J Natl Cancer Inst 2005;97:577-586.
"The interaction between the associations of plasma HIV RNA level and CD4+ T-cell count with HPV infection was striking"
"Results showing that detectable HPV infection can recur regardless of sexual activity raises the possibility that periodic cervical cancer screening by Pap smear and/or testing for cervical HPV DNA may be necessary for HIV-positive and other immune-compromised women, even among those who are currently negative for HPV DNA and in a long-term monogamous relationships or who are celibate."
Author Discussion: Despite the high rates of HPV infection, cervical cancer rates are only moderately elevated in women with HIV/AIDS. This observation may largely be accounted for by the effectiveness of cervical cancer screening and treatment, but the modest associations of immune status with HPV persistence and with risk of HPV16 infection may also be relevant factors, especially in populations with limited cervical cancer screening. However, neoplastic cervical lesions remain very common in HIV-positive women, and because HIV-positive women live longer on highly active antiretroviral therapy, for the first time entering in large numbers the older age groups in which cervical cancer is more common, it will be important to monitor their cervical cancer rates. Despite our greater understanding of the natural history of HPV in HIV-positive women, we do not yet know the full long-term relationship between HIV coinfection and risk of HPV tumorigenesis in the era of highly active antiretroviral therapy.
We examined the association between HIV coinfection and the natural history of HPV, the viral cause of cervical cancer, in a large prospective cohort. Our main observations were 1) that plasma HIV RNA level and CD4+ T-cell count in combination appear to have a strong and statistically interactive association with both prevalent and incident detection of HPV, 2) that HIV RNA/CD4+ strata have a much stronger association with incident detection of HPV than with the persistence of HPV infection, and 3) that a substantial fraction of incident HPV detection in immune-compromised HIV-positive women is not related to recent sexual activity. These findings may have implications for our understanding of the natural history of HPV in HIV-positive women and could even provide insight into why, despite very high rates of HPV infection, the incidence of cervical cancer–albeit elevated–has not reached more epidemic proportions in women with HIV/AIDS.
These findings are consistent with previous data showing that both CD4+ T-cell count and plasma HIV RNA level contribute to the risk of opportunistic infections and AIDS.
A third major observation from this study was that both increased sexual transmission and a source (or sources) of HPV that is not related to recent sexual activity help account for the high rates of HPV detection in HIV-positive women. The sexual association of HPV infection was indicated by the statistically significant relationship observed between the number of sex partners in the last 6 months and incident detection of HPV.
Our results in women who have been celibate for at least 18 months provide strong evidence that incident HPV detection can occur in the absence of recent sexual activity.
Although reactivation of latent HPV infection is a likely explanation for these findings, other possibilities exist, such as autoinoculation (e.g., from the anal mucosa or other anogenital tissue reservoirs of HPV). It should be noted that nonpenetrative sexual contact in virgins is associated with risk of genital HPV infection (45), and there are laboratory data that suggest that HPV can resist desiccation, making it possible for HPV to be transmitted through indirect contact.
….findings are in keeping with the causal relation between HPV and SILs. We could not, however, study progression to high-grade SILs because of the small number of such lesions in our study.
"……Even those HIV-positive women with the best immune status (i.e., women with a CD4+ T-cell count of >500 cells per mm3 and an HIV RNA level of <4000 copies per mL) had a higher prevalence of HPV (PR = 1.970 than HIV-negative women. Moreover, in HIV-positive women with a CD4+ T-cell count of more than 500 cells per mm3 or 200—500 cells per mm3, the prevalence ratio increased with increasing HIV RNA levels (both Ptrend<.001)…." (see article text following Reuters report).
"….Even one male sexual partner among married women (presumably a monogamous relationship with the subject's husband) was associated with risk of incident HPV detection, and this risk increased with the number of male sexual partners up to at least three partners in the past 6 months. Condom use was associated with a statistically significant reduced risk of incident HPV detection only among women with at least three sexual partners…"
"….., the detection rate was, as expected, statistically significantly greater among the sexually active women….. a source (or sources) of HPV unrelated to recent sexual activity, such as reactivation of previously acquired HPV, may account for a substantial fraction of incidently detected HPV in HIV-positive women…."
NEW YORK (Reuters Health) - In HIV-infected women, the plasma HIV RNA level and the CD4+ cell count are strong determinants of the ability to detect human papillomavirus (HPV) on Pap smear, new research shows. This suggests that a weakening immune system may facilitate reactivation of HPV.
"Our data suggest that undetectable HPV infections become active much more frequently in HIV-positive women, which helps explain the extremely high rates of HPV infection in these women," lead author Dr. Howard D. Strickler, from the Albert Einstein College of Medicine in New York, said in a statement.
"By extension, our findings suggest that HIV-positive women and other women with poor immune status, such as transplant patients, may benefit from continued cervical screening even if they've long been celibate, since old HPV infections can rise again," he added.
Dr. Strickler's team analyzed data from 1848 HIV-positive and 514 HIV-negative women to assess factors that influence HPV detection. The findings are published in the April 20th issue of the Journal of the National Cancer Institute.
The HIV RNA level and the CD4+ cell count were strongly linked to both prevalent and incident HPV detection. The highest odds of detecting HPV were seen with HIV RNA levels > 100,000 copies/mL and CD4+ cell counts of < 200 cells/mL. The likelihood of detecting squamous intraepithelial lesions showed a similar association with HIV RNA level and CD4+ cell count.
The number of recent sexual partners was directly related to incident HPV detection in all women. However, 22% of sexually inactive HIV-infected women with low CD4+ cell counts still had at least one incidently detected HPV type.
The association between HIV coinfection and HPV persistence was more moderate, which could help explain why epidemic proportions of cervical cancer are not yet seen in HIV-infected women, the authors point out.
J Natl Cancer Inst 2005;97:577-586.
"Natural History and Possible Reactivation of Human Papillomavirus in Human Immunodeficiency Virus—Positive Women"
Journal of the National Cancer Institute
April 20, 2005
Howard D. Strickler, Robert D. Burk, Melissa Fazzari, Kathryn Anastos, Howard Minkoff, L. Stewart Massad, Charles Hall, Melanie Bacon, Alexandra M. Levine, D. Heather Watts, Michael J. Silverberg, Xiaonan Xue, Nicolas F. Schlecht, Sandra Melnick, Joel M. Palefsky
Affiliations of authors: Albert Einstein College of Medicine, Bronx, NY (HDS, RDB, MF, KA, CH, XX, NS); Maimonides Medical Center, Brooklyn, NY (HM); Southern Illinois University School of Medicine, Springfield (LSM); Georgetown University Medical Center, Washington, DC (MB); University of Southern California, Los Angeles (AML); National Institute of Child Health and Human Development, Bethesda, MD (DHW); Johns Hopkins University, Baltimore, MD (MJS); National Cancer Institute, Bethesda, MD (SM); University of California, San Francisco (JMP)
INTRODUCTION
Women with human immunodeficiency virus (HIV)/AIDS are at elevated risk of cervical cancer, as shown by studies of population-based U.S. AIDS and cancer registries (1,2). HIV-positive women also have extremely high rates of infection with human papillomavirus (HPV) (3—7), the sexually transmitted DNA virus that causes essentially all cervical cancer (8,9), and high rates of squamous cervical lesions (10—14). However, we lack information about several fundamental aspects of these relationships, such as the association between immune status–as characterized by both plasma HIV RNA level and CD4+ T-cell count–and the natural history of HPV. No prospective studies, to our knowledge, have assessed possible interactions between the associations of CD4+ T-cell and HIV RNA levels with either the incident detection or persistence of HPV (15,16), although a large cross-sectional investigation found a strong association between HIV RNA levels and HPV prevalence among women with a CD4+ T-cell count of greater than 200 cells per mm3 but not among women with lower CD4+ T-cell counts (3). It is, in addition, unknown whether the high rates of HPV infection in HIV-positive women are attributable mainly to increased incident detection of HPV infection, to increased HPV persistence, or to a combination of these. Furthermore, despite speculation that reactivation of previously acquired latent HPV accounts for much of the HPV detected in immune-compromised patients (13,17—19), this hypothesis has not been investigated carefully. Insight into the existence and nature of HPV reactivation, including its association with immune status, could have broad implications. For example, reactivation of latent HPV could contribute to the high rates of HPV infection and high-grade squamous intraepithelial lesions (SILs) observed in certain populations of elderly HIV-negative women, who may be undergoing age-related immune senescence (20).
We studied all available HPV DNA data from the Women's Interagency HIV Study, a large, long-term prospective cohort of HIV-positive and HIV-negative women, to assess how the combination of plasma HIV RNA level and CD4+ T-cell count is associated with the natural history of HPV infection. To seek evidence of HPV reactivation, we studied separately the subset of these women who were sexually inactive.
RESULTS
Subjects
A total of 1848 HIV-positive and 514 HIV-negative women with available HPV test results contributed data to our analysis of HPV natural history, including the prevalent and incident detection of HPV infection and its persistence (i.e., time to clearance). These data reflected a median of six follow-up visits of both HIV-positive and -negative women and a total of 4256 and 1405 person-years of observation, respectively, or 5661 person-years of observation total. Table 1 shows the baseline characteristics of the women in our analysis. Compared with HIV-positive women, HIV-negative women had a larger number of recent sexual partners (e.g., 23% of HIV-negative versus 15% of HIV-positive women reported two or more partners) and were more likely to currently smoke (61% versus 55%, respectively) and drink alcohol (62% versus 57%, respectively). HIV-negative women were also younger, had lower parity, and were less likely to report a history of injection drug use.
HPV Natural History
The associations of CD4+ T-cell count (measured as cells per mm3) and serum HIV RNA level (measured as copies per mL) with prevalent and incident detection and persistence (i.e., time to clearance) of HPV infection are shown in Fig. 1. HIV-negative women were used as the reference group in each analysis. Three strata of CD4+ T-cell count (>500, 200—500, and <200 T cells per mm3) and four strata of HIV RNA level (<4000, 4000—20 000, 20 001—100 000, and >100 000 copies per mL) were used to assess their combined associations (CD4+/HIV RNA strata) with HPV natural history. Adjustment for the covariates shown in Table 1 did not alter these results. Therefore, the results in Fig. 1 are for each CD4+/HIV RNA stratum, unadjusted for other covariates.
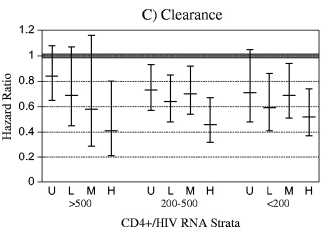
Figure 1.
Plasma human immunodeficiency virus (HIV) RNA level, CD4+ T-cell count, and the natural history of human papillomavirus (HPV) infection, including prevalent and incident detection of HPV and HPV persistence (as measured by time to clearance). HIV RNA strata are as follows: U (undetectable) = <4000 copies per mL, L (low) = 4000—20 000 copies per mL, M (moderate) = 20 001—100 000 copies per mL, and H (high) = >100 000 copies per mL. CD4+ T-cell count strata are as follows: >500 T-cells per mm3, 200—500 T-cells per mm3, and <200 T-cells per mm3. The estimates (bar) and their 95% confidence intervals (error bars) are shown. HIV-negative women served as the reference group. (A) Prevalent detection of HPV. The estimate is a prevalence ratio (i.e., the prevalence of HPV in a given CD4+/HIV RNA stratum relative to that in HIV-negative women), determined in generalized estimating equation models that adjusted for repeated measures. (B) Incident detection of HPV. The estimate for the incident detection is a hazard ratio, determined with multivariable Cox regression models. (C) Clearance of HPV infection. The estimate for the clearance of an incidentally detected HPV infection is a hazard ratio determined with multivariable Cox models (clearance was defined as two or more sequential negative results for each incidentally detected HPV type). The thick shaded horizontal line in each figure is the null result for each analysis and was added as a visual aid.
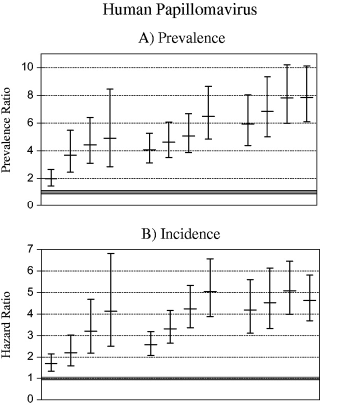
HPV prevalence and its association with CD4+/HIV RNA strata was assessed by use of prevalence ratios that were estimated in generalized estimating equation models that adjusted for repeated measures (Fig. 1, A). Even those HIV-positive women with the best immune status (i.e., women with a CD4+ T-cell count of >500 cells per mm3 and an HIV RNA level of <4000 copies per mL) had a higher prevalence of HPV (PR = 1.97; 95% confidence interval [CI] = 1.45 to 2.66) than HIV-negative women. Moreover, in HIV-positive women with a CD4+ T-cell count of more than 500 cells per mm3 or 200—500 cells per mm3, the prevalence ratio increased with increasing HIV RNA levels (both Ptrend<.001). Adjustment for the exact CD4+ T-cell count within these CD4+ T-cell strata did not alter the association of HIV RNA level with HPV prevalence, thus excluding the possibility that HIV RNA level might be acting only as a surrogate marker of lower CD4+ T-cell count within either CD4+ T-cell stratum.
We then divided women with a CD4+ T-cell count of less than 200 cells per mm3 into two groups, those with a CD4+ T-cell count of less than 50 cells per mm3 and those with a CD4+ T-cell count of 50—199 cells per mm3. As shown in Table 2, only among women with a CD4+ T-cell count of less than 50 cells per mm3 was there no association between HIV RNA level and HPV prevalence; in these women the prevalence ratios were all 7.81 or higher and did not vary with HIV RNA level (Ptrend = .68). In contrast, among women with a CD4+ T-cell count of 50—199 cells per mm3, the HIV RNA level had a statistically significant independent association with HPV prevalence (Ptrend = .004). Adjustment for covariates, including the use of highly active antiretroviral therapy modeled as a time-dependent covariate, did not change these findings (data not shown); these results are consistent with the principle that changes in the CD4+ T-cell count and HIV RNA level largely account for the relationship between highly active antiretroviral therapy and HPV. Finally, we confirmed the apparent interaction between the associations of CD4+ T-cell count and HIV RNA level with HPV prevalence: The rate of change in HPV prevalence per unit of HIV RNA decreased statistically significantly as the CD4+ T-cell count decreased (Pinteraction = .002).
Incident HPV detection (Fig. 1, B) was similar to HPV prevalence in its associations with CD4+/HIV RNA strata. The risk of incident HPV detection, as measured by hazard ratio, was higher in HIV-positive women than HIV-negative women, even among those HIV-positive women who had a CD4+ T-cell count of more than 500 cells per mm3 and an HIV RNA level of less than 4000 copies per mL (HR = 1.70, 95% CI = 1.35 to 2.15). Furthermore, there was an almost linear increase in hazard ratio with higher HIV RNA level (both Ptrend<.001) in women with a CD4+ T-cell count of more than 500 cells per mm3 or 200—500 cells per mm3. Adjustment for the exact CD4+ T-cell count within each CD4+ T-cell stratum did not alter the association of HIV RNA level with incident detection of HPV DNA in these women. Unlike the case with detection of prevalent HPV, however, an increased HIV RNA level was not associated with increased incident HPV detection among women with a CD4+ T-cell count of less than 200 cells per mm3, and the incident detection of HPV did not increase further (P = .88) with greater reduction of CD4+ T-cell count (Table 2). In fact, the risk of incident HPV detection was maximal (i.e., HR range = 4.0—5.0) in women with a CD4+ T-cell count of less than 200 cells per mm3 or an HIV RNA level of more than 100 000 copies per mL. The interaction between the associations of CD4+ T-cell count and HIV RNA level with incident detection of HPV was highly statistically significant (Pinteraction = .001).
The rate of clearance of an incidently detected HPV (Fig. 1, C) was statistically significantly lower in HIV-positive women than in HIV-negative women (HR = 0.67, 95% CI = 0.56 to 0.81). However, HIV RNA level was statistically significantly (and monotonically) associated with clearance only in women with a CD4+ T-cell count of more than 500 cells per mm3 (Ptrend = .03). Moreover, the association between CD4+/HIV RNA stratum and HPV clearance was clearly weaker than the association between CD4+/HIV RNA stratum and incident HPV detection. Hazard ratios for HPV clearance reached a trough in the range of 0.5—0.6 (an association roughly equivalent to a hazard ratio in the range of 1.7—2.0 for a positive relationship, as estimated by inversing the observed hazard ratios), whereas (as above) peak hazard ratios for incident detection were in the range of 4.0—5.0. The difference in the strength of the association of CD4+/HIV RNA stratum with incident HPV detection as compared with HPV clearance was highly statistically significant (P<.001).
Incident Detection of HPV in Sexually Active and Inactive Women
To study the extent to which the detection of HPV in HIV-positive women is related to recent sexually acquired HPV infection, we measured the association between the number of recent male sexual partners and the rate of incident HPV detection, controlling for the self-reported use of condoms (Table 3). Multivariable Cox models were used to adjust these results for CD4+ T-cell count; there were insufficient data to concurrently stratify by HIV RNA level. Even one male sexual partner among married women (presumably a monogamous relationship with the subject's husband) was associated with risk of incident HPV detection (HR = 1.19, 95% CI = 1.00 to 1.40), and this risk increased with the number of male sexual partners up to at least three partners in the past 6 months (Ptrend<.001). Condom use was associated with a statistically significant reduced risk of incident HPV detection only among women with at least three sexual partners (HR = 0.61, 95% CI = 0.41 to 0.92). Unexpectedly, condom users with three or more male sexual partners had a lower risk of incident HPV detection than condom users with two male sexual partners–an inconsistency that could suggest the reduced risk associated with condom use in those with at least three sexual partners was spurious. Unlike the number of sexual partners in the past 6 months, the lifetime number of sexual partners was not associated with incident detection of HPV, indicating that recent sexual exposure was the relevant sexual factor.
ABSTRACT
Background: Little is known in human immunodeficiency virus (HIV)—positive women about how the combination of plasma HIV RNA level and CD4+ T-cell count is associated with the natural history of human papillomavirus (HPV) infection or about HPV reactivation–whether it occurs and with what frequency in HIV-positive women.
Methods: HIV-positive (n = 1848) and -negative (n = 514) women were assessed at semiannual visits (total person-years = 5661) for cervicovaginal HPV with polymerase chain reaction assays and for squamous intraepithelial lesions (SILs) by Pap smear. We studied the prevalent detection of HPV and SILs with generalized estimating equations and the incident detection and persistence of HPV and SILs with multivariable Cox models. All statistical tests were two-sided.
Results: We observed a strong interaction between the associations of CD4+ and plasma HIV RNA strata with both prevalent (Pinteraction = .002) and incident (Pinteraction = .001) detection of HPV. Indeed, the hazard ratio for incident HPV detection peaked between 4.0 and 5.0, with either a CD4+ count of less than 200 cells per mm3 or an HIV RNA level of more than 100 000 copies per mL. Although incident HPV detection in all women was associated with the number of recent sex partners (Ptrend<.001), 22% of sexually inactive HIV-positive women with a CD4+ count of less than 200 cells/mm3 also had at least one incidently detected HPV type. The association between CD4+/HIV RNA strata and HPV persistence was statistically significantly smaller (P<.001) than for incident HPV detection. SIL prevalence, incident detection, and persistence had similar associations with CD4+/HIV RNA strata as HPV (above).
Conclusion: In HIV-positive women, plasma HIV RNA level and CD4+ count in combination appear to have a strong and statistically interactive association with incident detection of HPV, some of which may reflect HPV reactivation (e.g., in sexually inactive women). The more moderate association between HIV coinfection and HPV persistence could partly explain why cervical cancer rates have not reached more epidemic proportions in HIV-positive women.
FULL AUTHOR DISCUSSION
We examined the association between HIV coinfection and the natural history of HPV, the viral cause of cervical cancer, in a large prospective cohort. Our main observations were 1) that plasma HIV RNA level and CD4+ T-cell count in combination appear to have a strong and statistically interactive association with both prevalent and incident detection of HPV, 2) that HIV RNA/CD4+ strata have a much stronger association with incident detection of HPV than with the persistence of HPV infection, and 3) that a substantial fraction of incident HPV detection in immune-compromised HIV-positive women is not related to recent sexual activity. These findings may have implications for our understanding of the natural history of HPV in HIV-positive women and could even provide insight into why, despite very high rates of HPV infection, the incidence of cervical cancer–albeit elevated–has not reached more epidemic proportions in women with HIV/AIDS (1,2).
The interaction between the associations of plasma HIV RNA level and CD4+ T-cell count with HPV infection was striking. A high level of HIV RNA was strongly associated with prevalent and incident detection of HPV in women with either a high (>500 cells per mm3) or moderate (200—500 cells per mm3) CD4+ T-cell count but not in women with a low CD4+ T-cell count (<50 cells per mm3 for prevalent HPV detection and <200 cells per mm3 for incident HPV detection). As a result, there was often extensive overlap between the prevalent and incident HPV results in one CD4+ T-cell stratum and the next stratum, depending on the HIV RNA level, suggesting that a failure to categorize patients by combined CD4+/HIV RNA strata could cause substantial misclassification (the grouping of unalike patients) and lead to an imprecise estimation of the association between HIV coinfection and HPV prevalence. These findings are consistent with previous data showing that both CD4+ T-cell count and plasma HIV RNA level contribute to the risk of opportunistic infections and AIDS (33—39) but also raise an important concern. Few studies have a sufficiently large number of subjects to categorize HIV-positive patients by combined CD4+/HIV RNA strata. Even in this study we could not meet this requirement in certain analyses. This concern has research implications that go beyond our ability to characterize the association between HIV coinfection and HPV natural history. For example, in studies to elucidate the cofactors for cervical tumorigenesis (e.g., Chlamydia trachomatis or herpes simplex virus 2) in HIV-positive women, inadequate control for the interaction of HIV RNA and CD4+ strata could result in residual confounding. The greatest concern, though, relates to clinical trials. Studies to assess prevention (e.g., HPV vaccines) and treatment (e.g., ablative/destructive versus excisional treatments for cervical neoplasia) strategies for cervical cancer in HIV-positive women will have to have a sufficient number of participants to stratify patients by CD4+/HIV RNA strata to avoid a null bias, such as a failure to detect a decreased risk of high-grade cervical neoplasia in patients with a high CD4+ T-cell count because those with an HIV RNA level of more than 100 000 copies per mL did not respond.
A reasonable alternative to using multiple individual CD4+/HIV RNA strata might be to combine those strata that share similar associations with HPV infection and SILs, thereby reducing the number of subgroups and participants required in clinical trials and other studies. However, further investigation involving independent cohorts and perhaps a meta-analysis across studies is warranted before determining exactly which CD4+/HIV RNA strata should be combined when conducting clinical research.
Our second major observation was that HPV persistence, although elevated in HIV-positive women, was less strongly associated with CD4+/HIV RNA strata than was incident HPV detection. It appears, therefore, that the high rates of HPV infection in immune-compromised HIV-positive women are accounted for more by a pronounced increase in incident detection of HPV than by increases in persistence. Although a few smaller studies (15,16,40) have found stronger associations with HPV persistence than those that we observed, their main analyses combined prevalent (baseline) with incident HPV data. This approach would have caused a positive bias, because an already prevalent infection is more likely to persist longer than a new incident HPV infection (41). The limited subanalyses of incident HPV infections that were reported in those studies were not performed on a type-specific basis (40) or included a comparatively small number of infections and did not directly measure the duration of HPV infection (i.e., time to clearance) (15). In addition, a recent study among adolescent girls found an association between HIV coinfection and type-specific HPV persistence that was statistically significant but of moderate size, similar to our results (i.e., an approximately twofold-increased risk of HPV persistence in the most immune-compromised subjects) (42).
If further confirmed, our finding that HPV persistence is only moderately increased by HIV coinfection could provide important insight into why cervical cancer rates are only moderately elevated in women with HIV/AIDS. Persistent HPV infection is necessary for cervical tumorigenesis; therefore, a modest or moderate association between HIV coinfection and HPV persistence might limit the risk of cervical cancer in women with HIV/AIDS. We previously reported another possible reason that cervical cancer rates might be only moderately increased in HIV-positive women (7): Both incident and prevalent detection of HPV16 (the most important oncogenic HPV type, which alone accounts for approximately half of all cervical cancers) may be more weakly associated with immune status than those of other HPV types (7). If these observations are correct, then it is perhaps not so surprising that cervical cancer rates, although elevated in women with HIV/AIDS, do not approach the epidemic proportions of other virally related AIDS-associated malignancies, such as Kaposi's sarcoma or non-Hodgkin lymphoma (1,2), even in populations with limited cervical cancer screening (43,44).
A third major observation from this study was that both increased sexual transmission and a source (or sources) of HPV that is not related to recent sexual activity help account for the high rates of HPV detection in HIV-positive women. The sexual association of HPV infection was indicated by the statistically significant relationship observed between the number of sex partners in the last 6 months and incident detection of HPV. The data also seemed to suggest that condoms might protect against incident HPV in HIV-positive women with multiple recent sex partners. However, the results among condom users did not show the expected gradient across strata of sexual activity, complicating interpretation of the condom results.
Our results in women who have been celibate for at least 18 months provide strong evidence that incident HPV detection can occur in the absence of recent sexual activity. Moreover, the rate of incident HPV detection in celibate women was strongly associated with immune status; at least one new HPV type was detected in 22% of HIV-positive women with a CD4+ T-cell count of less than 200 cells per mm3, 9% of HIV-positive women with a CD4+ T-cell count of 200—500 cells per mm3, 7% of HIV-positive women with CD4+ T-cell count of more than 500 cells per mm3, and 5% of HIV-negative women. These rates were statistically significantly less than those in women who remained sexually active, but the absolute differences were fairly small. The findings undoubtedly underestimate the importance of sexual transmission because the limited data made it impossible to consider the specific number of recent sexual partners in the sexually active comparison group. However, these results suggest not only that there are sources of HPV unrelated to recent sexual behavior but also that these sources account for a high fraction of all newly detected HPV in HIV-positive women. Consistent with this, we found that more than 80% of women who met a stringent criterion thought to be consistent with HPV reactivation were HIV-positive and had a CD4+ T-cell count of less than 500 cells per mm3.
Although reactivation of latent HPV infection is a likely explanation for these findings, other possibilities exist, such as autoinoculation (e.g., from the anal mucosa or other anogenital tissue reservoirs of HPV). It should be noted that nonpenetrative sexual contact in virgins is associated with risk of genital HPV infection (45), and there are laboratory data that suggest that HPV can resist desiccation, making it possible for HPV to be transmitted through indirect contact (46). Thus, in theory, autoinoculation might occur through even routine activities related to personal hygiene (such as wiping when using the toilet), particularly in HIV-positive women who have high rates of anal HPV and a susceptibility to cervicovaginal HPV infection. In short, we cannot exclude autoinoculation as a source of cervicovaginal HPV infection in HIV-positive women. Despite this uncertainty, the clinical implications are straightforward. Results showing that detectable HPV infection can recur regardless of sexual activity raises the possibility that periodic cervical cancer screening by Pap smear and/or testing for cervical HPV DNA may be necessary for HIV-positive and other immune-compromised women, even among those who are currently negative for HPV DNA and in a long-term monogamous relationships or who are celibate.
An additional indication of the clinical significance of our HPV results was the similarity between the associations of CD4+/HIV RNA strata with both the natural history of SILs (i.e., prevalent and incident detection of SILs and the time to their clearance) and the natural history of HPV infection. These findings are in keeping with the causal relation between HPV and SILs. We could not, however, study progression to high-grade SILs because of the small number of such lesions in our study.
Our study has several additional limitations. The most important of these was the need to rely on patients' self-reported sexual behavior, which undoubtedly caused some degree of misclassification. That said, whether one is celibate or not is fairly straightforward, and it would have required gross misrepresentation by a number of subjects to account for the high rates of incident HPV detection among those who reported being sexually inactive.
Another important limitation was the lack of data regarding HIV viral load in cervical specimens. This lack of data is a concern because our findings might be interpreted as evidence against the hypothesis that HIV tat protein coactivates HPV in vivo (47—49). That is, if HIV had a direct biologic effect on HPV replication, we would not have expected the observed decline in the association between HIV RNA level and HPV infection as the CD4+ T-cell count declined. However, this issue is clouded by recent findings that HIV may be compartmentalized, as shown by at least moderate differences in the HIV viral load in blood and in cervical specimens (50,51). Consequently, future studies on similar cohorts should directly measure the HIV RNA levels in cervical specimens. These future studies could also help to determine whether cervical HIV RNA level has a greater association with HPV natural history than does plasma HIV RNA level and would provide insight into the importance of the local mucosal milieu as a factor in cervical neoplasia.
Although important questions remain, the data in this study, taken as a whole, add to a growing understanding of the relationship between HIV coinfection and HPV natural history. To summarize, our results suggest that the high rates of HPV infection in HIV-positive women may largely be accounted for by a very strong association of incident HPV detection with immune status, as characterized by the combination of CD4+ T-cell count and plasma HIV RNA level strata. When the CD4+ T-cell count is very low or the plasma HIV RNA level is very high, however, the other factor appears to be less biologically relevant. Furthermore, the source of incident HPV detection may vary. Greater susceptibility to the sexual transmission of HPV partly explains the high rates of incident HPV detection in HIV-positive women, but a large fraction of newly detected HPV may be unrelated to recent sexual activity. Whether this finding reflects the reactivation of latent cervical HPV or autoinoculation cannot be entirely determined from our results, but it is clear that immune status plays a major role, as shown by the strong association of CD4+ T-cell count with incident HPV detection in celibate women.
Despite the high rates of HPV infection, cervical cancer rates are only moderately elevated in women with HIV/AIDS. This observation may largely be accounted for by the effectiveness of cervical cancer screening and treatment, but the modest associations of immune status with HPV persistence and with risk of HPV16 infection may also be relevant factors, especially in populations with limited cervical cancer screening. However, neoplastic cervical lesions remain very common in HIV-positive women, and because HIV-positive women live longer on highly active antiretroviral therapy, for the first time entering in large numbers the older age groups in which cervical cancer is more common, it will be important to monitor their cervical cancer rates. Despite our greater understanding of the natural history of HPV in HIV-positive women, we do not yet know the full long-term relationship between HIV coinfection and risk of HPV tumorigenesis in the era of highly active antiretroviral therapy.
PATIENTS AND METHODS
Subjects and Specimens
Between October 1, 1994, and November 15, 1995, 2058 HIV-seropositive and 568 HIV-seronegative women older than 13 years provided written informed consent and were enrolled in the Women's Interagency HIV Study from similar clinical and outreach sources in Brooklyn and Bronx, NY; Chicago, IL; Los Angeles and San Francisco, CA; and Washington, DC (21). The Women's Interagency HIV Study's protocol was approved by each local institutional review board. Baseline data showed that HIV-positive women in the Women's Interagency HIV Study had demographic characteristics and risk behaviors similar to those of women with AIDS nationwide in the United States (21). Women in the Women's Interagency HIV Study cohort undergo a structured interview and a physical and gynecologic examination semiannually. Exfoliated cervicovaginal cells for HPV DNA testing are obtained by cervicovaginal lavage, and Pap smear material is collected with a wooden Ayre's spatula and a cytologic brush. The details of these methods have been previously described (7).
Clinical Laboratory Data
All available Pap smears were examined centrally at Dianon Systems (New York, NY) and categorized by use of the 1991 Bethesda System for cytologic diagnosis (22). Two independent cytotechnologists examined all Pap smears. Any Pap smear considered abnormal by either cytotechnologist, as well as 10% of all negative smears, were examined by a cytopathlogist. T-cell subsets were analyzed by flow cytometry in laboratories participating in the AIDS Clinical Trials Quality Assurance Program, as described previously (23,24). Plasma HIV RNA levels were measured through visit 7 with a nucleic acid sequence—based amplification technique (Organon Teknika, Durham, NC), the lower threshold of detection of which was 4000 copies per mL. Similar methods with greater sensitivity were used thereafter, as they became clinically available in these same quality-assured laboratories (e.g., quantitative polymerase chain reaction [PCR] assay by Roche Molecular Systems, Branchburg, NJ; lower limit of sensitivity = 400 copies per mL) (24,25).
Detection of HPV DNA
HPV DNA was detected with L1 consensus primer MY09/MY11/HMB01 PCR assays. Primer set PC04/GH20, which amplifies a 268—base-pair cellular {beta}-globin DNA fragment, was included in each assay as an internal control to assess the adequacy of amplification. Details of these methods have been previously reported (26), and the results were shown to have high reproducibility, sensitivity, and specificity (3,27,28). In brief, after proteinase K digestion, 2—10 µL of each cell digest was used in reaction mixtures containing 10 mM Tris—HCl, 50 mM KCl, 4 mM MgCl2, all four deoxyribonucleotide triphosphates (each at 200 µM), 2.5 U of AmpliTaq DNA polymerase, and 0.5 µM of each primer. There were 35 amplification cycles (95 °C for 20 seconds, 55 °C for 30 seconds, and 72 °C for 30 seconds), with a 5-minute extension period at 72 °C in the last cycle. Amplification products were probed for the presence of any HPV DNA with a generic probe mixture and probed for HPV DNA with filters individually hybridized with type-specific biotinylated oligonucleotide probes for HPV6, 11, 13, 16, 18, 26, 31, 32, 33, 34, 35, 39, 40, 42, 45, 51, 52, 53, 54, 55, 56, 57, 58, 59, 61, 62, 64, 66, 67, 68, 69, 70, 71 (AE8), 72, 73 (PAP238A), 81 (AE7), 82 (W13B and AE2), 83 (PAP291), 84 (PAP155), 85 (AE5), 89 (AE6), AE9, and AE10, as described previously (26,27).
Statistical Methods
Contingency tables were constructed from the data for each study visit. CD4+ T-cell counts were categorized into three conventional clinical strata that have been associated with HPV infection in previous studies [i.e., >500, 200—500, and <200 CD4+ cells per mm3 (3,7); for some analyses, the lowest stratum was divided further into 50—199 and <50 CD4+ cells per mm3]. Plasma HIV RNA levels were also divided a priori into four categories that were related to detection of HPV in an earlier study (i.e., <4000, 4000—20 000, 20 001—100 000, and >100 000 copies per mL) (3). The prevalence of HPV DNA was expressed as the percentage of women with adequate HPV test results (i.e., those in whom amplification of {beta}-globin was detected–an indicator that the PCR performed satisfactorily). Incident detection of HPV was defined as a positive test result for any HPV type that was not present at baseline or at any other earlier visit in a given woman. We use the term "incident detection" rather than "incidence" because the relative contributions of newly acquired HPV infections and reactivation of previously acquired latent HPV infections cannot be determined in a population with many years of sexual activity, regardless of the HIV status of that population. HPV persistence was measured as the time to clearance of a newly detected HPV type (making it possible to estimate the time of onset), with clearance defined by the stringent criterion of at least two sequential negative results to minimize concerns that a single false-negative result might affect the findings and also by the less stringent criterion of a single negative result. Both definitions of clearance gave similar results, and only findings that are based on the stringent criterion are presented.
To evaluate the study population in terms of additional factors (covariates), we initially divided the data by HIV status because HIV-negative women were used as the reference group in most analyses. The distributions of covariate data in HIV-positive and-negative women were compared by use of standard Pearson chi-square tests for simple contingency data or by the Mantel extension test when examining trends in ordinal data. These same variables were also assessed in the multivariable analyses described below to determine whether their inclusion in these models altered the estimated associations of HIV RNA level and CD4+ T-cell count strata with detection of HPV or SILs.
The cross-sectional associations of HIV RNA level and CD4+ T-cell count strata with prevalent detection of HPV DNA were measured by use of prevalence ratios (PRs), with HIV-negative women as the reference group (i.e., the prevalence of HPV in a given CD4+/HIV RNA stratum among HIV-positive women relative to that in HIV-negative women). These prevalence ratios were determined by use of marginal binary response—generalized estimating equation regression models that assumed an independence correlation structure to adjust the standard errors for repeated measures (i.e., summarizing data across all HPV types and across study visits). This approach addresses the facts that women may have multiple concurrent HPV infections and that the same women are being assessed over repeated visits, but it makes few assumptions regarding the nature of the intraindividual (i.e., subject specific) correlations. We have previously shown that the results in this dataset are unchanged by using other possible correlation structures (7), and, in general, model estimates are robust with respect to the correlation structure imposed (29). Because of the central etiologic role of HPV in essentially all neoplastic cervical lesions (a single causal pathway), generalized estimating equation models of HPV prevalence were not adjusted for the presence of lesions detected by Pap smear, which is consistent with the approach used in an earlier study (7). Treatment of cervical lesions was analyzed as a time-dependent covariate rather than by censoring at the time of treatment because previous analyses in the Women's Interagency HIV Study demonstrated that both approaches gave similar results (7). Other time-dependent covariates assessed included use of highly active antiretroviral therapy, HIV RNA level, CD4+ T-cell count, recent sexual behavior, and demographic variables that change over time (e.g., age). Time-independent variables included such factors as ethnicity. The two-sided Wald test was used to measure the statistical significance of the variables in these models.
To investigate the association of CD4+/HIV RNA strata with the incident detection of HPV, we used Cox regression models (30). The time of each incident event was defined as the midinterval (i.e., the midpoint calendar date between two consecutive visits), and HIV-negative women were used as the reference group. The Wei Lin Weissfeld marginal model approach was used to adjust the results for possible correlations between multiple incident infections (31) because, as above, it was important to control for the fact that woman with multiple infections contributed more than one observation. Both time-dependent and time-independent covariates were assessed in these multivariable Cox models. Patients who had missing data for two visits in a row for any reason were censored at the time of their last visit with complete data. Reanalysis of the dataset with censoring of women at the time they missed a single visit did not alter the results (data not shown). The two-sided Wald test was used to measure the statistical significance of the variables in the models. Because the variables of interest, HIV RNA level and CD4+ T-cell count, were necessarily modeled as time-dependent covariates, the proportional hazards assumption–that the instantaneous statistical associations being measured (i.e., hazard ratios [HRs]) are constant over the entire follow-up period–does not apply to this analysis (32). Instead, with the inclusion of time-dependent covariates in a multivariable Cox regression model, the instantaneous associations can vary with changes in the time-dependent covariates (32).
A similar Cox/Wei Lin Weissfeld model approach was used to measure the association of CD4+/HIV RNA strata with HPV persistence (i.e., the hazard ratio for HPV clearance). To determine whether the strength of association of CD4+/HIV RNA strata was greater for persistence versus incident detection of HPV, we used a single Wei Lin Weissfeld model stratified by outcome (i.e., persistence or incident detection) and the two-sided Wald test to calculate the P value for the hypothesis that there was no difference in the strength of the two associations.
To study possible incident detection of HPV that was not related to recent sexual exposure (e.g., HPV reactivation), we identified women who were sexually inactive (defined as the absence of any insertional vaginal or anal intercourse or oral—genital contact with either a man or a woman) for 18 months or more, the maximum period practical in our dataset before the number of subjects who met the selection criteria became too small for relevant analysis. For comparison, we identified a group of sexually active women who reported having at least one sexual partner during each of the three prior 6-month visits for a total of 18 months. We then measured the incident detection of a new HPV type among women in each group after a fixed period of follow-up (i.e., 2.5—3 years of follow-up; specifically, visit 5 or 6–whichever of the two visits in which a given subject first met the selection criteria for their group). It was important to carefully control for the period of follow-up because the number of different HPV types that can be newly or incidently detected may be reduced with every visit (i.e., once a particular type of HPV is detected, it is excluded from the pool of possible "new" HPV types that can be detected at later visits). The results were not altered by changing the period of sexual inactivity to 12 months (instead of 18 months) or by changing the exact visits selected for analysis (data not shown). The number (and percentage) of women in each group who had at least one incident HPV type detected was determined by CD4+ T-cell count stratum. We measured the associations between CD4+ T-cell count and incident HPV detection in each group with the Mantel extension test (for linear trends) and compared the incident detection rates between the sexually active and inactive groups (while accounting for the CD4+ T-cell count strata) with the Mantel—Haenszel chi-square test.
Our analyses of SILs–to determine the associations of CD4+/HIV RNA strata with prevalent detection, incident detection, and clearance of SILs–were conducted with the same generalized estimating equation and Cox/Wei Lin Weissfeld models described above, including the use of two separate definitions for clearance, one stringent (i.e., at least two sequential negative results) and one less stringent (i.e., a single negative result). Both definitions of clearance gave similar results, and only the results from the stringent criteria are presented. All statistical tests were two-sided.