, 39], virologic and biochemical response has been seen in up to 70% to 96% of patients receiving 48 weeks of lamivudine therapy versus less than 10% among patients on placebo. Histologic improvement has been seen in 20% to 95% of cases [40]. The majority of patients, however, relapse once therapy is stopped [40, 41]. Continued treatment, however, results in the development of drug- resistant mutants and response is not maintained; in one study, complete response (biochemical and virologic) was seen in 81% of patients at 1 year--this was decreased to 69% during the second year. Lamivudine-resistant mutants increased from 19% at the end of year 1 to 44% at the end of year 2 [42].
Lamivudine has also been studied in previous nonresponders to IFN treatment. In one study of 238 IFN nonresponders, patients were randomized to receive lamivudine alone for 52 weeks, combination of lamivudine plus IFN for 24 weeks, or no treatment. Histologic response was significantly more common in patients treated with lamivudine (52% vs placebo 25%, P = 0.002) or the combination regimen (32%, P = 0.01). HBeAg loss was also more common with lamivudine (33% vs 13% vs 21%); HBeAg seroconversion was seen in 18%, 12%, and 13% of patients, respectively [43]. These data suggest that patients who failed IFN treatment have similar virologic response to lamivudine as treatment-naive patients and re-treatment with combination of IFN and lamivudine did not confer any added benefit compared with lamivudine monotherapy.
When lamivudine is stopped, the majority of patients maintain HBeAg seroconversion. In the Asian study, 83% of patients maintained seroconversion after 6 to 36 (median 19) months [44]. However, the duration of lamivudine treatment after HBeAg seroconversion may be important to prevent early relapse after stopping treatment [44]. In one study of 67 Korean patients treated with lamivudine, those who received lamivudine for at least 4 months after HBeAg seroconversion had a much lower rate of relapse compared with those who received treatment for less than 2 months after HBeAg seroconversion (32% vs 74% at 2 years) [45]. Based on these findings, it is recommended that lamivudine be continued for at least 4 to 6 months after HBeAg seroconversion to decrease the likelihood of relapse [46].
The initial controlled studies have shown that 1-year treatment with lamivudine is associated with histologic improvement in more than 50% of patients [29, 30, 31]. With longer duration of treatment, however, drug-resistant mutations may reverse this initial histologic benefit. In the Asian trial, 62 patients were evaluated with liver histology at 0, 1, and 3 years. Whereas necroinflammatory scores continued to improve at 1 and 3 years among patients without resistance, histologic scores worsened at 3 years after an initial improvement at 1 year in those with resistant mutations [47]. These data, along with other reports suggesting that patients with advanced disease who develop resistance can have further disease progression and decompensation, raise concerns over the continued use of lamivudine after development of drug resistance and the long-term clinical impact of lamivudine-resistant mutants.
The recommended dose of lamivudine for adults is 100 mg/d given orally, with dose reduction reserved for patients with renal insufficiency. The recommended dose for children is 3 mg/kg up to 100 mg. Patients who have HBV and HIV coinfection should be treated with 150-mg doses twice a day in addition to other antiretroviral therapies.
Lamivudine is generally well tolerated. In patients who do not achieve HBeAg seroconversion (among those who are HBeAg positive) or biochemical plus virologic response (in HBeAg-negative chronic hepatitis B), lamivudine can be continued as long as one is aware of the potential risks of developing resistance. If treatment is discontinued, patients should be closely monitored because acute exacerbations of hepatitis and hepatic decompensation have been reported even in patients with HBeAg seroconversion [34, 48, 49*, 50]. If this occurs, lamivudine can be reinstituted and is usually effective in controlling exacerbations if the patients have not developed a breakthrough infection. In patients with HBeAg-negative chronic hepatitis B, a longer course of treatment is recommended to prevent relapse; however, the optimal duration is unclear.
ADEFOVIR DIPIVOXIL
(ed note regarding tenofovir & coinfection: a recent study was presented at the 12th CROI meeting finding that in HBV/HIV coinfected patients tenofovir was at least as potent as adefovir in suppressing HBV in coinfected patients. This was a non-inferiority study so the study was stopped at week 24 when it was clear that tenofovir had equal efficacy. But HBV DNA reductions were greater for tenofovir than for adefovir (at week 48: -4.03 vs -3.12 log, ITT). Gilead Sciences is planning studies to show that tenofovir is superior to adefovir in mono and coinfected patients. In coinfection, often HAART therapy includes tenofovir plus 3TC or FTC when the clinician decides to treat the HIV & the HBV at once. The link to the CROI report is:
http://www.natap.org/2005/CROI/croi_24.htm
Adefovir dipivoxil is an orally bioavailable prodrug of adefovir, a nucleotide analogue of adenosine monophosphate [2**, 51, 52, 53*]. It effectively inhibits both the reverse transcriptase and DNA polymerase activity of HBV.
HBeAg-positive chronic hepatitis B
In the multicenter placebo-controlled phase III study, 515 patients were randomized to receive 10 or 30 mg of adefovir or placebo for 48 weeks. Histologic response (defined as greater than 2-point decrease in Knodell necroinflammatory score with no worsening of fibrosis) was seen more commonly with both doses of adefovir compared with placebo (53% to 59% vs 25%) [52]. HBeAg loss was seen in 11%, 24%, and 27% and HBeAg seroconversion in 6%, 12%, and 14% of patients receiving placebo, adefovir 10 and 30 mg, respectively. No adefovir-associated resistant mutations were identified at the end of 48 weeks of therapy. More recent data suggest that as with lamivudine, continued treatment with adefovir will also increase rates of HBeAg seroconversion, increasing from 12% at week 48 to 29% at week 96 and 43% at week 144 of therapy [53*].
HBeAg-negative chronic hepatitis B
In the multicenter phase III trial of HBeAg-negative chronic hepatitis B, 185 patients were randomized to receive 10 mg of adefovir versus placebo in a 2:1 ratio for 48 weeks. Histologic response was significantly higher among the treated patients, 64% versus 33% of control subjects (P < 0.001) [54*]. Normalization of serum ALT and undetectable HBV DNA by PCR was achieved in 72% versus 29% and 51% versus 0%, respectively (P < 0.001). In addition, no adefovir-resistant mutations were identified at 48 weeks.
Use of adefovir in lamivudine-resistant hepatitis B
Adefovir has been shown to be effective in suppressing HBV DNA in both wild-type and lamivudine-resistant HBV infection [51, 55, 56]. In cases of lamivudine resistance, it is generally recommended to start adefovir while continuing lamivudine until a biochemical and virologic response is achieved before stopping lamivudine. If such a response is not seen (~ after 3 months) and there is no evidence of any flare, lamivudine can be withdrawn but the clinical course should be carefully followed because adefovir-resistant strains of HBV appear to arise more frequently in patients with prior lamivudine resistance.
Adefovir is generally well tolerated but a potential toxicity with adefovir is the development of a dose-dependent type 4 renal tubular acidosis. The recommended dose of adefovir is 10 mg/d. Dosing intervals should be adjusted in patients with renal insufficiency. Increases in serum creatinine (> 0.5 mg/dL) have been observed in approximately 20% of patients with decompensated cirrhosis and post-liver transplant patients [57]. Therefore, close monitoring is required in these clinical settings when using adefovir.
Management of Patients with Chronic Hepatitis B and Advanced Liver Disease and Post-liver Transplantation
Interferon
Patients with compensated cirrhosis appear to tolerate IFN and respond as well as patients with precirrhotic chronic hepatitis B. However, IFN is associated with a wide spectrum of side effects and there is substantial risk of serious infections and potentially fatal exacerbations of hepatitis even with low doses of IFN among patients with decompensated cirrhosis [58, 59]. Therefore, standard or pegylated IFN should not be used in patients with decompensated HBV cirrhosis.
Lamivudine
Lamivudine is a safer treatment than IFN in patients with advanced liver disease, including those with decompensated cirrhosis [60]. Improvement in liver disease with decrease in Child-Turcotte-Pugh score, along with decreased need for liver transplantation, has been observed in patients who completed a minimum of 6 months of treatment [61]. In a recent large study of 651 patients with advanced fibrosis or cirrhosis, patients were randomized in a 2:1 ratio to receive lamivudine (100 mg/d) or placebo for a maximum of 5 years. The study was terminated after a median duration of 32.4 months of treatment due to a significant reduction in the time to disease progression among patients receiving lamivudine compared with placebo (17.7% vs 7.8%, hazard ratio 0.45, P = 0.02) [62**]. Genotypic resistance on lamivudine developed in 49% of patients and patients with these mutations were more likely to have disease progression as determined by Child-Turcotte-Pugh score. Some patients with advanced liver disease continue to have progression despite antiviral therapy. For example, in an analysis of 154 patients with decompensated liver disease, the benefit of lamivudine was evident only in the 84% of patients who survived the initial 6 months of treatment [63].
The combined prophylactic use of hepatitis B immune globulin and lamivudine has reduced the incidence of recurrent HBV infection after transplant to less than 10% [2**, 64]. A recent review of 166 patients transplanted for hepatitis B over a 17-year period at a single center showed that survival is significantly improved, with 1- and 5-year patient survival rates of 86% and 72% [65]. Lamivudine has also been used to treat patients with recurrent hepatitis B. In one multicenter study, 52 patients with recurrent hepatitis B were treated with open-label lamivudine for 52 weeks [66]. HBV DNA became undetectable in 60%, and 31% of HBeAg-positive patients lost HBeAg. Loss of HBsAg was seen in 6%. Virologic response was accompanied by biochemical and histologic improvement; however, lamivudine-resistant mutations were detected in 27% of patients and in some this was accompanied by clinical deterioration.
Adefovir
Recently, the addition of adefovir with continued use of lamivudine in patients with lamivudine resistance was reported in 135 patients with chronic hepatitis B [67]. Ninety-five patients with compensated chronic hepatitis B were randomized to receive adefovir 10 mg/d or placebo for 52 weeks while continuing lamivudine. Forty patients with decompensated disease or post-transplantation received open-label adefovir along with lamivudine. HBV-DNA response (defined as decline in serum HBV DNA level to 105 copies/mL or greater than 2 log10 reduction baseline) was seen in 85% of patients on combination therapy, compared with only 11% on lamivudine plus placebo. In the patients with decompensated disease or post-transplantation receiving open-label combination therapy, HBV-DNA response was seen in 92%. In another study of 196 patients with recurrent hepatitis B post-liver transplantation, adefovir at a dose of 10 mg induced a 3 to 4 log reduction in HBV DNA at 48 weeks [57].
Recently, the effect of adefovir given alone or in combination with lamivudine in patients with lamivudine-resistant hepatitis B was reported. In this study, 59 HBeAg-positive patients were randomized to receive adefovir 10 mg/d, lamivudine 100 mg/d, or combination therapy (adefovir and lamivudine) [68]. Rapid reduction in HBV DNA was seen as early as 4 weeks in the adefovir and adefovir plus lamivudine groups, which continued in both groups through 48 weeks. These studies suggest that long-term use of combination therapy may not be needed, because a similar effect, at least on HBV-DNA suppression, can be achieved with adefovir alone in patients with lamivudine-resistant hepatitis B. Although these data are promising, longer follow-up is needed to establish the long-term efficacy of adefovir dipivoxil in patients with lamivudine-resistant HBV mutants.
Drug Resistance in Antiviral Therapy of Chronic Hepatitis B
Lamivudine-resistant HBV
One of the main concerns with continued use of lamivudine is the development of drug-resistant mutants, which involve the polymerase (P) gene of the HBV genome. Three mutations have been well defined: these include methionine to valine or isoleucine (rtM204V/I) substitutions involving the YMDD locus in domain C and leucine to methionine substitution (rtL180M) in domain B [69]. Patients who develop resistance usually have a virologic and biochemical breakthrough with reappearance of HBV DNA in serum using nonamplified assay after being undetectable and this is also followed by rise in serum liver enzymes [70]. Factors related to the development of resistance included non-Asian ethnicity, high pretreatment serum HBV-DNA level, male sex, and high body mass index [37]. High pretreatment ALT level and serum HBV greater than 103 copies/mL after 6 months of treatment have also been shown to predict the development of lamivudine resistance [71].
When patients develop lamivudine resistance, management options include continuing lamivudine especially if a patient has normal or low ALT levels, to discontinuing treatment and monitoring closely for hepatitis flares, or adding another antiviral agent such as adefovir. If a patient has cirrhosis or is immunosuppressed, however, stopping lamivudine can lead to clinical decompensation if a flare develops. In this circumstance, it may be best to consider adding another agent such as adefovir and stop lamivudine once ALT is normalized or stable and HBV-DNA level is suppressed.
Adefovir-resistant HBV
Although adefovir-resistant mutations in the DNA polymerase region have not been seen in earlier studies, emerging data suggest that with longer duration adefovir resistance is being reported. In a recent report of long-term adefovir, rates of adefovir-related resistant mutations (rtN236T and rtA181V) were noted to be 3.9 at 96 weeks and 5.9% at 144 weeks, respectively [52]. Further data in larger number of patients are needed before we know the full impact of adefovir-resistant mutations on the clinical course of chronic hepatitis B.
|
|
 |
| |
Newer Therapies on the Horizon
Several new drugs are currently being evaluated for the management of chronic hepatitis B (both HBeAg-positive and HBeAg-negative cases). As new drugs are approved, the choices for using different agents either alone or in combination are expected to increase. A major part of the decision making will include ease of administration, duration of treatment, efficacy (virologic, serologic, and biochemical), side-effect profile, and development of drug-resistant mutations. Drugs that are currently being evaluated include entecavir, emtricitabine (Coviracil/FTC; Triangle Pharmaceuticals, Durham, NC), Clevudine (Triangle Pharmaceuticals; L-FMAU, 2'-fluoro-5-methyl-b-L-arabinofuranosyl), and L-nucleosides [12, 72]. The role of combination therapies (both currently approved treatments and newer agents) is unclear at this time in the management of chronic hepatitis B, despite some studies showing virologic, serologic, and biochemical benefit.
Ed note: March 11, the FDA is holding a public hearing to review approval of entecavir for hepatitis B treatment.
References and Recommended reading
Recently published papers of particular interest have been highlighted as:
* Of importance
** Of major importance
1. Lee WM: Hepatitis B virus infection. N Engl J Med 1997, 337:1733-1745.
2.** Lok AS: Management of hepatitis B: 2000--summary of a workshop. Gastroenterology 2001, 120:1828-1853.
This is an excellent summary of the NIH workshop on hepatitis B. It includes a summary of the natural history of chronic hepatitis B, and recommended definitions of biochemical, serologic, and virologic profiles to define disease status and treatment response.
3.Kidd-Ljunggren K: Genetic variability in hepatitis B viruses. J Gen Virol 2002, 83:1267-1280.
4.Chu CJ: Clinical significance of hepatitis B virus genotypes. Hepatology 2002, 35:1274-1276.
5.Chu CJ, et al.: Hepatitis B virus genotypes in the United States: results of a nationwide study. Gastroenterology 2003, 125:444-451.
6.Chu CJ: Hepatitis B virus genotype B is associated with earlier HBeAg seroconversion compared with hepatitis B virus genotype C. Gastroenterology 2002, 122:1756-1762.
7.Kao JH: Genotypes and clinical phenotypes of hepatitis B virus in patients with chronic hepatitis B virus infection. J Clin Microbiol 2002, 40:1207-1209.
8.Kao JH, et al.: Hepatitis B genotypes and the response to interferon therapy. J Hepatol 2000, 33:998-1002.
9.Zhang X, et al.: Analysis of hepatitis B virus genotypes and pre-core region variability during interferon treatment of HBe antigen negative chronic hepatitis B. J Med Virol 1996, 48:8-16.
10.Yang HI, et al.: Hepatitis B e antigen and the risk of hepatocellular carcinoma. N Engl J Med 2002, 347:168-174.
11.Realdi G, et al.: Survival and prognostic factors in 366 patients with compensated cirrhosis type B: a multicenter study. The Investigators of the European Concerted Action on Viral Hepatitis (EUROHEP). J Hepatol 1994, 21:656-666.
12.Wong DK, et al.: Effect of alpha-interferon treatment in patients with hepatitis B e antigen-positive chronic hepatitis B. A meta-analysis. Ann Intern Med 1993, 119:312-323.
13.Craxi A: Interferon alpha for HBeAg positive chronic hepatitis B: systematic review. Paper presented at the Proceedings of the European Association for the Study of the Liver (EASL) International Consensus Conference on Hepatitis B. Geneva, Switzerland; September 13-14, 2002.
14.Bortolotti F, et al.: Long-term effect of alpha interferon in children with chronic hepatitis B. Gut 2000, 46:715-718.
15.Niederau C, et al.: Long-term follow-up of HBeAg-positive patients treated with interferon alfa for chronic hepatitis B. N Engl J Med 1996, 334:1422-1427.
16.Sokal EM, et al.: Interferon alfa therapy for chronic hepatitis B in children: a multinational randomized controlled trial. Gastroenterology 1998, 114:988-995.
17.Fattovich G, et al.: Delayed clearance of serum HBsAg in compensated cirrhosis B: relation to interferon alpha therapy and disease prognosis. European Concerted Action on Viral Hepatitis (EUROHEP). Am J Gastroenterol 1998, 93:896-900.
18.Lau DT, et al.: Long-term follow-up of patients with chronic hepatitis B treated with interferon alfa. Gastroenterology 1997, 113:1660-1667.
19.Lin SM, et al.: Long-term beneficial effect of interferon therapy in patients with chronic hepatitis B virus infection. Hepatology 1999, 29:971-975.
20.Lok AS: Long-term follow-up of chronic hepatitis B patients treated with interferon alfa. Gastroenterology 1993, 105:1833-1838.
21.Papatheodoridis GV: The long-term outcome of interferon-alpha treated and untreated patients with HBeAg-negative chronic hepatitis B. J Hepatol 2001, 34:306-313.
22.Hoofnagle JH: The treatment of chronic viral hepatitis. N Engl J Med 1997, 30:336:347-356.
23.** Lok AS: Chronic hepatitis B: update of recommendations. Hepatology 2004, 39:857-861. This is an excellent updated summary of the guidelines for management of chronic hepatitis B provided by the American Association for the Study of Liver Diseases.
24.Janssen HL, et al.: Interferon alfa for chronic hepatitis B infection: increased efficacy of prolonged treatment. The European Concerted Action on Viral Hepatitis (EUROHEP). Hepatology 1999, 30:238-243.
25.Brunetto MR, et al.: Outcome of anti-HBe positive chronic hepatitis B in alpha-interferon treated and untreated patients: a long term cohort study. J Hepatol 2002, 36:263-270.
26.Cooksley WGE, et al.: Peginterferon alfa-2A (40 kDa): an advance in the treatment of HBeAg positive chronic hepatitis B. J Viral Hepat 2003, 10:289-305.
27.Janssen HLA, et al.: Peginterferon alfa-2b and lamivudine combination therapy compared with peginterferon alfa-2b for chronic HBeAg positive hepatitis B: a randomized controlled trial in 307 patients. Hepatology 2003, 38(suppl 1):724A.
28.* Marcellin P, et al.: Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. N Engl J Med 2004, 351:1206-1217.
This is a recent report of the large randomized controlled study of pegylated IFN compared with lamivudine and combination (pegylated IFN plus ribavirin) in patients with HBeAg-negative chronic hepatitis B.
29.Lai CL, et al.: A one-year trial of lamivudine for chronic hepatitis B. N Engl J Med 1998, 339:61-68.
30.Dienstag JL, et al.: Lamivudine as initial treatment for chronic hepatitis B in the United States. N Engl J Med 1999, 341:1256-1263.
31.Schalm SW, et al.: Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: a randomized trial. Gut 2000, 46:562-568.
32.Liaw YF, et al.: Effects of extended lamivudine therapy in Asian patients with chronic hepatitis B. Asia Hepatitis Lamivudine Study Group. Gastroenterology 2000, 119:172-180.
33.Leung NW, et al.: Extended lamivudine treatment in patients with chronic hepatitis B enhances hepatitis B e antigen seroconversion rates: results after 3 years of therapy. Hepatology 2001, 33:1527-1532.
34.Guan R, et al.: Efficacy and safety of 5 years lamivudine treatment of Chinese patients with chronic hepatitis B [abstract 187]. J Gastroenterol Hepatol 2001, 16(suppl 1):A60.
35.Jonas MM, et al.: Clinical trial of lamivudine in children with chronic hepatitis B. N Engl J Med 2002, 346:1706-1713.
36.Chien RN, et al.: Pretherapy alanine transaminase level as a determinant for hepatitis B e antigen seroconversion during lamivudine therapy in patients with chronic hepatitis B. Hepatology 1999, 30:770-774.
37.Perrillo RP, et al.: Predictors of HBeAg loss after lamivudine treatment for chronic hepatitis B. Hepatology 2002, 36:186-194.
38.Rizzetto M: Efficacy of lamivudine in HBeAg-negative chronic hepatitis B. J Med Virol 2002, 66:435-451.
39.Gaia S, et al.: Lamivudine in anti-HBeAg positive chronic hepatitis B: response after one year of treatment. J Hepatol 2000, 32(suppl 2):112.
40.Tassopoulos NC, et al.: Efficacy of lamivudine in patients with hepatitis B e antigen-negative/hepatitis B virus DNA-positive (precore mutant) chronic hepatitis B. Lamivudine Precore Mutant Study Group. Hepatology 1999, 29:889-896.
41.Santantonio T, et al.: Long-term follow-up of patients with anti-HBe/HBV DNA-positive chronic hepatitis B treated for 12 months with lamivudine. J Hepatol 2000, 32:300-306.
42.Buti M, et al.: Two years of lamivudine therapy in anti-HBe-positive patients with chronic hepatitis B. J Viral Hepat 2001, 8:270-275.
43.Schiff ER, et al.: Lamivudine and 24 weeks of lamivudine/interferon combination therapy for hepatitis B e antigen-positive chronic hepatitis B in interferon nonresponders. J Hepatol 2003, 38:818-826.
44.Leung NW, et al.: Durable HBeAg response in Chinese patients treated with lamivudine [abstract]. Hepatology 2001, 34:348A.
45.Song BC, et al.: Hepatitis B e antigen seroconversion after lamivudine therapy is not durable in patients with chronic hepatitis B in Korea. Hepatology 2000, 32:803-806.
46.Chien RN, et al.: Determinants for sustained HBeAg response to lamivudine therapy. Hepatology 2003, 38:1267-1273.
47.Leung N: The effect of longer duration of harbouring lamivudine-resistant hepatitis B virus (YMDD mutants) on liver histology during 3 years lamivudine therapy in Chinese patients [abstract]. Hepatology 2001, 34:348A.
48.Chang TT, et al.: Incremental increases in HBeAg seroconversion and continued ALT normalization in Asian chronic HBV (CHB) patients treated with lamivudine for four years. Antivir Ther 2000, 5(suppl 1):44.
49.* Leung NY, et al.: Long-term safety of lamivudine in Asian and Caucasian patients with chronic hepatitis B. J Hepatol 2001, 34(suppl 1):160.
This study reports the effects of long-term therapy with lamivudine in chronic hepatitis B with regard to treatment efficacy (HBeAg seroconversion) and the development and consequences of drug-induced viral-resistant mutants.
50.Honkoop P, et al.: Acute exacerbation of chronic hepatitis B virus infection after withdrawal of lamivudine therapy. Hepatology 2000, 32:635-639.
51.Gilson RJ, et al.: A placebo-controlled phase I/II study of adefovir dipivoxil in patients with chronic hepatitis B virus infection. J Viral Hepat 1999, 6:387-395.
52.Marcellin P, et al.: Long term efficacy and safety of Adefovir dipivoxil (ADV) in HBeAg+ chronic hepatitis B patients: increasing serologic, virologic and biochemical response over time [abstract]. Hepatology 2004, 40:655A.
53.* Marcellin P, et al.: Adefovir dipivoxil for the treatment of hepatitis B e antigen-positive chronic hepatitis B. N Engl J Med 2003, 348:808-816.
This is a recent report of the large randomized, double-blind, placebo-controlled study of adefovir dipivoxil in patients with HBeAg-positive chronic hepatitis B.
54.* Hadziyannis SJ, et al.: Adefovir dipivoxil for the treatment of hepatitis B e antigen-negative chronic hepatitis B. N Engl J Med 2003, 348:800-807. This is a recent report of the large randomized, double-blind, placebo-controlled study of adefovir dipivoxil in patients with HBeAg-negative chronic hepatitis B.
55.Perrillo R, et al.: Adefovir dipivoxil for the treatment of lamivudine-resistant hepatitis B mutants. Hepatology 2000, 32:129-134.
56.Perrillo R, et al.: The addition of adefovir dipivoxil to lamivudine in decompensated chronic hepatitis B patients with YMDD variant HBV and reduced response to lamivudine-preliminary 24 week results [abstract]. Hepatology 2001, 34:349A.
57.Schiff ER, et al.: Adefovir dipivoxil (ADV) for the treatment of chronic hepatitis B in patients pre- and post- liver transplantation (OLT) with lamivudine resistant (LAM-R) hepatitis B virus (HBV) patients [abstract]. Hepatology 2002, 36:371A.
58.Hoofnagle JH: Interferon alfa for patients with clinically apparent cirrhosis due to chronic hepatitis B. Gastroenterology 1993, 104:1116-1121.
59.Perrillo R, et al.: Low-dose, titratable interferon alfa in decompensated liver disease caused by chronic infection with hepatitis B virus. Gastroenterology 1995, 109:908-916.
60.Perrillo RP, et al.: A multicenter United States-Canadian trial to assess lamivudine monotherapy before and after liver transplantation for chronic hepatitis B. Hepatology 2001, 33:424-432.
61.Fontana RJ, et al.: Effect of lamivudine treatment on survival of 309 North American patients awaiting liver transplantation for chronic hepatitis B. Liver Transpl 2002, 8:433-439.
62.** Liaw YF, et al.: Lamivudine for patients with chronic hepatitis B and advanced liver disease. N Engl J Med 2004, 351:1521-1531.This study reports the significant beneficial effects on lamivudine in patients with advanced liver disease due to chronic hepatitis B.
63.Fontana RJ, et al.: Determinants of early mortality in patients with decompensated chronic hepatitis B treated with antiviral therapy. Gastroenterology 2002, 123:719-727.
64.Colquhoun SD, et al.: Transplantation in the hepatitis B patient and current therapies to prevent recurrence. Semin Liver Dis 2000, 20(suppl 1):7-12.
65.Anselmo DM, et al.: New era of liver transplantation for hepatitis B: a 17-year single-center experience. Ann Surg 2002, 235:611-619.
66.Perrillo R, et al.: Multicenter study of lamivudine therapy for hepatitis B after liver transplantation. Lamivudine Transplant Group. Hepatology 1999, 29:1581-1586.
67.Perrillo R, et al.: Adefovir dipivoxil added to ongoing lamivudine in chronic hepatitis B with YMDD mutant hepatitis B virus. Gastroenterology 2004, 126:81-90.
68.Peters MG, et al.: Adefovir dipivoxil alone or in combination with lamivudine in patients with lamivudine-resistant chronic hepatitis B. Gastroenterology 2004, 126:91-101.
69.Stuyver LJ, et al.: Nomenclature for antiviral-resistant human hepatitis B mutations in the polymerase region. Hepatology 2001, 33:751-757.
70.Liaw YF: Results of lamivudine in Asian trials. Paper presented at the Proceedings of the European Association for the Study of the Liver (EASL) International Consensus Conference on Hepatitis B. Geneva, Switzerland; September 13-14, 2002.
71.Zollner B, et al.: Subtype-dependent response of hepatitis B virus during the early phase of lamivudine treatment. Clin Infect Dis 2002, 34:1273-1277.
72.Lok ASF: Management of chronic hepatitis B. J Hepatol 2003, 38(suppl 1):S90-S103.
|
|
| |
| |
|
|
|