| |
PK of Ribavirin & NRTIs in APRICOT Study
|
| |
| |
"Effect of Ribavirin on Intracellular and Plasma Pharmacokinetics of Nucleoside Reverse Transcriptase Inhibitors in Patients with Human Immunodeficiency Virus-Hepatitis C Virus Coinfection: Results of a Randomized Clinical Study"
Antimicrobial Agents and Chemotherapy, October 2005,
M. Rodriguez-Torres,1* F. J. Torriani,2 V. Soriano,3 M. J. Borucki,4 E. Lissen,5 M. Sulkowski,6 D. Dieterich,7 K. Wang,8 J.-M. Gries,8 P. G. Hoggard,9 D. Back,9 for the APRICOT Study Group
Conclusions.
In patients with HIV-HCV coinfection who are receiving stable antiretroviral therapy, peginterferon alfa-2a (40KD) 180 µg/week plus RBV 800 mg/day does not perturb intracellular levels of the triphosphate anabolites of 3TC, d4T, or ZDV. RBV does not modify the plasma concentration-time profile of these NRTIs and control of HIV-1 RNA replication is not adversely affected. When the results of this pharmacokinetic analysis are considered together with the overall outcome of APRICOT, it is clear that combination therapy with peginterferon alfa-2a (40KD) plus RBV is both effective and safe and can be recommended for patients with HIV-HCV coinfection and compensated liver disease.
DISCUSSION
Our results suggest that RBV at a dosage of 800 mg/day does not have a clinically significant effect on the plasma pharmacokinetics or intracellular phosphorylation of 3TC, d4T, or ZDV in HIV-HCV-coinfected patients. There were no statistically significant differences in the plasma AUC0-12 h or Cmax of 3TC, d4T, or ZDV between patients treated with RBV or placebo. Moreover, the intracellular AUC0-12 h ratios of the triphosphate anabolites of 3TC, d4T, and ZDV to the corresponding endogenous nucleoside triphosphates in PBMCs were not changed significantly by treatment with RBV. This is important because the triphosphorylated anabolites of NRTIs, produced by intracellular kinases, drive both the efficacy and toxicity of these agents by competitively inhibiting reverse transcriptase in HIV-infected cells.
As evidenced by the wide standard deviations and broad confidence intervals, interpatient variation in the plasma and intracellular triphosphate concentrations for the three NRTIs was large but consistent throughout the study. The 95% confidence intervals are smaller for the 3TC data than ZDV and d4T (due to more patients receiving 3TC), and the evidence for an absence of an effect is largest for 3TC and less for ZDV and d4T. Nevertheless, these findings are not unexpected and are consistent with data from other studies of the NRTIs by several research groups using different methodologies. Large interpatient variation in both intracellular phosphorylation and plasma exposure of NRTIs have been reported previously (4, 17, 24, 28, 31, 33, 34, 38). In addition, wide variation also exists in the intracellular concentrations of endogenous nucleoside triphosphates (16, 17, 24, 37).
The mean steady-state plasma Cmax (2,771 ng/ml) and AUC0-12 (23,476 ng · h/ml) values of RBV in our study are in good agreement with those obtained with the same dosage of the drug (400 mg twice daily) in HIV-infected patients in an earlier study (2,440 ng/ml and 24,546 ng · h/ml, respectively) (25).
Our findings contrast with the results of in vitro studies that suggest that RBV inhibits the phosphorylation of ZDV (19, 38, 42) and d4T (15). These studies measured the total concentration of the phosphorylated metabolites of ZDV and d4T in PBMCs and human cell lines, including the monophosphate, diphosphate and triphosphate metabolites. RBV had the greatest impact on ZDV monophosphate levels, although the ratio of dTTP:ZDV-TP also increased in the presence of RBV (38). The results suggest that RBV increases intracellular formation of dTTP, which reduces the activity of thymidine kinase through feedback inhibition. The end result is a reduction in the phosphorylation of ZDV and d4T.
There are several possible explanations for the discrepancy between the findings in our study in patients with HIV-HCV coinfection and the findings of in vitro studies. The in vivo pharmacokinetics of RBV may be responsible to some extent for the observed differences. RBV is preferentially taken up by erythrocytes in vivo, which contributes to the large interpatient variability; a 60:1 red blood cell:plasma concentration ratio at steady state has been reported in HIV-infected patients receiving 800 mg/day (25). In addition to PBMCs, human cell lines were used in these investigations, which have different properties than cells in patients with HCV-HIV coinfection.
We measured the intracellular concentrations of endogenous nucleoside triphosphates and the triphosphorylated anabolites of 3TC, d4T, and ZDV after 8 to 12 weeks of continuous exposure to RBV, which is considerably longer than the brief exposure in the in vitro experiments. The longer treatment duration, large number of patients and the inclusion of a placebo group may also be responsible for the differences between this and the in vitro studies. It is possible that, after weeks of treatment, homeostatic mechanisms corrected transient perturbations that occurred shortly after the initiation of study treatment. Finally, it should be noted that other discrepancies between in vitro and in vivo studies have been reported: hydroxyurea has been consistently shown to increase intracellular levels of NRTIs in vitro (11, 12, 21) though the magnitude of changes in vivo are minimal.
The most important evidence of the absence of a clinically significant drug interaction between RBV and the nucleoside triphosphates is provided by HIV-1 levels in the two treatment groups. HIV-1 RNA levels did not increase in either treatment group during our study. This finding was also evident in the overall APRICOT population at the end of treatment: in patients who had detectable HIV-1 RNA at baseline, HIV-1 RNA levels were lower at the end of treatment (week 48) than at baseline in those treated with peginterferon alfa-2a (40KD) plus placebo and peginterferon alfa-2a (40KD) plus RBV (41). Moreover, the decreases were of similar magnitude (-0.7 to -0.8 log10 copies/ml). Reductions in HIV-1 levels have also been reported in patients with HIV-HCV coinfection after treatment with conventional interferon plus RBV combination therapy (6, 34). The CD4+ percentage increased in patients receiving combination therapy for HCV in APRICOT and in the other studies, a further indicator of ongoing control of HIV disease (6, 34, 41). These data should allay concerns that RBV may negatively affect control of HIV replication in HIV-HCV coinfected patients receiving antiretroviral therapy.
A study in patients with HIV-HCV coinfection who were randomized to conventional interferon, 3 MIU thrice weekly, plus RBV, 1,000 or 1,200 mg/day, or no treatment has also reported results contrary to those of in vitro investigations (34). RBV did not increase intracellular dTTP concentrations as predicted by in vitro studies; rather a transient decrease in dTTP concentrations was observed during the first month of RBV treatment. Serum HIV-1 RNA levels remained well controlled in both groups, similar to the findings in our study, and the combination regimen was judged to be effective and well tolerated for the treatment of HCV (34). The study reported a nonsignificant trend toward lower median peak and trough intracellular d4T-TP concentrations, a trend toward a decrease in the d4T-TP:dTTP ratio, and large intra- and interpatient variability (34). An increase in d4T-TP levels in the control group makes interpretation of the results difficult, as does the absence of a conventional interferon monotherapy control group. The overall findings are in agreement with those in our study that there is no evidence of a clinically significant interaction between RBV and d4T in patients with HIV-HCV coinfection.
Our study focused on the intracellular phosphorylation and plasma pharmacokinetics of 3TC, ZDV, and d4T and thus cannot be used to draw conclusions about the intracellular pharmacokinetics of all NRTIs in combination with RBV. Nonetheless, a broad spectrum of agents was used during the course of APRICOT, which suggests that antiretroviral therapy is compatible with peginterferon alfa-2a (40KD) plus RBV in HIV-HCV coinfected patients. The one possible exception to this generalization is didanosine (DDI), which has been associated with pancreatitis when given as monotherapy and when used in combination with RBV (29, 39). Only two patients received DDI during our nested pharmacokinetic study. An analysis of data from 14 cirrhotic patients who experienced hepatic decompensation during or after treatment in APRICOT revealed that use of DDI, among other factors, was significantly associated with this adverse event. Importantly, hepatic decompensation occurred with similar frequency in patients treated with or without RBV and thus did not appear to be exacerbated by the use of this drug. This is consistent with the absence of a pharmacokinetic interaction between these two drugs, as reported in a previous study, (20) but does not preclude an intracellular interaction. Nonetheless, there have been reports of severe mitochondrial toxicity, including fatalities, in patients receiving concurrent DDI and RBV (7-9, 13). Thus, use of DDI should be avoided in patients with HIV-HCV coinfection, particularly in those receiving RBV and those with cirrhosis (27, 39).
Conclusions. In patients with HIV-HCV coinfection who are receiving stable antiretroviral therapy, peginterferon alfa-2a (40KD) 180 µg/week plus RBV 800 mg/day does not perturb intracellular levels of the triphosphate anabolites of 3TC, d4T, or ZDV. RBV does not modify the plasma concentration-time profile of these NRTIs and control of HIV-1 RNA replication is not adversely affected. When the results of this pharmacokinetic analysis are considered together with the overall outcome of APRICOT, it is clear that combination therapy with peginterferon alfa-2a (40KD) plus RBV is both effective and safe and can be recommended for patients with HIV-HCV coinfection and compensated liver disease.
RESULTS
A total of 56 patients were enrolled in the pharmacokinetic substudy, 25 of whom were randomized (according to the APRICOT study protocol) to peginterferon alfa-2a (40KD) plus RBV and 31 to peginterferon alfa-2a (40KD) plus placebo. The baseline characteristics of the patients are summarized in Table 1. At the design of the trial, the pharmacokinetic population was defined as patients who received at least one dose of either study drug and had completed the baseline pharmacokinetic assessments. Baseline pharmacokinetic assessments were obtained in all but one patient (in the peginterferon alfa-2a [40KD] plus placebo group). During the course of the study five patients withdrew prematurely for non-safety-related reasons leaving 51 patients who completed treatment including 23 recipients of peginterferon alfa-2a (40KD) plus RBV and 28 of peginterferon alfa-2a (40KD) plus placebo.
Baseline antiretroviral therapy was well balanced between the treatment arms and was representative of antiretroviral therapy used at the time the trial was conducted (Table 1). The most common NRTI combination was 3TC plus d4T. The range, of sampling times for the steady state was 7.3 to 14.3 weeks.
Pharmacokinetic parameters were calculated using all available measurements. In some cases, data were missing because of laboratory errors, damaged samples or samples not being taken. In some patients who completed the study, concentrations below the limit of quantitation or missing data prevented calculation of the AUC0-12 h. As these cases were randomly distributed in both baseline and steady state data, they were considered unlikely to impact the Cmax evaluations. However, as a result, the numbers of patients with evaluable data were not always the same throughout the study period or for all the parameters reported.
Plasma pharmacokinetics of 3TC, d4T, and ZDV.
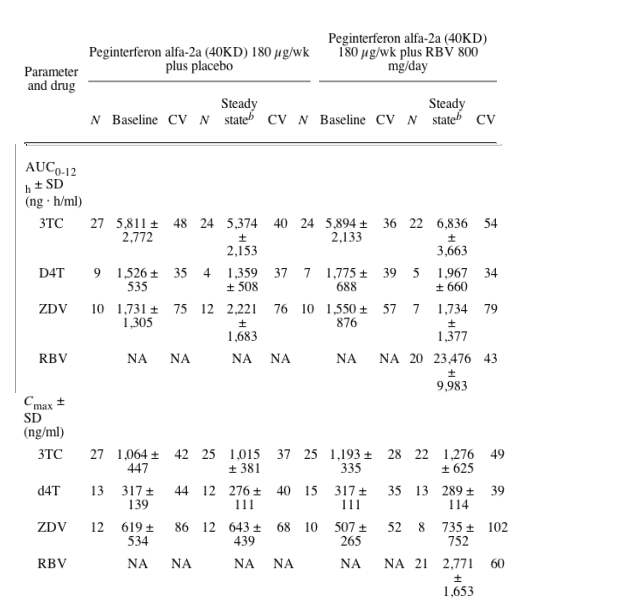
Mean plasma AUC0-12 h and Cmax values of 3TC, d4T, and ZDV during the 12-hour sampling interval at baseline and steady state are presented in Table 2. Based on an analysis of covariance, there were no statistically significant differences in the steady state plasma AUC0-12 h or Cmax values for 3TC, d4T, and ZDV between patients treated with RBV and placebo: the ratios of the least squares mean AUC0-12 h values (RBV:placebo) were 1.17 (95% confidence interval [CI], 0.91 to 1.51) for 3TC, 1.44 (95% CI, 0.58 to 3.60) for d4T and 0.85 (95% CI, 0.50 to 1.45) for ZDV, and the corresponding ratios for Cmax were 1.33 (95% CI, 0.99 to 1.78) for 3TC, 1.06 (95% CI, 0.68 to 1.65) for d4T, and 0.84 (95% CI, 0.46 to 1.53) for ZDV.
The changes in mean plasma concentration with time for 3TC, d4T, and ZDV are presented in Fig. 2. The profiles show considerable variability over the 12-h sampling time. In addition, some of the patient profiles, particularly for d4T show small increases at the end of the dosing interval. These findings were not significant and have not been noted previously and are most likely the result of the considerable variability. Importantly, the concentration-time profiles for each of the NRTIs show similar drug exposure at baseline and after 8 to 12 weeks of study treatment.
Intracellular phosphorylation of 3TC, d4T, and ZDV.
The intracellular AUC0-12 h values for the phosphorylated NRTIs and their corresponding endogenous nucleoside triphosphates are presented in Table 4. There was considerable variability in the mean AUC0-12 h values of dCTP and dTTP at baseline and steady state and there is considerable overlap of the standard deviations of the means when the mean values are compared. Moreover, there was no evidence of a significant interaction between RBV and either 3TC, d4T, or ZDV based on the ratio of the intracellular AUC0-12 h values of the respective NRTI triphosphates to the corresponding endogenous nucleoside triphosphates in PBMCs.
The differences at steady state between RBV and placebo in the least squares mean ratios of AUC0-12 h for each NRTI triphosphates to its corresponding endogenous nucleoside triphosphates are presented in Table 5. The differences (RBV minus placebo) were 0.274 (95% CI, -0.37 to 0.91) for 3TC-TP:dCTP, 0.009 (95% CI, -0.06 to 0.08) for d4T-TP:dTTP and -0.081 (95% CI, -0.40 to 0.24) for ZDV-TP:dTTP. The lack of any significant effect of treatment is further evidenced by the AUC ratios of intracellular NRTI triphosphates to the corresponding endogenous nucleoside triphosphates at baseline and steady state in individual patients (Fig. 3). Comparison of the ribavirin and placebo arms shows a similar pattern -considerable intrapatient variation was observed, but no trends were associated with treatment. Figure 3 also illustrates that there were no apparent changes in the AUC ratios of intracellular NRTI triphosphates to the corresponding endogenous nucleoside triphosphates between baseline and steady state in both the placebo and ribavirin treatment arms.
There were no statistically significant differences between the least squares mean AUC0-12 h values for the endogenous nucleoside triphosphates in patients treated with RBV and placebo for 8 to 12 weeks.
The mean intracellular concentration-time profiles for 3TC-TP, d4T-TP, ZDV-TP, dCTP and dTTP are presented in Fig. 4. The concentration-time profiles for each of these metabolites were similar at baseline and steady state with a broad overlap in the standard deviations at each time point. In agreement with previous studies (3, 31, 33), the intracellular triphosphate profiles of the NRTIs are much flatter than the plasma profiles of the parent drug.
Pharmacokinetics of RBV.
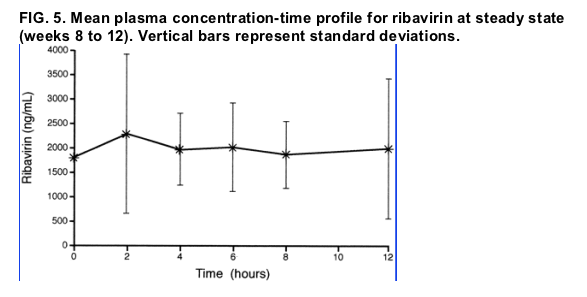
The plasma AUC0-12 h and Cmax values for RBV at steady state are presented in Table 2 and the plasma concentration-time profile at steady state is presented in Fig. 5. The mean steady-state plasma Cmax was 2,771 ng/ml, and the mean AUC0-12 h was 23,476 ng · h/ml.
Pharmacodynamic parameters.
At baseline most patients had undetectable serum HIV-1 RNA levels. There was no significant change from baseline in the median HIV-1 RNA level at week 4, 8, or 12 in either treatment group. There was no evidence of any increase in mean HIV-1 RNA levels in patients treated with either peginterferon alfa-2a (40KD) plus placebo or peginterferon alfa-2a (40KD) plus RBV at weeks 4, 8, and 12.
Mean and median CD4+ cell counts decreased in both treatment groups over the 12 week study period. The median change in CD4+ count at week 12 was -72 cells/µl (range, -377 to 756 cells/µl) and -87.5 cells/µl (range, -284 to 101 cells/µl) in patients treated with placebo and RBV, respectively.
Safety.
The safety profiles of peginterferon alfa-2a (40KD) plus placebo and peginterferon alfa-2a (40KD) plus RBV in this substudy reflected that reported in APRICOT (41). Modifications in the dose of peginterferon alfa-2a (40KD) because of adverse events or laboratory abnormalities were consistent with those reported in APRICOT.
In this substudy, serious adverse events were reported in 3 (10%) patients treated with peginterferon alfa-2a (40KD) plus placebo and 2 (8%) patients treated with peginterferon alfa-2a (40KD) plus RBV. Only two serious adverse events were considered related to treatment (pancreatitis in one placebo recipient and anemia in one RBV recipient). No deaths occurred during the pharmacokinetic substudy.
INTRODUCTION
Fundacion de Investigacion de Diego, Santurce, Puerto Rico,1 University of California, San Diego, California,2 Hospital Carlos III, Madrid, Spain,3 University of Texas Health Center, Tyler, Texas,4 Virgen del Rocío University Hospital, Seville, Spain,5 Johns Hopkins University School of Medicine, Baltimore, Maryland,6 Mt. Sinai School of Medicine, New York, New York,7 Roche, Nutley, New Jersey,8 University of Liverpool, Liverpool, United Kingdom9
Since the introduction of potent antiretroviral therapy, the life expectancy of patients with human immunodeficiency virus (HIV) infection has increased significantly. Coinfection with hepatitis C virus (HCV) in patients with HIV infection is common, and liver disease has emerged as a major cause of morbidity and mortality in HIV-HCV-coinfected patients (5, 39). It has been estimated that approximately 250,000 persons in the United States have HIV-HCV coinfection, which amounts to 10% of the total number of patients with chronic hepatitis C (2, 36), and approximately one-third of HIV-infected persons in the United States and Europe have HCV coinfection (32). Effective treatment for HCV is urgently needed in this population.
Sustained virological response rates of 52 to 63% have been obtained after 48 weeks of treatment with pegylated interferon plus ribavirin (RBV) in pivotal phase III studies in patients with HCV monoinfection (10, 14, 26, 43). As a result, this combination is recognized as the treatment of choice in this population (1, 39, 40).
RBV significantly enhances the efficacy of interferon-based therapies in the treatment of HCV. Whether RBV interferes with the pharmacokinetics of antiretroviral drugs, however, is an important and, as yet, unanswered question relevant to the treatment of HCV in HIV-infected persons. Concerns have also been raised regarding the safety of RBV in patients with HIV-HCV coinfection receiving antiretroviral therapy (39).
RBV inhibits IMP dehydrogenase and thereby alters various intracellular nucleotide pools. The drug reduces in vitro phosphorylation of certain pyrimidine analogue nucleoside reverse transcriptase inhibitors (NRTIs) (15, 19, 38, 42). The antiviral activity of NRTIs relies on conversion to pharmacologically active triphosphorylated moieties that competitively inhibit reverse transcriptase. The clinical significance of altered in vitro phosphorylation of NRTIs by RBV requires clarification because combination therapy with pegylated interferon plus RBV offers the best hope of a cure for HCV in patients with HIV coinfection (39).
In the randomized, multinational AIDS PEGASYS RBV International Coinfection Trial (APRICOT), the combination of peginterferon alfa-2a (40KD) plus RBV produced an overall SVR of 40%, which was significantly greater than that achieved with peginterferon alfa-2a (40KD) monotherapy (20%, P < 0.0001) or conventional interferon plus RBV (12%, P < 0.0001) (41). A nested pharmacokinetic study was incorporated into the design of APRICOT to determine the impact of RBV on the intracellular phosphorylation and plasma pharmacokinetics of NRTIs, the results of which are reported in this paper.
ABSTRACT
The intracellular triphosphorylation and plasma pharmacokinetics of lamivudine (3TC), stavudine (d4T), and zidovudine (ZDV) were assessed in a pharmacokinetic substudy, in 56 human immunodeficiency virus-hepatitis C virus (HIV-HCV) coinfected patients receiving peginterferon alfa-2a (40KD) 180 µg/week plus either placebo or ribavirin (RBV) 800 mg/day in the AIDS PEGASYS Ribavirin International Coinfection Trial.
There were no significant differences between patients treated with RBV and placebo in plasma pharmacokinetics parameters for the nucleoside reverse transcriptase inhibitors (NRTIs) at steady state (weeks 8 to 12): ratios of least squares mean of area under the plasma concentration-time curve (AUC0-12 h) were 1.17 (95% confidence interval, 0.91 to 1.51) for 3TC, 1.44 (95% confidence interval, 0.58 to 3.60) for d4T and 0.85 (95% confidence interval, 0.50 to 1.45) for ZDV, and ratios of least squares mean plasma Cmax were 1.33 (95% confidence interval, 0.99 to 1.78), 1.06 (95% confidence interval, 0.68 to 1.65), and 0.84 (95% confidence interval, 0.46 to 1.53), respectively.
Concentrations of NRTI triphosphate (TP) metabolites in relation to those of the triphosphates of endogenous deoxythymidine-triphosphate (dTTP) and deoxcytidine-triphosphate (dCTP) were similar in the RBV and placebo groups.
Differences (RBV to placebo) in least squares mean ratios of AUC0-12 h at steady state were 0.274 (95% confidence interval, -0.37 to 0.91) for 3TC-TP:dCTP, 0.009 (95% confidence interval, -0.06 to 0.08) for d4T-TP:dTTP, and -0.081 (95% confidence interval, -0.40 to 0.24) for ZDV-TP:dTTP. RBV did not adversely affect HIV-1 replication.
In summary, RBV 800 mg/day administered in combination with peginterferon alfa-2a (40KD) does not significantly affect the intracellular phosphorylation or plasma pharmacokinetics of 3TC, d4T, and ZDV in HIV-HCV-coinfected patients.
|
|
| |
| |
|
|
|