| |
Impact of tenofovir-containing ART on chronic HBV in a cohort co-infected with HIV
|
| |
| |
Journal of Antimicrobial Chemotherapy 2005 56(6):1087-1093
Christoph Stephan1,*, Annemarie Berger2, Amina Carlebach1, Thomas Lutz3, Markus Bickel1, Stephan Klauke4, Schlomo Staszewski1 and Martin Stuermer2
1 HIV Research and Treatment Unit at Center for Internal Medicine, Johann Wolfgang Goethe-University Hospital Frankfurt, Germany; 2 Medical Virology Institute, Johann Wolfgang Goethe-University Hospital Frankfurt, Germany; 3 HIV Medical Office Gruneburgweg Frankfurt, Germany; 4 HIV Outpatient Clinic Stresemannallee Frankfurt, Germany
Abstract
Objectives: We studied the impact of tenofovir disoproxil fumarate, given as an antiretroviral medication, on patients with chronic hepatitis B virus (HBV) co-infection.
Methods: The polymerase gene-sequence evolution and quantitative HBV loads (HBVL) were observed for 48 weeks in patients taking tenofovir-containing antiretroviral therapy. The patients were grouped according to baseline strata: high-replicative virus (>6 log copies/mL), low-replicative virus at detectable virus loads (<6 log) and HBs-antigen-positive, HBV-DNA-negative individuals.
Results: Thirty-one patients were evaluated. The median decline in 20 patients with high-replicative HBV infection was -5.37 log (range: 3.57-7); 11 out of 20 decreased to undetectable levels (lower limit of detection = < 200 copies/mL) and another three were below 400 copies/mL. Out of six patients with detectable HBV-DNA at week 48 (HBVL result: range 3.36-4.32 log10), we were able to carry out a re-sequence in four patients. We did not observe relevant emerging resistance mutations, or a relevant virus load re-increase from nadir (>+0.5 log). The patients with low-replicative virus (n = 9) and the baseline DNA-negative patients (n = 2) had an undetectable HBV-DNA at week 48. Two patients became HBeAg-negative; one DNA-negative patient became HBsAg-negative.
Conclusions: Tenofovir is effective in treating HBV infection in HIV patients. Patients with high-replicative virus may benefit from this treatment strategy by a reduction in replicative status, a precondition for improved hepatic function. A few patients showed low-level HBV replication. Indicators for clinical HBV-resistance to tenofovir were not observed.
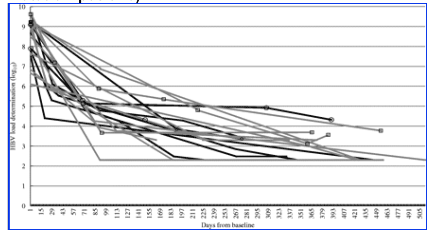
Figure 1. Hepatitis B virus load determination over the individual observation time for the high-replicative HBV patient group (n = 20). Grey lines represent individual virus load courses with a detectable baseline YMDD-mutation, black lines without YMDD-mutation. Six patients at suboptimal response (> lower limit of detection = 200 copies/mL) are pointed out by circles (YMDD-mutation-negative) and boxes (YMDD-mutation-positive).
Introduction
Approximately 5-10% of the general population infected with human immunodeficiency virus (HIV) suffer from an additional chronic co-infection with hepatitis B virus (HBV). Besides hepatitis C virus, HBV co-infection is an important morbidity and mortality factor for the HIV-infected population. Compared with the monoinfected, the course of a chronic HBV infection is more aggressive. Individuals with HIV co-infection tend to have lower transaminase elevation, higher HBV loads (HBVL), lower inflammatory activity and a higher prevalence of cirrhosis.1 In order to prevent the progression of HBV disease towards end-stage cirrhosis and hepatocellular carcinoma, it is crucial for the individual patient to convert from a high-replicative HBV infection into a low-replicative state.
Nucleoside analogues possess high antiviral efficacy and are well tolerated.2,3 They represent the primary treatment option for hepatitis B in the presence of an HIV co-infection4 and appear to be superior to interferon-.5 Currently available inhibitors of the HBV polymerase lamivudine and its sibling drug emtricitabine, adefovir and entecavir (recently approved for the US) have replaced famciclovir as treatment options for chronic HBV infection. Further drugs such as clevudine and telbivudine are currently under investigation. One problem in long-term lamivudine use as monotherapy of chronic hepatitis B infection is the emergence of viral resistance, as conferred by a mutation in the tyrosine-methionine-aspartate-aspartate motif (YMDD; rtM204V/I, where rt stands for reverse transcriptase) of the HBV polymerase gene. The presence of the YMDD mutation has been reported in 14-32% of patients who were not immunocompromised after 1 year on lamivudine monotherapy and in 69% of patients after 5 years. The emergence of viral resistance to lamivudine is even more rapid in individuals with HIV/HBV co-infection: the YMDD mutation was present in samples from 50% of such patients after 2 years and in 90% of patients after 4 years of lamivudine therapy. However lamivudine-resistant HBV is still susceptible to adefovir (Jules' NOTE: and entecavir). This nucleotide HBV-polymerase inhibitor seems to provide a higher barrier to viral resistance as emergence of adefovir resistance is rare during therapy though possible in patients with suboptimal virological response.
Tenofovir disoproxil fumarate is a nucleotide reverse transcriptase inhibitor (NRTI) and approved for combination antiretroviral treatment in HIV patients. Tenofovir has been shown to be effective in the treatment of high-replicative HBV infection in patients co-infected with HIV in a prospective study over 48 weeks. A sub-group analysis of a larger study compared the effect of tenofovir disoproxil fumarate versus tenofovir disoproxil fumarate + lamivudine on HBV, although with very small patient numbers.16 Cohort observations usually showed a favourable impact on chronic HBV co-infection if tenofovir was part of highly active antiretroviral therapy (HAART).17,18 One study demonstrated activity of tenofovir against lamivudine-resistant HBV in vivo for 24 weeks.19 Although large-scale studies with tenofovir are not yet available, it has been reported that this drug is at least not inferior to adefovir in the treatment of chronic hepatitis B infection in HIV patients.20
The objective of the current study was to follow the changes in HBVL and the evolution of the HBV genome in individuals with HIV/HBV co-infection who were receiving tenofovir as part of combination anti-HIV therapy. The secondary endpoints in the study were the HB seroconversion markers, including antibody status, surface- (HBs-) and e- (HBe-) antigen. This was an open observational subgroup analysis of all chronic HBV/HIV co-infected individuals from the Frankfurt HIV cohort. It began at the end of 2001 when tenofovir was available as an anti-HIV drug and ended in 2003. The median observation time was 48 weeks.
Discussion
This article describes the antiviral effect of tenofovir on HBV in 31 HBV/HIV co-infected patients from the Frankfurt HIV cohort. The main finding was that individuals with chronic HBV infection and detectable HBV at baseline responded well to this tenofovir-containing HAART, regardless of whether they had a high- or low-replicative HBV infection. All individuals with high-replicative HBV disease achieved a reduction of at least -3.5 log in HBVL after an individual observation time of a median of 48 weeks and most of these individuals achieved an HBVL of <400 copies/mL. All patients with low-replicative HBV infection at baseline turned to undetectable by HBV-DNA (<200 copies/mL) and HBsAg carriers remained DNA-negative. Patients with a detectable YMDD mutation at baseline, which confers resistance to lamivudine, responded as well to the tenofovir-containing antiretroviral regimen as those with wild-type HBV. The virological decay kinetics of HBV replication, when HIV co-infected patients were exposed to tenofovir, has been demonstrated recently.30 In contrast this study confirmed its impact on a clinical cohort.
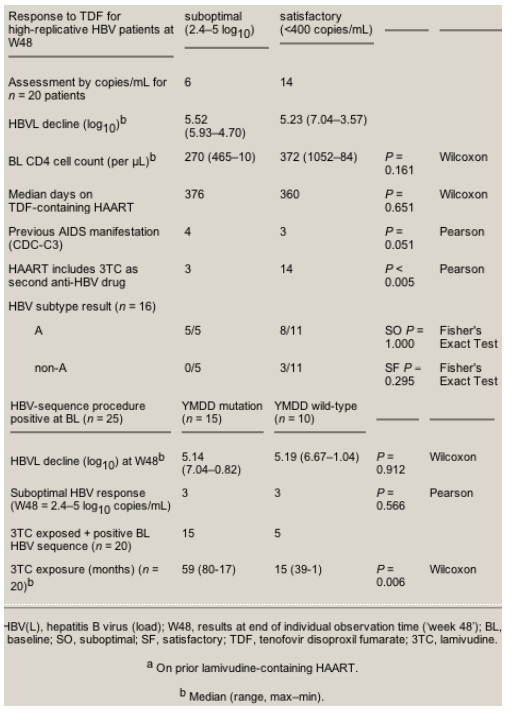
Although all high-replicative HBV carriers achieved low replicative status, they did not all achieve complete HBV suppression during the study period. A minority showed a suboptimal response characterized by low-level HBV replication while receiving tenofovir-containing HAART. The reason for this incomplete virus suppression is unknown, but suboptimal virus response in a proportion of patients at week 48 has been reported in previous studies.12,31,32 In the present study, this was not due to a shorter time on treatment for these patients. A lower CD4 cell count at baseline and experience of clinical AIDS was not significantly associated with better virological response; however the addition of lamivudine as second active anti-HBV drug was.
Hepatitis B antiviral monotherapy may result in the selection of resistance mutations leading to impaired drug susceptibility. This has been demonstrated for lamivudine10 and adefovir13,14 but not yet for tenofovir. This study confirmed once more that the emergence of YMDD mutations is significantly associated with exposure time to lamivudine. During the 48 weeks of tenofovir-containing HAART, we observed neither clinical resistance nor emergence of relevant new HBV mutations in patients with a suboptimal virological response on tenofovir. The patients with detectable YMDD mutations at baseline maintained them through the observation time.
We did not observe any true antibody seroconverters in our study-not surprising due to a rather short cumulative treatment and observation time. Usually a long treatment period is required in order to detect seroconverters.3 Nevertheless, HBeAg became undetectable in two patients while they were receiving tenofovir-containing HAART. One antiretroviral naive patient became HBsAg-negative, although he had been a documented HBsAg carrier for at least 7 years, which is a remarkable finding in an HBV/HIV co-infected patient.
We believe that HBVL measurements are superior to the serological control parameters when assessing the virological responses to treatment. High-replicative HBeAg minus variants may be missed when using only serological parameters. In our cohort we discovered two female HBeAg-negative patients with chronic aggressive HBV/HIV co-infection. This underscores the importance of using quantitative HBV determinations in HIV co-infected HBsAg carriers in order to assess individual treatment options.
For a decade, the primary interest in lamivudine was its anti-HIV activity. Perhaps understandably its anti-hepatitis B impact was neglected when faced with the global burden of HIV/AIDS. A consequence of long-term lamivudine experience is that many individuals with HBV/HIV co-infection exhibit HBV resistance to this drug. Moreover, the natural history of chronic HBV infection is critically distinct in the presence of HIV co-infection.1 This means that a robust and safe HBV treatment option is needed. Our results confirm in a clinical setting that tenofovir used as part of combination HAART regimens is an excellent treatment option for both lamivudine-naive and -experienced individuals-it provides an opportunity to 'hit two viruses' with one drug. The long term effect of this decline in HBV replication from high- to low- or undetectable DNA status cannot be assessed to date. However for high-replicative HBV co-infected HIV patients this is one precondition to delay liver disease progression. The limitations of our study are mainly due to its observational nature and 'real-life cohort' conditions. Since large prospective studies are unlikely to be conducted in this area, observational clinical cohort studies such as this are important in assessing the clinical effects of tenofovir.
RESULTS
Study cohort determination
Approximately 78% (n = 1854) of the 2369 patients who regularly attended the Frankfurt HIV cohort clinics in 2002 received HAART. During this first year of availability as an antiretroviral drug, 274 patients in the Frankfurt cohort received tenofovir, that is, a rate of 14.8% of all patients on HAART. In the whole Frankfurt HIV cohort 122 individuals were confirmed carriers of HBsAg, i.e. an HBV/HIV co-infection rate of 5.2%. The HBV/HIV co-infected group was more likely to receive HAART (n = 105/122, 86%), if compared with the whole cohort and a higher proportion of the co-infected received tenofovir in 2002 (n = 34/105, 32%).
Baseline study cohort characteristics and demographics
We were able to analyse data from 31 HBV/HIV co-infected patients who met the study criteria. None of the patients had a history of hepatic insufficiency. During the investigational tenofovir-containing HAART we could neither observe transaminase elevations grade 3 or 4 (WHO) nor abnormal liver function tests indicating any advanced liver disease (cirrhosis Child-Pugh class B or C). One patient also had an untreated chronic hepatitis C virus infection and all 31 individuals were naive to immunomodulator interferon-a or to other specific viral hepatitis-targeted therapy. None of the patients tested positive for hepatitis D serology.
All antiretroviral experienced patients (n = 24) were also lamivudine-experienced, and out of these four were subsequently placed on a lamivudine-sparing regimen, due to HIV resistance. All HAART-naive patients (n = 7, 22.6%) received lamivudine as second anti-HBV active component.
HBVL determination
All patients responded according to the previously defined definitions and showed a virus load decline during their individual observation times. The last sample was collected at day 366 (median) on the tenofovir-containing regimen, i.e. 'week 48' (range 149-556 days). None of the patients showed a virus load rebound of >+0.5 log during the observed tenofovir treatment which would indicate a clinically relevant resistance.
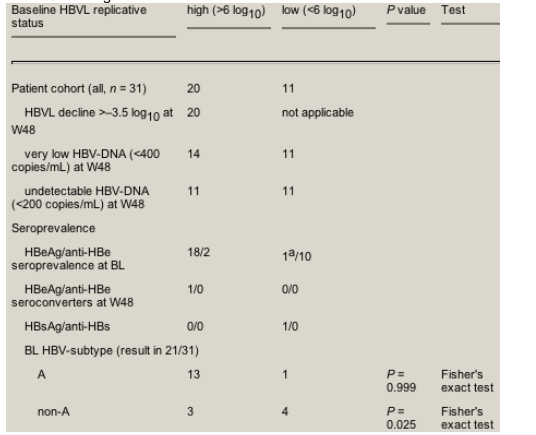
At baseline, the majority of patients (n = 20, 64.5%) had a high-replicative chronic HBV co-infection indicated by an HBVL of >6 log. Among these, two individuals were HBeAg-negative. The median virus load decline for all in this group was -5.37 log10 (range 7.04-3.57) (see Table 3). Eleven patients (35.5%) had an HBV replication activity of <6 log. Among these were nine patients with detectable HBV-DNA, including one HBeAg-positive individual on lamivudine and two with undetectable DNA (<200 copies/mL). All low-replicative patients decreased to undetectable DNA during tenofovir-containing HAART, the DNA-negative patients did not relapse. Another patient, known to be HBsAg-positive for 7 years and DNA-negative at baseline, became HBsAg-negative at week 48 on his first HAART that included lamivudine; seroconversion in HBs-antibody was not observed. A previously antiviral treatment-naive patient presented with the highest HBVL (4.32 log = 21 000 copies/mL at day 390) at week 48. His initial HAART regimen also included lamivudine as a second HBV-active drug. He reported compliance problems.
Table 3. Comparative subgroup analysis of HBVL and sequencing procedures of TDF-containing HAART
Genotyping
A baseline HBV sequence procedure was positive in 25/31 patients. A YMDD mutation was found in 15 out of these 25 HBV sequences. We were able to perform a positive HBV sequence in 20/24 patients with lamivudine-experience. Within these, five individuals (25%) had wild-type YMDD. The HBV sequence procedure was also positive in five of the 11 patients who had a baseline HBVL of <6 log10 copies/mL. Interestingly, in three of these five patients we were able to detect YMDD mutations.
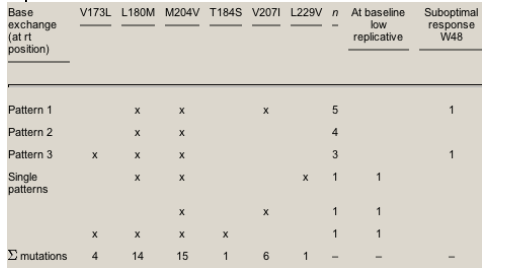
All YMDD mutational patterns that we were able to determine are highlighted in Table 4. The primary lamivudine mutation at position rt M204V was present in all.
Table 4. Baseline resistance patterns of YMDD mutations: findings in 15 HIV-positive, chronically HBV co-infected individuals with lamivudine experience
Due to the virological response to the tenofovir-containing HAART, the HBV-DNA from most patients with a baseline YMDD mutation could not be sequenced for a second time at the end of the individual observation time. However we were able to obtain follow-up sequences in four out of six patients with a suboptimal virological response: two patients with a baseline wild-type and two with a YMDD mutation. All four re-sequenced HBV strains showed the same pattern of mutations as those found at baseline-either wild-type HBV or the YMDD mutations, as were previously detected. One YMDD mutation-positive patient additionally showed a mixture base sequence change at positions rt M129M/V and rt V103V/I. These positions cannot be attributed to any specific domain (A-E) and are not regarded as relevant for emerging resistance. The other patient with a YMDD mutation and the two patients with wild-type HBV-DNA showed the identical base sequences that were found at baseline.
Patients and methods
Patients and design
The Frankfurt HIV cohort is a large German HIV treatment cohort of about 2500 continuously treated patients. At the time of first presentation the routine laboratory screening programme includes HBV-core antibody (anti-HBc) and HBs-antigen (HBsAg) and in the case of a positive result, the HBs- and HBe-antibody and HBe-antigen tests. A positive HBsAg result, confirmed after >6 months, sets the diagnosis of a chronic HBV infection, and the patient undergoes subsequent hepatitis D serology.
A database query identified patients who regularly visited the Frankfurt HIV cohort clinics (=/>3 visits per year) with chronic HBV co-infection and switch to tenofovir-containing HAART. A quantitative baseline HBVL assessment and an HBV polymerase sequence was obtained at the time of the switch to the tenofovir-containing HAART or within 12 weeks before from a frozen sample. Plasma samples for prospective HBVL assessments were subsequently collected. HBs- and HBe-antigen and -antibody tests were performed at baseline and at the end of the observation period.
All chronic HBV/HIV co-infected patients who switched to tenofovir disoproxil fumarate-containing HAART in 2002 or earlier, and for whom at least two subsequent samples on investigational treatment were available, were included in this cohort study. The intended observation time on treatment was 48 weeks. In the case of <48 weeks on tenofovir, the last available sample result was carried forward and analysed as the final result.
Patients who were lost to follow-up or whose anti-HIV therapy was changed or interrupted after <21 weeks on tenofovir or patients who underwent treatment with any other HBV-active drug except lamivudine were excluded from this observation. The demographic characteristics, individual anti-HIV drug history and the reason for changing anti-HIV therapy were recorded for all eligible patients.
The response to tenofovir treatment at the end of the individual observation time had to be analysed according to a pre-defined baseline HBVL stratification (Table 1).
Table 1. HBVL stratification

The routine laboratory tests included transaminase determinations and liver function tests, such as thromboplastin time by INR (where INR stands for international normalized ratio), serum cholinesterase and albumin. These parameters were observed in the course but not analysed as study endpoints.
Clinical HBV resistance was assumed if more than a +0.5 log re-increase from nadir in two subsequent quantitative HBVL determinations was detected. The prevalence of baseline YMDD mutations (rtM204V/I) was evaluated in relation to the individual time of previous lamivudine exposure. In the case of persistently detectable HBVL after at least 24 weeks on tenofovir, a second HBV sequence was carried out and compared with the baseline result in order to detect newly emerged mutations.
Methods
The hepatitis B serology tests used were the IMx-AxSYM HBc, HBe and HBs antibody tests and the microparticle enzyme immunoassay HBe/HBs antigen tests and hepatitis delta virus antibody tests (Abbott Laboratories, North Chicago, IL, USA). All tests were performed in the IMx or AxSYM system in accordance with the manufacturer's specifications.
The quantitative HBVL test assays provided a test result by virus equivalents in copies per mm3 of serum. The following two-step test kit combination was used. A quantitative hybridization method (measuring range: 1.4 x 105-1.7 x 109 copies/mL; Hybrid Capture II HBV test, Digene, MD, USA) determined high virus loads for all patients. If a patient went below the lower limit of detection of this method, the more sensitive quantitative PCR method (measuring range: 200-200 000 copies/mL; HBV DNA Monitor, Roche, Switzerland) was used.
Sequencing procedure
After viral DNA purification (using a QIAamp blood kit, Qiagen, Hilden, Germany) we amplified a 667 nt fragment using primers described by Thibault et al.21 The primers were modified at the 5' end by attaching the m13 universal and reverse primer sequence as previously described.22 The HBV-cycle-sequencing reaction was performed using the Big Dye Terminator Cycle Sequencing Kit version 2.0 (Applera Deutschland, Darmstadt, Germany) on an ABI 377-96 sequencer.
Sequence alignment and phylogenetic analysis
For the alignment of the HBV polymerase fragments, all isolates were trimmed to a similar range including amino acids rt79 to rt278 of the HBV polymerase using the amino acid nomenclature suggested by Stuyver et al.23 corresponding to positions 366-1025 in the HBV clone pEco63 (genotype A) genome (GenBank no. AY128092). Sequence alignments were performed using the MegAlign module of the LaserGene software24 and the Clustal V algorithm.25,26 Phylogenetic tree analyses were conducted using the Mega 2 software.27 Bootstrap tests with 10 000 replications based on the neighbour-joining method28 with the Kimura 2-parameter substitution module29 were performed to construct the phylogenetic trees.
Special attention was paid to mutations in the HBV polymerase protein which are known to be significantly associated with drug resistance to HBV treatment with famciclovir, lamivudine or adefovir.23 The focus of interest was turned to the following rt positions, with documented antiviral resistance for certain drugs (in parentheses), the corresponding primary mutations being underlined. Domain A: H90N/Y (famciclovir); domain B: F166L (lamivudine), V173L (famciclovir, lamivudine), P177L (famciclovir), L180M/V (lamivudine, famciclovir), A181V (adefovir), T184S (lamivudine, famciclovir), A200V (lamivudine); domain C: M204V/I (lamivudine), V207I (lamivudine, famciclovir), L229V (lamivudine); domain D: N236T (adefovir), N/S/H/A238S (famciclovir).
|
|
| |
| |
|
|
|