| |
Natural history of chronic HBV infection: A cohort study with up to 12 years follow-up in North Greece (part of the Interreg I-II/EC-project)
|
| |
| |
Journal of Medical Virology Oct 2005
Volume 77, Issue 2 , Pages 173 - 179
G.H. Zacharakis 1 2 *, J. Koskinas 2, S. Kotsiou 3, M. Papoutselis 1, F. Tzara 1, N. Vafeiadis 1, A.J. Archimandritis 2, K. Papoutselis 1
1Unit of Preventive Medicine, Social Security Institute, Alexandroupolis, Greece
2Second Department of Internal Medicine, Athens University, School of Medicine, Hippokration General Hospital, Greece
3A Academic Department of Internal Medicine, University of Thrace, Alexandroupolis General Hospital, Greece
.....In our study population, the phase of HBeAg(+) chronic HBV infection accounts for only 5.3% of patients whereas the HBeAg(-)/anti-HBe(+) phase
was observed in 94.7%.....
.....In contrast to the inactive carriers, patients with HBeAg(-)/anti-HBe(+) chronic infection and elevated ALT levels had active liver disease with moderate to
severe fibrosis observed in 46% of them. At the 4th year follow up biopsy 8% of them had developed deterioration of liver disease. The annual rate risk of cirrhosis and HCC was estimated 1% and 0.5%, respectively.....
..... The last two decades a lot of prospective studies on the prognosis of chronic HBV infection have been reported. In these series, a great variability in the rate of progression to cirrhosis and HCC exists that relates to differences in the demographic features and the selection of the study population or the presence of preexisting cirrhosis at enrollment.....
.....in our population-based study, it was found that the course and outcome of the inactive carriers, who constitute the majority of HBeAg(-) chronic HBV infected patients, is benign, as has been reported previously [Hoofnagle et al., 1987; Lok and McMahon, 2001] although, in some studies, slightly increased morbidity and mortality rates have been observed [Velasco et al., 1978; Sakuma et al., 1982; Alward et al., 1985; Villeneuve et al., 1994; Manno et al.,
2004]. In contrast, in HBeAg(-) patients with active liver disease the annual rate of development cirrhosis or HCC was 1% and 0.5%, respectively. Finally, an interesting point in the current study was the observation that serum HBV-DNA levels <104 copies/ml (10,000) were closely associated with the inactive carrier state over the follow-up period of up to 12 years."
Abstract
The aim of the study was to assess the long-term outcome of chronic hepatitis B surface antigen (HBsAg) carriers in the general population in North Greece (Thrace), an area with an intermediate endemicity. This was a part of the Interreg I-II EC project.
Two hundred sixty three chronic HBsAg(+) carriers, median age 34 years (20-65), were evaluated prospectively for a median follow-up of 5 years (2-12).
Hepatitis B virus (HBV) markers and ALT were examined every 6 months and serum HBV-DNA every 12 months. Liver biopsy was undertaken at presentation and every 2-4 years.
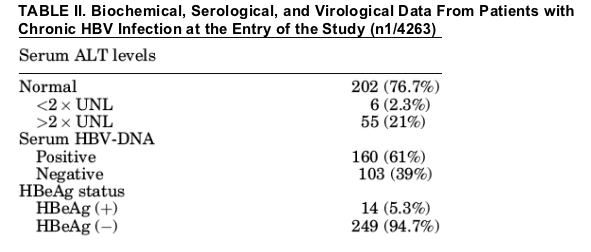
Fourteen of 263 (5.3%) subjects were HBeAg(+) and 249/263 (94.7%) HBeAg(-)/anti-HBe(+) of whom 48 (19.3%) had elevated ALT, and HBV-DNA levels ranging from 1.4 X105-4 X107 copies/ml.
Inactive carriers (98/195 (50.3%)) had detectable HBV-DNA (median 2.6 X103 range 0.042 X104-1.9 X104 copies/ml); 4/195 (2%) exhibited HBV reactivation during the observation period (all had HBV-DNA >104 copies/ml at presentation).
Patients (7/14 (50%) HBeAg(+)) developed anti-HBe(+), annual rate 10%. Subjects (16/195 (8%)) lost HBsAg, all were inactive carriers; 10 developed anti-HBs (annual rate 1%).
Liver biopsy was normal or with minimal changes in 92/95 (97%) inactive carriers and remained so at 4 years follow-up.
In contrast, 4/48 (8.3%) HBeAg(-)/anti-HBe(+) patients with active disease had deterioration of liver histology.
In this cohort study: (a) the annual seroconversion rate was 1% for the HBsAg and 10% for the HBeAg, (b) 23.6% of the HBsAg(+) carriers had active liver disease and 39% moderate fibrosis at presentation of whom a small proportion deteriorated over the observation period, (c) HBsAg carriers with HBV-DNA level <104 copies/ml had persistently normal ALT and unchanged liver histology over the follow-up period of up to 12 years.
Preliminary results of this study were presented at the 39th Annual Meeting of The European Association for the Study of the Liver.
DISCUSSION
This is a long-term prospective study evaluating the natural history of chronic HBV infection in the general population in Thrace, Northern Greece. Chronic HBV infection has a dynamic natural history with various phases that reflect the viral-host interplay and have different impact on disease prognosis [Chisari
and Ferrari, 1995; Lee, 1997; Hadziyannis and Vassilopoulos, 2001a,b]. In our study population, the phase of HBeAg(+) chronic HBV infection accounts for only 5.3% of patients whereas the HBeAg(-)/anti-HBe(+) phase was observed in 94.7%. This HBeAg(-)/anti-HBe(+) proportion of the patients in this study stands true for the entire HBV population in Greece as reported in previous studies [Hadziyannis and Vassilopoulos, 2001a,b]. Similarly, other long-term studies reported that >90% of HBV carriers infected early in life become HBeAg(-) at the fifth decade of life [Dragosic et al., 1987; Bortolloti et al., 1990; McMahon et al., 2001].
In a present study, during the follow-up period, 50% of HBeAg(+) patients developed anti-HBe(+) immunity, with a yearly incidence of 10%. After HBeAg seroconversion a small number of patients remained with active liver disease, findings that have been observed in other studies [Fattovich et al., 1986; Evans et al., 1997; Bortolotti et al., 1998; Hsu et al., 2002]. The persistence of hepatitis after HBeAg seroconversion is probably due to emergence and activation of HBV variants, most likely the precore mutant variants [Hadziyannis and Vassilopoulos, 2001b]. In the literature, the reported annual HBeAg to anti-HBe seroconversion rate varies from 8%to 15% in children or adults with increased ALT levels [Fattovich et al., 1986; Bortolloti et al., 1990; Evans et al., 1997; Papatheodoridis et al., 2001; Lok and McMahon, 2001; Yuen et al., 2001]. Several determinants
for HBeAg seroconversion have been reported including gender, age, ALT levels, histological activity, and more recently HBV genotype [Liaw et al., 1983;
Alward et al., 1985; Lok et al., 1987; McMahon et al., 2001; Sanchez-Tapias et al., 2002]. With respect to activity of liver disease, the seroconversion rate was
reported to be 15%-20% in patients with severe hepatitis reaching to 60% in those with ALT >5 UNL, UNL, but less than 5%-10% in those with minimal
histological activity [Liaw et al., 1984; Liaw, 1997].
A large proportion of our chronic HBV patients (76%) were at the inactive carrier state. Almost all (98%) remained with normal ALT levels during the observation
period of up to 12 years; roughly 50% had detectable serum HBV-DNA. Almost all (97%) inactive carriers with detectable serum HBV-DNA had normal or minimal changes on liver biopsy that remained unchanged at 4 years follow-up evaluation. Similarly, in a large group of French HBsAg inactive carriers the histology showed mild activity in all individuals and rarely evidence of fibrosis [Martinot-Peignoux et al., 2002].
A very small minority (2%) of the inactive carriers developed HBV reactivation with ALT flares during the follow-up period. On this issue there are conflicting data in the literature. Some studies report a 20%-30% spontaneous reactivation rate [Lok et al., 1987; Perillo, 2001;Hsuet al., 2002], others with a long-term follow-up period (up to 18 years), showed much lower rate [Dragosic et al., 1987; De Franchis et al., 1993; Bellentani et al., 2002]. These discrepancies may be
due to differences in the study group (ethnicity, age, etc,) and definition of the inactive carrier state.
Most importantly, all our inactive carriers with detectable serum HBV-DNA levels <104 copies/ml (10,000), at enrollment, remained inactive during the observation
period in contrast to those who developed reactivation who had HBV-DNA levels >104 copies/ml. In the literature, the cut-off level of serum HBV-DNA, as a clinical relevant marker of HBV replicative activity was proposed to be the 105 copies/ml [Lok et al., 2001] although lower levels have been suggested [Martinot-
Peignoux et al., 2002; Manesis et al., 2003].
In contrast to the inactive carriers, patients with HBeAg(-)/anti-HBe(+) chronic infection and elevated ALT levels had active liver disease with moderate to
severe fibrosis observed in 46% of them. At the 4th year follow up biopsy 8% of them had developed deterioration of liver disease. The annual rate risk of cirrhosis and HCC was estimated 1% and 0.5%, respectively.
The last two decades a lot of prospective studies on the prognosis of chronic HBV infection have been reported. In these series, a great variability in the rate of progression to cirrhosis and HCC exists that relates to differences in the demographic features and the selection of the study population or the presence of preexisting cirrhosis at enrollment.
Firstly, in untreated patients with HBeAg(-) chronic hepatitis and without preexisting cirrhosis, progression to cirrhosis and development of HCC was reported to range from 0 to 0.12 and from 0 to 0.25 per 100 person
years, respectively [Di Marco et al., 1999; Papatheodoridis et al., 2001; Brunetto et al., 2002]. Secondly, in studies that have evaluated asymptomatic patients,
mostly blood donors aged <50 years and without histological follow-up, for a period ranging from 3.5 to 30 years, the estimated annual risk of cirrhosis andHCC was 0%-0.5% and 0%-0.2%, respectively [Dragosic et al., 1987; De Franchis et al., 1993; Villeneuve et al., 1994; McMahon et al., 2001; Manno et al., 2004]. Lastly, in studies that included either male patients with verified chronic liver disease or high age or HDV coinfection the liver disease associated complications developed more frequently [Weissberg et al., 1984; Fattovich et al., 1988; Fattovich et al., 1991; Hadziyannis et al., 1991; De Jongh et al., 1992]. Additionally, in areas with high endemicity of HBV infection, the risk of
HCC in male patients who maintained HBeAg positivity at the age of 50 years was1%per year [Yang et al., 2002]. Loss of HBsAg during the follow-up evaluation was observed in 8.2% patients being at the inactive carrier
state, but anti-HBs immunity was seen in 5.1%, with a yearly incidence of 1%. In western countries, where HBV infection is usually acquired in adulthood, the
incidence of delayed HBsAg clearance has been estimated to be 1%-2% per year [Fattovich et al., 1998; Papatheodoridis et al., 2001; Manno et al., 2004]. A
lower rate from 0.05% to 0.8% per year has been described in endemic areas, where HBV infection is acquired mostly perinatally or in early childhood as in
the case of Thrace [Mahon et al., 2001; Yuen et al., 2001].
Development of anti-HBs immunity was seen only in 63% of our patients who lost HBsAg, this is an uncommon phenomenon in chronic HBV infection
[Chisari and Ferrari, 1995; Lok and McMahon, 2001; Hadziyannis and Vassilopoulos, 2001a]. During this stage,HBV-DNA levels may still be detectable by PCR in serum and more often in the liver [Chemin et al., 2001]. In rare cases of severe immune suppression, such as cancer chemotherapy or after organ transplantation, HBV can be reactivated in patients with resolved hepatitis B [Kawatani et al., 2001]. However, no such cases were found in the present study.
In conclusion, in our population-based study, it was found that the course and outcome of the inactive carriers, who constitute the majority of HBeAg(-) chronic HBV infected patients, is benign, as has been reported previously [Hoofnagle et al., 1987; Lok and McMahon, 2001] although, in some studies, slightly increased morbidity and mortality rates have been observed [Velasco et al., 1978; Sakuma et al., 1982; Alward et al., 1985; Villeneuve et al., 1994; Manno et al., 2004]. In contrast, in HBeAg(-) patients with active liver disease the annual rate of development cirrhosis or HCC was 1% and 0.5%, respectively. Finally, an
interesting point in the current study was the observation that serum HBV-DNA levels <104 copies/ml were closely associated with the inactive carrier state over the follow-up period of up to 12 years.
INTRODUCTION
Hepatitis B virus (HBV) infection is a global health problem responsible for 5,00,000-1.2 million deaths annually from cirrhosis and hepatocellular carcinoma
(HCC) [Maddrey, 2000]. The implementation of vaccination in many countries has reduced greatly the number of new infections, but a large reservoir of HBV
infected individuals remain, particularly in countries with low socioeconomic conditions [Mast et al., 1999]. Immigration, particularly during the last decades, from poor countries to the developed countries has a great impact on virus endemicity and health system policy.
Thrace, in South-East Europe, is located in the East side of Northern Greece and borders with Eastern Turkey and Northern Bulgaria. Since the implementation of mass vaccination program in 1994 in Thrace, there has been a significant reduction of 50% in perinatal and horizontal transmission of HBV infection [Papoutselis et al., 2001]. However, the area is still considered to be of intermediate endemicity, hepatitis B surface antigen (HbsAg) positivity ranges from 2% to 10%, because of increased immigration from areas of countries where HBV is endemic (the former USSR, Turkey, Bulgaria, Northeast Asia) and because of endemic regions with low socioeconomic conditions where Greek Muslims born and live in Thrace with their own religious and cultural characteristics [Papoutselis et al., 2001].
The clinical course and outcome of hepatitis B infection is complex and heterogeneous, and is influenced by many factors, including viral factors (HBV
genotype, viral mutations, level of HBV replication), host factors (gender, age, and immune status), and other factors such as HCV, HDV co-infections, or alcohol consumption [Lee, 1997; Sanchez-Tapias et al., 2002; Farci, 2003; Lok and McMahon, 2004]. As the result of viral-host interplay, chronicHBVinfection is characterized by an early replicative phase with enhanced viral replication (phase of immune tolerance), the immune phase with active liver disease (hepatitis B e antigen (HBeAg)-positive chronic hepatitis), and a late, low or non-replicative phase with HBeAg seroconversion and remission of liver disease (inactive carrier state) [Chisari and Ferrari, 1995; Lee, 1997]. After HBeAg seroconversion, some patients may continue to exhibit active hepatitis due to the emergence and activation of HBV variants not expressing HBeAg (HBeAg-negative chronic hepatitis) [Hadziyannis and Vassilopoulos, 2001a,b]. Approximately 15%-40% of people who develop chronic HBV infection progress to cirrhosis and end-stage liver disease, and 25% of these will develop HCC [Hoofnagle, 2000; Sanchez-Tapias et al., 2002].
The clinical course and long-term prognosis of HBV infection have been investigated in a number of longitudinal studies in the Western world [Dragosic et al., 1987; De Franchis et al., 1993; Villeneuve et al., 1994; Manno et al., 2004]. However, these studies have no follow-up of histological activity. Some other studies have either evaluated a heterogeneous study population in relation to the severity of liver disease at enrollment [Weissberg et al., 1984; Liaw et al., 1988; De Jongh et al., 1992] or have included both treated and untreated patients [Di Marco et al., 1999] or have included selected groups such as blood donors [Dragosic et al., 1987; De Franchis et al., 1993; Manno et al., 2004]. Studies
providing Information about the outcome of chronic HBV infection in the general population are lacking.
The long-term outcome of chronic HBsAg carriers in the general population of Thrace, during a follow-up period of up to 12 years (1992-2004) is described, and clinical, biochemical, virological, and histological parameters were evaluated prospectively. This study is a part of the Interreg I and II EC project.
PATIENTS AND METHODS
Protocol
The aim of the protocol was to study prospectively the natural history of patients with chronic HBV-infection in the general population in Thrace, a geographical region in the North of Greece with an estimated population of 350,000 people. Patients eligible for this prospective study were native individuals of Greek origin born in Thrace who were HBsAg(.) for at least 6 months. All patients were referred from the EC Interreg I and II cohort-studies, carried out between 1992-1994 and 1998-2002, with the aim of investigating the endemicity of HBV in the general population in Thrace.
Patients with active liver disease who had been treated in the past or started treatment during the study period were excluded from further evaluation.
Patients withHCV co-infection (all had detectable HCVRNA and undetectable HBV-DNA) were also excluded from the study. Patients meeting the inclusion criteria were asked to give an informed consent in order to participate in a long-term follow-up study that included regular clinical, biochemical, serological examinations, and liver biopsies. At entry, patients were given a questionnaire aimed to collect demographic data as well as information on alcohol or hepatotoxic drug use and possible sources of infection.
Patients
Two hundred sixty three HBsAg(+) individuals, 161 males, and 102 females with a median age of 34 years (range 20-65) were observed consecutively over a 12
years-period time (1992-2004), with a median follow-up of 4.9 years, at the Unit of Preventive Medicine of Social Security Institute in Alexandroupolis.
During the observation period, physical, biochemical, and serological examinations were carried out every 6 months and measurement of serum HBV-DNA every 12 months. Liver biopsy was undertaken at presentation and every 2 years in patients with active disease, and every 4 years in those patients been at the inactive carrier state. Patients with normal liver biochemistry and undetectable serum HBV-DNA were excluded for liver biopsy.
The patients were divided into two groups according to the HBeAg status and then subdivided according to the presence of HBV-DNA and ALT levels.
Patients with HBsAg(+), HBeAg(-), anti-HBe(+) with persistently normal ALT levels for >12 months were considered to be at the inactive carrier state. Patients with elevated aminotransferases >1.5 the upper normal limit (UNL), either HBeAg positive or negative were considered to have active liver disease.
Methods
HBsAg, HBeAg, anti-HBe, anti-HBs, anti-d, and anti- HCV were measured by commercial immunoassays (Abbott Diagnostics, Chicago, IL).
Serum HBV-DNA analyzed quantitatively with the Amplicor HBV-Monitor kit (Roche Diagnostics Systems, F. Hoffmann-La Roche Ltd, Basel, Switzerland.). The assay is based on amplification of a known standard amount added to each tube. According to the manufacturers'package insert, the Amplicor HBV monitor Test shows linearity from 4 102 (lower detection limit) to 4x 107 HBV-DNA copies/ml. Predilution of serum with higher HBV-DNA levels was performed to extend the detection limit.
Histological evaluation. Liver biopsies were carried out with a Mengini needle (Hepafix 16 mm, Braun Meslurgens, Germany). All specimens were blind
examined by the same pathologist according to standard criteria [Scheur, 1991].
Statistical Analysis
Statistical analysis, mainly descriptive in nature, was carried out with the SPSS software (version 11.5, SPSS, Inc., Chicago, IL).
RESULTS
Demographics, Transmission of HBV, and Classification of Patients at Entry to the Study
The demographic features and the data regarding the possible route of HBV-infection of the 263 adults enrolled in the study are given in Table I. The mode of transmission was considered unknown when the source of infection could not be identified (35.5%). The prevalent mode of transmission was interfamily transmission (49%). HBsAg-positive mother (30%).
At enrollment, 249 of the 263 (94.7%) individuals were HBeAg(-)/anti-HBe(+) and only 14/263 (5.3%) HBeAg(+). In the HBeAg(+) group, normal or slightly
elevated serum ALT (<2 UNL) was observed in seven patients; all had detectable HBV-DNA in serum (median HBV-DNA levels 2.5 107 copies/ml (250 million), range 6.2 106-7.1 107). In the HBeAg(-)/anti-HBe(+) group, 201/249 (80.7%) individuals had normal serum ALT levels of whom 98 (48.75%) had detectable serum HBV-DNA (median HBV-DNA levels 2.6 103 copies/ml (26,000), range
0.042 104-1.9 104), and the rest 48/249 (19.3%) patients had elevated ALT (>1 UNL) with median serum HBV-DNA levels 3.8 106 (38 million) copies/ml (range
1.4 105-4 107 copies/ml). In total 160 of 263 (61%) had detectable serum HBV-DNA levels (Table II).
Natural History
ALT activity and HBV DNA viraemia.
An inactive carrier state was observed in 201 individuals, but 6 of them were excluded from further evaluation because of drug or alcohol related liver disease;
detectable HBV-DNA was observed in 98/195 (50.3%), at entry to the study. During the follow-up period, 6 patients lost serum HBV-DNA while 4/195 (2.1%)
patients exhibited HBV reactivation with increased ALT levels, a yearly incidence of 0.4%. These four inactive carriers had serum HBV-DNA levels >104
copies/ml at presentation. The 97 inactive carriers (49 males) with undetectable serum HBV-DNA at enrollment remained so during the follow-up period.
In contrast, among the 48 HBeAg(-)/anti-HBe(+) patients with active liver disease 12 (25%) had either flares of ALT activity with intermediately normal levels
(erratic pattern) and the rest 36 (75%) persistently abnormal ALT levels. In this group, the serum HBV-DNA levels during the observation period ranged from
7.1 104 to 6.9 107 copies/ml. In particular, the group characterized by the erratic pattern of ALT activity had serum HBV-DNA levels ranging from 7.1 104 to 3.2 107 copies/ml, while the group with persistent elevation of ALT serum HBV-DNA levels ranging from 2 105 to 6.9 107 copies/ml.
In the HBeAg(+) group, 7/14 (50%) remained HBeAg(+) with high HBV-DNA levels and normal or slightly elevated aminotransferases (<2 UNL) during
the observation period. The rest seven patients lost HBeAg and developed anti-HBe, an annual seroconversion rate of 10%; five patients entered biochemical
remission with undetectable serum HBV-DNA. A schematic presentation of the natural history of the chronic HBsAg carriers is shown in Figure 1.
Liver histology
Patients at the Inactive carrier state. Liver biopsy was carried out in 95 of 98 individuals with a median age of 36 years (range 20-45) who were at the inactive
carrier state and had detectable serum HBV-DNA; 92/95 (97%) had normal or near normal histological findings and 3/95 (3%) mild fibrosis. In almost all of them, 90/95 (95%), the liver histology remained unchanged at the 4 years follow-up liver biopsy. Only two of four patients who developed HBV reactivation showed deterioration of the histological features during the follow-up period (Table III).
Patients with active liver disease. At the entry of the study,
the 48 HBeAg(-)/antiHBe(+) patients with active liver disease, median age 46 years (range 30-65) males 31(65%), underwent liver biopsy. Histology showed moderate or severe necro-inflammatory changes in 29 (60%) and moderate to severe fibrosiscirrhosis in 22 (46%), (Table IV). During the observation period 4/48 (8%) had deterioration of the necro-inflammatory changes and 2/48 (4%) had progression of fibrosis, one patient with already cirrhosis at presentation
developed HCC (Table IV).
In the group of HBeAg(+) patients liver biopsy at enrollment showed mild necro-inflammatory changes in 9/14 (64%) and moderate in 5/14 (36%), moderate fibrosis was found in 3/14 (21%). Of those who remained HBeAg(+) during the follow-up period the liver histology remained unchanged. Among the seven patients who developed anti-HBe seroconversion, the five patients who entered biochemical remission had improvement of liver histology at the follow-up liver biopsy.
Seroconversions
HBeAg seroconversion.
HBeAg to anti-HBe seroconversion was observed in7 (5 females) of the 14 (50%) patients, with an estimated annual rate of 10% (Table V). All patients who lost HBeAg and developed anti-HBe had ALT levels >2 UNL at presentation, while those who remained HBeAg(+) had ALT levels <2 UNL during the follow-up period.
HBsAg seroconversion. HBsAg to anti-HBs seroconversion was observed in 10/195 (5.1%) patients at the inactive carrier state with an estimated annual rate of 1% (Table V). Additionally, six patients lost HBsAg (3.1%) without developing anti-HBs immunity. All patients who lost HBsAg during the follow-up period
were HBeAg( )/anti-HBe(.), had undetectable serum HBV-DNA and normal ALT levels.
|
|
| |
| |
|
|
|