| |
Effects of chronic liver disease on bone mineral density and bone metabolism markers in postmenopausal women
|
| |
| |
European Journal of Obstetrics & Gynecology and Reproductive Biology
Volume 123, Issue 1, 1 November 2005, Pages 67-71
.... we suggest that the severity and prevalence of postmenopausal osteoporosis may significantly increase in patients with chronic liver disease....
..... In patients with liver disease, the mean BMD (p = 0.008) and T-score values (p = 0.005) were significantly lower at the lumbar spine than in control.....
......In this cross-sectional study, we showed that postmenopausal women with CLD have reduced BMD values compared with healthy postmenopausal women..... With the use of DEXA at four bone sites, we demonstrated a higher prevalence (72%) of osteoporosis in postmenopausal women with chronic liver disease than healthy postmenopausal women (33%).....
..... The treatment regimens to increase bone mineral density values in CLD patients are not well studied. In addition, safety and efficacy of estrogen therapy in preventing bone loss in these patients are obscure. In a cohort study, Menon et al. demonstrated that estrogen replacement therapy in postmenopausal women with primary biliary cirrhosis is safe and effective in reducing bone loss.....
1. Introduction
Osteoporosis is the predominant component of hepatic osteodystrophy and is present in 20-50% of patients with CLD, based on BMD measurements [1] and [2]. Osteomalacia, due to concomitant Vitamin D deficiency and calcium malabsorbtion, was traditionally considered to be the major bone disorder in patients with CLD, but is found in only a minority of patients [3].
Osteoporosis in patients with hepatic disease affects mainly trabecular bone and has been characterized by low bone turnover with reduced osteoblast function and low serum osteocalcin levels [4]. The pathogenesis of osteoporosis in CLD is unknown and there is much controversy about the risk factors for osteoporosis in CLD. The presence of liver cirrhosis, hypogonadism, steroid therapy and calcium malabsorbtion have been reported as risk factors for metabolic bone disease in CLD [5]. However, it is still uncertain whether CLD per se is a risk factor for osteoporosis in postmenopausal women and no study has so far examined this important issue.
We therefore conducted a cross-sectional, comparative study between 22 postmenopausal women with CLD and 30 postmenopausal healthy subjects. The aim of our study was to evaluate BMD and bone metabolism markers to characterize bone remodeling in these patients.
Authors- Sevinc Uretmena, Mert Golb, , , Dilek Cimrinc and Esra Irmakd
aIzmir Ataturk Research and Training Hospital, Department of Internal Medicine, Turkey
bDokuz Eylul University, Faculty of Medicine, Department of Obstetrics & Gynecology, Inciralti, Izmir, Turkey
cDokuz Eylul University, Faculty of Medicine, Department of Clinical Biochemistry, Turkey
dAtaturk Research and Training Hospital, Department of Internal Medicine, Turkey
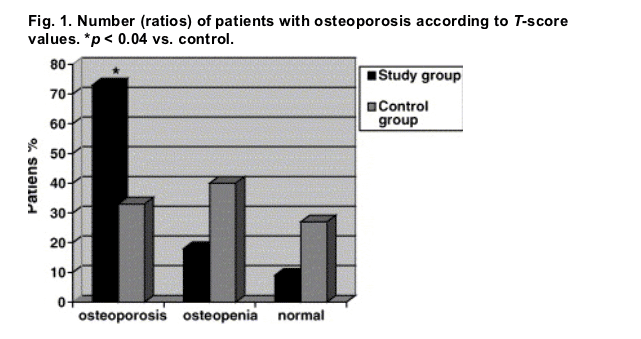
Fig. 1. Number (ratios) of patients with osteoporosis according to T-score values. *p < 0.04 vs. control.
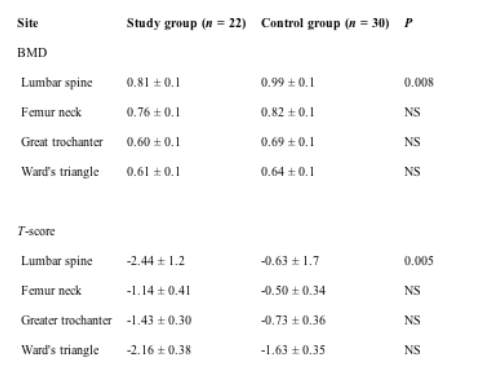
Bone mineral density values (g/cm2) and age-gender-adjusted bone density values (T-scores) of both groups are summarized in Table 3. In patients with liver disease, the mean BMD (p = 0.008) and T-score values (p = 0.005) were significantly lower at the lumbar spine than in control.
Table 3. Comparison of mean bone mineral density (BMD, g/cm2) and T-score values of the four sites studied between both groups
Abstract
Objectives: Patients with chronic liver disease (CLD) have an increased prevalence of osteoporosis. However, there is limited information about the effects of end-stage liver disease on bone metabolism and bone mineral density values in postmenopausal women. The aim of this study was to investigate the effects of chronic liver disease on bone mineral density (BMD) and bone metabolism markers in postmenopausal women.
Methods: We studied BMD by using dual-energy X-ray absorptiometry (DEXA), biochemical markers of bone turnover and calcium-parathyroid hormone (PTH)-Vitamin D axis in 22 postmenopausal women with CLD. Control group consisted of randomly recruited 30 healthy postmenopausal women.
Results
Based on WHO criteria, the prevalence of osteoporosis was significantly higher in patients with CLD (72 versus 33%) compared to healthy postmenopausal women. Bone loss was more significant at the lumbar spine than femur in the study group. Urinary excretion of bone resorption marker deoxypyridinoline was increased significantly in patients with CLD. There were no significant differences in bone formation markers (osteocalcin and bone alkaline phosphatase) between the groups.
Conclusions: CLD increases the prevalence of osteoporosis in postmenopausal women, particularly in the lumbar spine. Increased bone resorption seems to be the main reason for osteoporosis in these patients. We suggest that treatment strategies should be improved in these women to protect them from subsequent fractures.
3. Results
Etiologic diseases for chronic liver disease were: viral hepatic cirrhosis (14 patients), fatty liver steotosis (six patients), and primary biliary cirrhosis (two patients), respectively. The severity of chronic liver disease according to the Child classification were class B for every patient. None of the patients had undergone liver transplantation, nor had any bone fracture during the study.
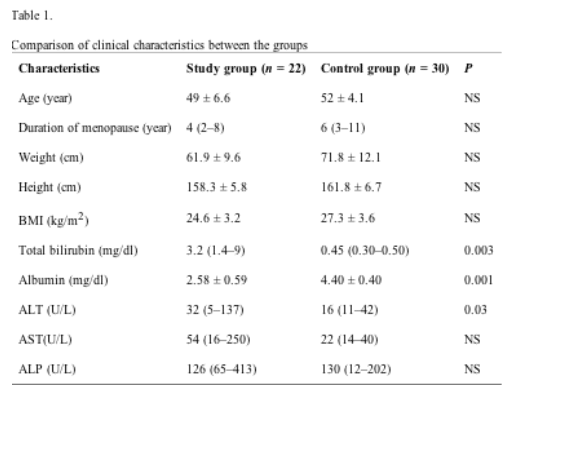
Clinical characteristics of the two groups are illustrated in Table 1. There were no significant differences in the mean age, duration of menopause, weight, height, and BMI values between the groups. In the study group, total bilirubin (p = 0.003) and albumin (p = 0.001) levels were significantly higher and lower compared to controls.
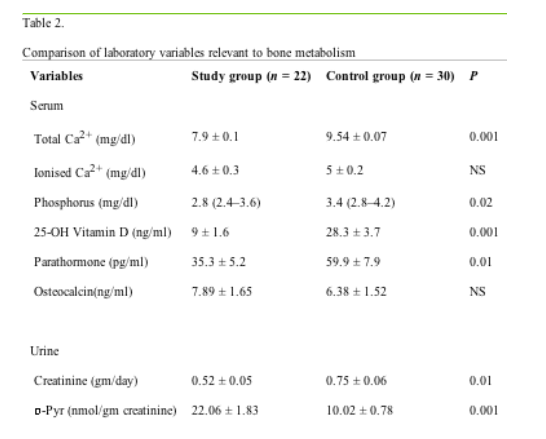
Table 2 shows values of laboratory variables particularly relevant to calcium, phosphorus and bone metabolism. The mean total calcium (p = 0.001) and phophorus (p = 0.02) levels were significantly lower in the study group than control; however, similar ionized calcium levels between the groups suggests that the decrease in total calcium was related to a decrease in levels of serum albumin in the study group. Although the mean serum parathormone and 25-hydroxy Vitamin D levels were within the normal reference ranges, serum 25-hydroxy Vitamin D (p = 0.001) and parathormone (p = 0.01) levels were significantly lower in the study group than control. There was no significant difference in the mean serum osteocalsin levels between the groups. Mean urinary excreation of creatinine was significantly lower in the study group than in control (p = 0.01), whereas mean urinary excreation of deoxypyridinoline was significantly higher in the study group than in control (p = 0.001). No significant difference was found in serum ALP levels between the groups.
Sixteen patients (72%) in the study group and 10 (33%) in the control group had osteoporosis. This difference was significant (p = 0.04) (Fig. 1).
Although the mean femur neck, Ward's triangle, greater trochanter BMD and T-score values were lower in the study group than control, these were not found to be statistically significant.
Significant correlations existed between bone densities at the four sites studied. Pearson correlation coefficients were as follows: lumbar spine and femoral neck, r = 0.807, 95% CI = 0.38-0.96; lumbar spine and Ward's triangle, r = 0.828, 95% CI = 0.27-0.83; lumbar spine and greater trochanter, r = 0.810, 95% CI = 0.43-1.49; femoral neck and Ward's triangle, r = 0.939, 95% CI = 0.58-1.03; femoral neck and greater trochanter, r = 0.935, 95% CI = 1.02-1.85 and Ward's triangle and greater trochanter r = 0.844, 95% CI = 0.78-2.23.
In the study group, serum total bilirubin levels showed significant negative correlation with lumbar spine (r = 0.91), Ward's triangle (0.96), and femoral neck (r = 0.90). All other measured parameters relevant to liver disease and bone metabolism did not show any correlations with bone mineral density values.
4. Discussion
In this cross-sectional study, we showed that postmenopausal women with CLD have reduced BMD values compared with healthy postmenopausal women. Diminished BMD has been reported in patients with CLD; nevertheless, most of these studies were performed in heterogenous groups regarding the gender, thus it is not clear whether CLD per se affects the incidence and severity of osteoporosis in postmenopausal women [6], [7] and [8].
BMD should be measured in women with strong risk factors for osteoporosis, such as cigarette smoking, thin habitus and early menopause. Several techniques are available for the measurement of BMD, but the most useful in clinical practise is DEXA. With the use of DEXA at four bone sites, we demonstrated a higher prevalence (72%) of osteoporosis in postmenopausal women with chronic liver disease than healthy postmenopausal women (33%). Direct comparisons cannot be performed between our study and previous studies due to the heterogenity of the diseases and subjects; however, we found high prevalence of osteoporosis in our study compared with these previous studies [6] and [7]. Crosbie et al. [6] reported 60% osteoporosis rate, whereas Bonkovsky et al. reported 40% osteoporosis rate in their study [7]. The low prevalence of osteoporosis in Crosbie's study compared to ours could also be the result of the younger age of subjects included in their study [6]. Furthermore, use of corticosteroid treatment in these studies could also have affected the results.
It has been shown that patients with cholestatic liver diseases, such as primary sclerosing cholangitis and primary biliary cirrhosis, have increased prevalence of osteoporosis than other chronic liver diseases [9] and [10]. Accordingly, Crosbie et al. [6] suggested that high prevalence of osteoporosis rate (60%) in their study is due to high number of patients, either with primary biliary cirrhosis or primary sclerosing cholangitis. In our study, the prevalence of osteoporosis was 72% in patients with primary biliary cirrhosis.
Bonkovsky et al. reported a prevalence of 40% osteoporosis rate in their study, in which there was also a high number of patients with cholestatic liver disease than the present study [7]. In agreement with our results, Ormarsdottir et al. [11] reported higher prevalence of osteoporosis in female subjects with chronic liver disease than the male subjects and suggested that female sex is one of the most important factors determining the outcomes in their study.
Most of the previous studies have shown similar prevalence of osteoporosis in all measured areas [6] and [7]. In contrast with these studies, we found significantly lower BMD values of the lumbar vertebra in the study group than in control. However, there were no significant differences in all measured femur sites between the groups. This difference between our study and previous studies may be the result of non-inclusion of male subjects in our study.
In the present study, we used current bone resorption and remodeling markers, such as deoxypyridinoline and osteocalsin. We found increased urinary excretion of deoxypyridinoline in the study group compared with the control, whereas serum osteocalsin levels were similar between the groups. These results may mean that the bone loss in chronic liver disease patients is not due to the diminished bone synthesis but due to the increased bone resorption in these patients. In agreement with our finding, Crosbie et al. [6] also found increased urinary excretion of deoxypyridinoline and normal serum osteocalsin levels in their subjects.
The importance of decreased serum 25-OH Vitamin D levels in chronic liver disease patients is not well-known. Two previous studies did not find any correlation between serum 25-OH Vitamin D levels and BMD values [7] and [12]. In contrast with these studies, Diamond et al. [3] reported a significant correlation between decreased level of 25-OH Vitamin D and BMD values in their patients with chronic liver disease.
In addition, Ormarsdottir et al. [11] concluded that bone loss in advanced liver disease is associated with decreased level of 25-OH Vitamin D. Although we found a decreased level of serum 25-OH Vitamin D in our subjects, we did not find significant correlation between serum 25-OH Vitamin D levels and BMD values. We suggest that serum 25-OH Vitamin D level is not a predictive marker for BMD values in patients with chronic liver disease. The same indefiniteness as in serum 25-OH Vitamin D levels also exists in serum PTH levels. There are conflicting results regarding the correlation of serum PTH levels and BMD values in chronic liver disease patients. Bonkovsky et al. [7] did not find any significant correlation between BMD values and serum PTH leves. In contrast, Crosbie et al. reported significant correlation between serum PTH levels and BMD values [6]. We also did not find any significant correlation between these two parameters and we suggest that osteoporosis may cause decreased BMD values in these patients, but not osteomalacia.
In this study, we found a significant negative correlation between serum total bilirubin levels and BMD values in CLD patients (Fig. 1). Gallego et al. [12] also reported a significant negative correlation between total bilirubin levels and BMD values in their subject. This may show the correlation between the severity of disease and decreased BMD values in patients with CLD.
The treatment regimens to increase bone mineral density values in CLD patients are not well studied. In addition, safety and efficacy of estrogen therapy in preventing bone loss in these patients are obscure. In a cohort study, Menon et al. [13] demonstrated that estrogen replacement therapy in postmenopausal women with primary biliary cirrhosis is safe and effective in reducing bone loss. A similar, recent study by Ormarsdottir et al. [14] showed effectiveness of transdermal estrogen use in increasing BMD values of patients with primary biliary cirrhosis. These studies seem to be promising regarding the use of estrogen therapy in this group of patients.
One of the limitations in our study could be the change in mean osteoporosis prevalence in the study group due to heterogenity of the etiologic diseases. It is well-known that patients with primary biliary cirrhosis have more decreased bone mineral density values compared to other etiologic diseases, such as viral hepatitis; therefore, osteoporosis rate in our study could be effected in correlation with the number of patients with primary biliary cirrhosis [6]. However, we had only two patients with PBC and after we excluded these two patients from the statistical analyses, the prevalence of osteoporosis did not change significantly in the study group. Accordingly, we do not think that the heterogenity of the etiologic diseases could have affected the overall result in the present study.
In summary, we suggest that the severity and prevalence of postmenopausal osteoporosis may significantly increase in patients with chronic liver disease. The main reason for this increase can be the result of augmented bone resorption in these patients. Further and large-scaled studies should be performed to reveal the exact mechanisms that underlie the etiology of osteoporosis of postmenopausal women with chronic liver disease. Subsequently, treatment strategies should be improved, particularly with anti-resorptive agents in these patients.
2. Materials and methods
Twenty-two postmenopausal women with chronic liver disease were included in this cross-sectional study. These women had their treatments and follow-up at the outpatient clinic of Gastroenterology Department at our instution. The diagnosis of liver disease was achived by biochemical, serological and histopathological investigations and with a duration of more than 6 months. Patients with malignancy or endocrine, cardiac or lung disease were not included in the study protocol. None of the patients were addicted to alcohol and were not smoking. Body weight and height were measured while the subjects were lightly clothed and shoeless. BMI was calculated as the weight (kg) divided by height (m2). Patients between the body mass index values of 20-30 kg/m2 were only included. Control group consisted of 30 healthy postmenopausal women. These women were selected by computer randomization method among postmenopausal women attending to our out-patient clinic who were not taking any drug related to the bone mineral metabolism. All subjects were postmenopausal (natural) with serum FSH levels greater than 30 mIU/ml and serum estradiol levels lower than 25 pq/ml.
None of the patients had received any of the medications related to bone mineral metabolism, such as calcium, estrogen, hormone replacement therapy, Vitamin D, corticosteroid, calcitonin, or biphosphonates for at least six months prior to the study. Patients and controls gave their informed consent to participation in the study.
2.1. Measurements of bone mineral density
The bone mineral density (g/cm2) of the second, third and fourth lumbar spine and three sites (femoral neck, Ward's triangle, and the greater trochanter) of the left proximal femur (if unaffected by disease, otherwise the right proximal femur) was measured. All measurements were conducted by (DEXA) (Hologic QDR1000, Hologic Inc., Waltham, MA). The in vivo DEXA precision was 0.76% for the lumbar spine and 1.0% for the femoral neck.
The WHO's definition of osteoporosis, BMD less than -2.5 standard deviations from the mean value of lumbar or proximal femur BMD in a sex-matched, young adult female reference population (denominated as BMD T-scores), was applied to classify individuals as either osteoporotic or not.
2.2. Biochemical tests
Morning blood and urine samples were drawn after an overnight fast. Analyses of aminotransferases, alkaline phosphatase, total bilirubin, albumin, phosphate, total and ionized calcium were measured with an automatic technique. Serum was assayed using commercially available kits for intact parathyroid hormone (PTH, Diagnosis Products Corporation, CA, USA, intra- and interassay variation 5.7 and 6.3%, respectively), 25 OHD (RIA, Diagnosis Products Corporation, CA, USA, intra- and interassay variation <8%), osteocalcin (Diagnosis Products Corporation, CA, USA, intra- and interassay variation <6%), FSH (Diagnosis Products Corporation, CA, USA, intra- and interassay variation 6.3 and 5.5%, respectively), estradiol (Diagnosis Products Corporation, CA, USA, intra- and interassay variation 6 and 11%, respectively), and urinary deoxypyridinoline (DPD, Diagnosis Products Corporation, CA, USA, intra- and interassay variation 6 and 14%, respectively). Urinary creatinine was measured with the ACA analyzer (DU Pont CO) following the manufacturer's instructions.
Statistical analysis was performed using SPSS for windows (rel. 10.0, SPSS Inc., 2001). All results were expressed as the mean �} S.E.M. Normal distributions of the values in both groups were verified with the Kolmogorov-Smirnoff test. The mean values of both groups were compared by the unpaired Student's two-tailed t-test and the Chi-square test with a Fisher's exact test to adjust for small numbers or nonparametric Mann-Whitney U-test as appropriate. Correlation studies were performed with Pearson standard linear regression analysis (normal distribution) or the Spearman test (non-normal distribution). A P-value of <0.05 was considered statistically significant.
References
[1] J.E. Hay, K.D. Lindor and R.H. Wiesner, The metabolic bone disease of primary sclerosing cholangitis, Hepatology 14 (1991), pp. 257-261.
[2] K.D. Lindor, C.H. Janes, J.S. Crippin, R.A. Jorgensen and E.R. Dickson, Bone disease in primary biliary cirrhosis: does ursodeoxycholic acid make a difference?, Hepatology 21 (1995), pp. 389-392. Abstract
[3] T.H. Diamond, D. Stiel, M. Lunzer, D. McDowall, R.P. Eckstein and S. Posen, Hepatic osteodystrophy static and dynamic bone histomorphometry and bone Gla-protein in 80 patients with chronic liver disease, Gastroenterology 96 (1989), pp. 213-221.
[4] S.F. Diamond-Hodgson, E.R. Dickson, H.W. Wahner, K.A. Johnson, K.G. Mann and B.L. Riggs, Bone loss and reduced osteoblast function in primary biliary cirrhosis, Ann Inter Med 103 (1985), pp. 855-860.
[5] J. Rose, J.E. Compston, E.O. Crawley, C. Evans and P. Smith, Osteoporosis associated with chronic liver disease, Eur J Gastroenterol Hepatol 3 (1991), pp. 63-69.
[6] O.M. Crosbie, R. Freaney, M.J. McKenna and J.E. Hegarty, Bone density, Vitamin D status, and disordered bone remodeling in end-stage chronic liver disease, Calcif Tissue Int 64 (1999), pp. 295-300.
[7] H.L. Bonkovsky, M. Hawkins, S. Karen, T. Hersh, J. Galambos and J.M. Henderson, Prevalence and prediction of osteopenia in chronic liver disease, Hepatology 12 (1990), pp. 273-280.
[8] T. Diamond, D. Stiel and S. Posen, Osteoporosis in hemochromatosis: iron excess, gonadal deficiency, or other factors?, Ann Inter Med 110 (1989), pp. 430-436.
[9] C. Trautwein, P. Possienke, H. Schlitt, H. Klaus, H. Roland and R. Rudolph et al., Bone density and metabolism in patients with viral hepatitis and cholestatic liver diseases before and after liver transplantation, Am J Gastroenterol 95 (2000), pp. 2343-2351.
[10] H. Rosen, Primary biliary cirrhosis and bone disease, Hepatology 21 (1996), pp. 253-255.
[11] S. Ormarsdottir, O. Ljunggren, H. Mallmin, H. Brahm and L. Loof, Low body mass index and use of corticosteroids, but not cholestasis, are risk factors for osteoporosis in patients with chronic liver diseases, J Hepatol 31 (1999), pp. 84-90.
[12] R.F. Gallego, G.J. Calvin, M.M. Torres, J. Mundi, R.F. Perez and D.R. Moreno, Bone mineral density, serum insulin-like growth factor I, and bone turnover markers in viral cirrhosis, Hepatology (1998), pp. 695-699.
[13] K.V. Menon, P. Angulo, G.M. Boe and K.D. Lindor, Safety and efficacy of estrogen therapy in preventing bone loss in primary biliary cirrhosis, Am J Gastroenterol 98 (2003), pp. 889-892.
[14] S. Ormarsdottir, H. Mallmin, T. Naessen, M. Petren-Mallmin, U. Broome and R. Hultcrantz et al., An open, randomized, controlled study of transdermal hormone replacement therapy on the rate of bone loss in primary biliary cirrhosis, J Inter
|
|
| |
| |
|
|
|