| |
A Randomized Trial of the Efficacy and Safety of Fenofibrate versus Pravastatin in HIV-Infected Subjects with Lipid Abnormalities: AIDS Clinical Trials Group Study 5087
|
| |
| |
AIDS RESEARCH AND HUMAN RETROVIRUSES
Volume 21, Number 9, 2005, pp. 757-767
JUDITH A. ABERG,1 ROBERT A. ZACKIN,2,* SUSAN W. BROBST,3, SCOTT R. EVANS,2 BEVERLY L. ALSTON,4 W. KEITH HENRY,5 MARSHALL J. GLESBY,6 FRANCESCA J. TORRIANI,7 YIJUN YANG,2, SUSAN I. OWENS,8 and CARL J. FICHTENBAUM,9 for the ACTG 5087 STUDY TEAM
See end of article for affiliations.
*Dr. Robert Zackin was diagnosed with AIDS in 1992, and after receiving chemotherapy for Kaposi's sarcoma he developed heart failure in
1999. During this period he was integral in the design of A5087 and other AACTG studies. He underwent heart transplantation in February 2001.
His medical history was presented in N Engl J Med 2003;348:2323-2328 as the first successful heart transplant in an HIV-infected individual.
Throughout this time despite multiple hospitalizations, he remained active as the senior statistician participating on calls and sending us documents
electronically from his hospital bed even during his last hospitalization. Dr. Zackin died September 2, 2004.
See RESULTS & DISCUSSION following Abstract & Introduction.
ABSTRACT
There is a paucity of information on the safety and efficacy of lipid-lowering therapy for dyslipidemia associated with human immunodeficiency virus (HIV) and antiretroviral therapy. Our objective was to determine whether fenofibrate and pravastatin were equivalent for the treatment of combined dyslipidemia in HIV as measured by a composite of the National Cholesterol Education Project (NCEP) goals based on absolute values for low-density lipoprotein (LDL), triglycerides (TG), and high-density lipoprotein (HDL) and to compare the safety of these agents through 48 weeks.
This was a randomized, open-label trial with subjects assigned to fenofibrate 200 mg (n=88) or pravastatin 40 mg (n=86) daily. Subjects who failed to reach the
NCEP composite goal on monotherapy by week 12 received both drugs.
The composite goal at week 12 was achieved in 1% of fenofibrate and 5% of pravastatin subjects. At week 16, 69/88 subjects on fenofibrate added
pravastatin (FP) and 67/86 subjects on pravastatin added fenofibrate (PF). At week 48, 7% FP subjects and 3% PF subjects achieved the composite goal.
Median changes in LDL/HDL/TG/non-HDL were -8/ +5/ -144/ -50 and
-14/ +2/-66/ -34 mg/dl in subjects receiving FP and PF, respectively. There were few adverse events and no rhabdomyolysis reported.
Combination therapy with fenofibrate and pravastatin for HIV-related dyslipidemia provides substantial improvements in lipid parameters and appears safe, but is unlikely to achieve all NCEP targets for lipid levels.
INTRODUCTION
THERE IS GROWING CONCERN that the metabolic complications associated with HIV and antiretroviral therapy (ART) may lead to accelerated coronary heart disease (CHD).1-6 In addition to nonreversible risk factors such as male sex, age 40, and family history of premature CHD, other traditional cardiovascular
risk factors such as hypertension, dyslipidemia, diabetes mellitus, and visceral fat accumulation are increasingly seen in HIV patients on highly active antiretroviral therapy (HAART).1-6
In HIV-infected individuals on antiretroviral therapy, dyslipidemia is generally characterized by elevated cholesterol, triglycerides, LDL cholesterol, and low HDL cholesterol levels.6 The incidence of myocardial infarctions is higher in persons taking potent antiretroviral therapy, although the mechanism(s) for this association has not been clearly defined.1-4 In the D:A:D study, secondary analyses suggest that worsening of traditional risk factors like dyslipidemia is associated with a greater incidence of myocardial infarction.3
There are few published randomized studies of the treatment of dyslipidemia in HIV and no clinical end point studies documenting the efficacy of lipid-lowering therapy in this population. In the absence of clinical end point study data, it seems prudent to treat HIV-infected individuals with dyslipidemia in a manner similar to the general population per the recommendations of the National Cholesterol Education Program (NCEP).7-9
NCEP currently recommends that for patients with two or more CHD risk factors, the optimum LDL should be less than 100 mg/dl and triglycerides (TG) less than 200 mg/dl. In addition, NCEP recommends intensive therapeutic changes for individuals with the "metabolic syndrome," a condition increasingly being observed in persons with HIV10-12 and defined as the presence of three or more of the following: abdominal obesity, hypertriglyceridemia (TG 150 mg/dl), hypertension (>130/85 mm Hg), glucose intolerance (fasting blood sugar >110 mg/dl), and prothrombotic states. The metabolic syndrome has been associated with a very high risk for progression of coronary atherosclerosis in persons without HIV infection.13,14
Statins are the preferred lipid-lowering drug of choice for lowering LDL, whereas fibrates are the drug of choice for lowering TG.7 Combination therapy has been recommended for those who fail to respond to either class of medication alone.7
The safety and efficacy of these approaches in persons with HIV infection, particularly those taking potent antiretroviral therapy, are not known. In 1999, when this trial was designed, little was known about the mechanisms of HIV-associated dyslipidemia and there was uncertainty about whether specific lipid-lowering agents would work in this population. Furthermore, the type and incidence of specific lipid disorders in HIV were poorly characterized in 1999. Therefore, we designed a randomized trial comparing the NCEP-guided approaches in persons with HIV infection and combined dyslipidemia (elevated
LDL and TG), a population likely to be at greater risk of progression of atherosclerosis. Pravastatin and fenofibrate were chosen due to their lack of significant metabolism by the cytochrome P-450 system and results from pharmacokinetic studies15,16 suggesting that these drugs would be safe and tolerable in patients on potent antiretroviral therapy. We sought to determine
whether fenofibrate and pravastatin were equivalent with respect to clinical response and to define a therapeutic approach for management of dyslipidemia in HIV. Finally, we planned to compare the safety of these agents in the treatment of HIV-related dyslipidemia through 48 weeks.
RESULTS
One-hundred and seventy-four subjects were randomized-88 to fenofibrate and 86 to pravastatin. Baseline characteristics were balanced between groups (Table 1). One-hundred and seventy subjects had available 12-week lipid data. At week 12, one (1%) subject on fenofibrate and four subjects (5%) on pravastatin achieved composite goal (p=0.203, Table 2). Median changes in LDL/HDL/TG were +13/+ 4/ -118 and -30/0/ -27 mg/dl in subjects randomized to fenofibrate and pravastatin, respectively (Table 3). All p-values for within-treatment changes
were significant except both absolute and percent change in pravastatin HDL and pravastatin TG. Since only four subjects (2.3%) were missing primary end point data at week 12, sensitivity analyses revealed that results are robust to the handling of missing data. There were few ARV modifications during the initial 12-week period, and they were balanced between the arms (n=7 fenofibrate; n=8 pravastatin).
One-hundred and thirty-six subjects subsequently initiated dual therapy. At week 28, 64/88 subjects randomized to fenofibrate who subsequently added pravastatin (FP) and 66/86 subjects on pravastatin who added fenofibrate (PF) had lipid data available. Ten (16%) FP subjects achieved the composite goal, and three PF subjects (5%) achieved the goal (p=0.043, Table 2). Thirteen (20%) FP subjects and 14 (21%) PF subjects met the LDL goal (p=1.0). Forty-six (72%) FP subjects and 38 (58%) PF subjects met the HDL goal (p=0.101). Thirty-nine (61%) FP subjects met the TG goal, whereas only 25 (38%) PF subjects met the TG goal (p=0.014). Median changes in LDL/HDL/TG were -19/+ 6/ -158 and -15/ +2/ -81 mg/dl in subjects receiving FP and PF, respectively (p-values for withintreatment changes were significant for all absolute changes; percent
changes were significant except for FP LDL; only changes in TG were significantly different between treatments).
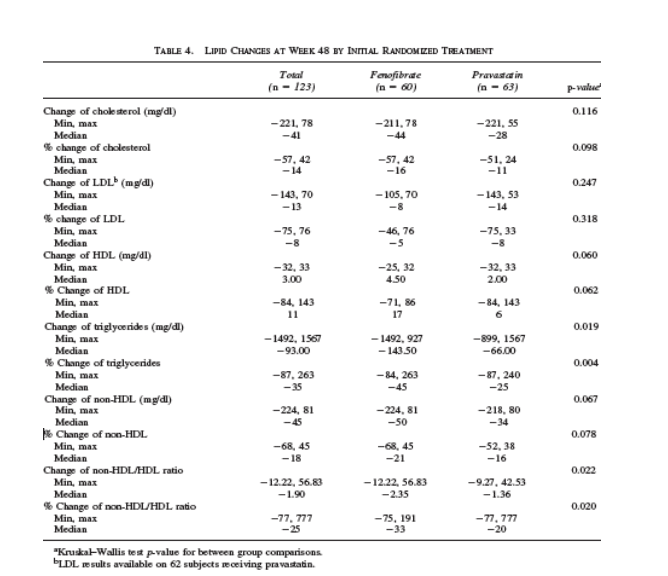
At week 48, 123 subjects had lipid data available; four (7%) FP subjects and two (3%) PF subjects achieved the composite goal (Table 2). Median changes in LDL/HDL/TG/non-HDL were -8/ +5/ -144/ -50 and -14/ +2/ -66/ -34 mg/dl in subjects receiving FP and PF, respectively (Table 4). There was a significant difference in both absolute and percent reduction of TG in subjects receiving fenofibrate first compared to those who started with pravastatin (p=0.019, p=0.004, respectively). p-values for within-treatment changes were significant for absolute changes except for FP LDL and PF HDL. For percent changes, all but PF HDL were significant.
Four subjects (three fenofibrate, one pravastatin) discontinued therapy due to protocol-defined toxicities in the first 12 weeks for the following reasons: (1) myalgias and elevated CK (not rhabdomyolysis), (2) asymptomatic elevation in CK, (3) pancreatitis (history of pancreatitis before study enrollment), and (4) asymptomatic elevation in lipase. Three subjects discontinued therapy between weeks 12 and 28 for protocol-defined toxicities: two with asymptomatic elevations in lipase and one with thrombocytopenia. Thirty-six percent of subjects experienced grade 3 or higher signs, symptoms, and/or laboratory toxicities. None of these events was attributed to study drugs. There were no reports of rhabdomyolysis or clinical hepatitis during the 48-week study period.
DISCUSSION
In this trial, we demonstrated that short-term use of pravastatin, fenofibrate, and the combination of the two appears to be safe and well-tolerated in persons with HIV infection. Subjects were using either protease inhibitor (PI)-containing (87%) or PI-sparing (13%) regimens. No significant changes from baseline were observed in HIV viral load or CD4 lymphocyte count in either treatment arm (data not shown) as were observed in the retrospective cohort study by Narayan et al.17 and the six subjects reported by del Real.18 There were no major safety issues; of importance, there were no cases of rhabdomyolysis. Though monotherapy with either pravastatin or fenofibrate provided significant reductions in lipid levels known to be associated with greater risk for the development of atherosclerosis, it was not effective in altering lipid levels to reach composite
goals in subjects with baseline elevations of both LDL and TG. This is an important strategic issue since clinicians typically initiate lipid-lowering therapy with the goal of reaching NCEP-recommended targets to reduce the risk of progression of atherosclerosis. Thus, with pravastatin monotherapy, 36% of
randomized subjects achieved LDL goals, 49% achieved HDL goals, and 18% achieved TG goals. Treatment with fenofibrate monotherapy led to 9%, 66%, and 48% of subjects achieving LDL, HDL, and TG goals, respectively.
Combination therapy resulted in 21%, 65%, and 49% of subjects meeting NCEP goals at week 28 for LDL, HDL, and TG, respectively. The strategy of whether to initiate a fibrate or statin first in combined dyslipidemia in persons with HIV infection is an important issue. We now recognize that there are specific mechanisms for the development of dyslipidemia related to the use of specific antiretroviral agents. Although many clinicians would recommend a statin initially, this approach may not be the most effective for drug-induced dyslipidemia. Of note, more subjects who initially received fenofibrate achieved a composite
goal at week 28 in contrast with those who initially received pravastatin (16% versus 5%). This significant difference disappeared by week 48, although a trend favoring fenofibrate was still present. This observation is most likely secondary to the reduction in TG that is associated with fibrates where the TG-depleted particles shift toward LDL, thereby increasing the LDL.19,20 This is best demonstrated in Fig. 2 in which the median LDL reduction at week 12 on pravastatin alone was 30 mg/dl and at week 28 following the addition of fenofibrate was only 14 mg/dl, demonstrating a loss of the beneficial LDL-lowering effect. Whereas the subjects on the fenofibrate arm had an increase in LDL at week 12, the median reductions of LDL at weeks 28 and 48 were similar to the pravastatin arm. The potential long-term clinical implications of these findings are unknown. An important objective of this study was to demonstrate in
a randomized trial that lipid-lowering agents traditionally used for non-HIV-associated dyslipidemia are effective for HIV-associated dyslipidemia. This information was not known at the time this trial was designed. Although combination therapy for combined dyslipidemia seems intuitively attractive, initial treatment with a statin plus a fibrate is generally not recommended for combined dyslipidemia. Furthermore, there were insufficient published data on the safety of statins and fibrates in HIV, thus a trial involving combination treatment with a statin plus a fibrate for combined dyslipidemia was warranted. In fact, the lipid-lowering effects on the individual TG and LDL values observed in our study are similar to those reported in trials of
pravastatin and fenofibrate in HIV-uninfected subjects.20-22 For example, in WOSCOPS (West of Scotland Coronary Prevention Study), 6595 men with elevated cholesterol levels who had not experienced MI were randomized to treatment with pravastatin or placebo and followed for an average of 5 years.21 In WOSCOPS, TGs were decreased by 20%, LDL was decreased by 26%, and HDL was increased by 5% with the use of pravastatin. This is similar to that seen in our pravastatin monotherapy arm. In a controlled trial of fenofibrate, 46 subjects with combined hyperlipidemia22 had reductions of 45% and 6% for
TG and LDL, respectively, while an increase of 15.3% was observed for HDL. The results of our study are also consistent with smaller, nonrandomized studies of statins and fibrates in persons with HIV infection.22-27 For example, Caramelli et al.23 reported a 45.7% reduction of TG in 13 patients treated with
fenofibrate. Calza et al.24 reported the use of fibrates resulted in reductions in TG of 41% and 23% for LDL while increasing HDL by 20% (n 69). Statins (n 37) reduced TG by 35% and LDL by 26% while HDL increased by 24%. Thus,
HIV-infected persons with combined dyslipidemia who are taking potent antiretroviral therapy can expect to derive significant benefits from lipid-lowering therapy.
Recent evidence28,29 suggests that aggressive lipid-lowering therapy in the general HIV-uninfected population with dyslipidemia will provide better outcomes than standard treatment. Using this treatment strategy for the HIV population could yield similar benefits. We used an aggressive approach to lipid-lowering
therapy in this trial with targets generally used for persons at greater risk of cardiovascular disease. There are emerging data to suggest that more potent lipid-lowering therapy results in regression of atherosclerosis.28 Thus, we thought it was important to target therapeutic goals that would be likely to imply a clinical benefit in the long term. Unfortunately few subjects reached these therapeutic targets in our study. Potential explanations for the overall modest response to reach the composite goal in A5087 include direct drug effects (especially from the PIs), drug-drug interactions, and host factors. Recent
data30 suggest that both PIs and nonnucleoside reverse transcriptase inhibitors reduce levels of pravastatin by about 50%. Thus, it is possible that higher doses of pravastatin might have improved the results observed in this trial. Indeed the use of other more potent statins may provide even further benefit. Nevertheless,
lowering of LDL will reduce the risk of CHD even among those who are unable to achieve goal.31 Furthermore, some of survival benefits of statin use may not be directly related to the effect on lowering lipids. There are data in HIV-infected
persons that inflammatory markers of atherosclerosis are elevated compared to the general population.32-35 Treatment with statins and fibrates affects many of these inflammatory markers, which may be independent of the alteration of circulating lipid levels. Results from the PROVE IT-TIMI 22 trial
demonstrated that patients who have low CRP levels after statin therapy have better clinical outcomes than those with higher CRP levels irrespective of the achieved level of LDL.36 Inflammatory marker analyses are planned to determine whether additional benefits were achieved in A5087 in the absence of
reaching the composite goal.
There are some important limitations of this trial. Because of the DSMB's decision to suspend enrollment into the monotherapy arms, the conclusions about the safety of these drugs in HIV should be considered preliminary. More safety data are needed to exclude the possibility of uncommon adverse
events. In non-HIV-infected individuals there are data suggesting that statins and fibrates may be combined safely for the treatment of dyslipidemia.37,38 Jones and Davidson37 reviewed reports of rhabdomyolysis obtained from an analysis of data from January 1, 1998 to March 31, 2002 from the United States Food and Drug Administration's Adverse Event Reporting System. They found that the use of fenofibrate in combination with statins results in fewer reports of rhabdomyolysis per million prescriptions dispensed than the use of gemfibrozil. In particular, of the total number of reports of rhabdomyolysis for fibrate and statin therapies, only 2.3% (14 of 606) were associated with fenofibrate/cerivastatin combination therapy, whereas 88% (533 of 606) were associated with gemfibrozil/cerivastatin combination therapy (140 reports per million prescriptions dispensed for fenofibrate versus 4600 reports for gemfibrozil). When used in combination with statins other than cerivastatin, the number of reports of rhabdomyolysis per million prescriptions dispensed was ~15 times lower for fenofibrate than for gemfibrozil (0.58 reports per million prescriptions dispensed
for fenofibrate versus 8.6 reports for gemfibrozil). Statin-fibrate combination treatment for up to 3 years in a cohort of 102 patients with coronary artery disease was not associated with serious disturbances in biochemical markers of muscle or liver function.38 Over a maximum 3 year follow-up no patient reported
myalgic symptoms and none had a measured CK activity >10 times above normal.
A5087 also suggests that the strategy to reduce lipids in combined dyslipidemia in HIV infection should begin with a fibrate. However, this may not apply in all circumstances where a more potent statin may be used. Furthermore, this approach may need to be modified based upon the likely mechanism of dyslipidemia. For example, if the primary mechanism is hypertriglyceridemia
due to induction of lipid production by ritonavir, a fibrate may be initially more successful than a statin. Whereas if hypertriglyceridemia is primarily due to insulin resistance with secondary adipocyte dysfunction, then an alternate approach might be more successful. Ultimately, these issues will require
further study. In conclusion, this study demonstrated that monotherapy with
either pravastatin or fenofibrate for HIV-related combined dyslipidemia appears safe but unlikely to achieve the composite goal.
Pravastatin appears to be mose effective in lowering LDL, while subjects receiving fenofibrate had larger increases in HDL and decreases in TG. Dual therapy appears safe. Dual therapy beginning with fenofibrate appears to result in additional lipid-lowering benefit. Pravastatin and fenofibrate provide reductions in TG and LDL and increases in HDL similar to those observed in other studies in HIV and non-HIV subjects. The clinical implications of this study suggest that HIV-infected persons with elevated LDL and TG will require more intensive lipid-lowering therapy to reach NCEP goals. It is important to note that the survival benefits derived from the use of statins are not solely related to their lipid-lowering effects. Thus, the failure to reach NCEP goals does not necessarily imply that lipid-lowering therapy is not beneficial. In general, clinicians should attempt to reduce LDL and TG levels below the NCEP guideline values when possible realizing that significant benefits are likely achieved with any reduction in these parameters. The relationship between LDL and CHD risk is continuous
so that lowering the LDL even if not reaching the NCEP goal can still be beneficial, especially at high LDL levels, since the relationship is log-linear.35 Further evaluation of different lipid-lowering agents including more potent statins, dosages, and novel strategies for the treatment of dyslipidemia is needed in persons with HIV infection and dyslipidemia.
MATERIALS AND METHODS
Treatment goals
The clinical goal of this study was based upon NCEP guidelines for the treatment of combined dyslipidemia in persons without HIV infection. We chose this end point because it reflects the general therapeutic approach of clinicians faced with
a patient with combined dyslipidemia. The following components were required to meet the goal: LDL levels 100 mg/dl (for subjects with two or more cardiovascular risk factors) or <130 mg/dl (for subjects with fewer than two risk factors); TG levels <200 mg/dl (for subjects with entry TG 200-800 mg/dl) or TG <400 mg/dl (for subjects with entry TG 800 mg/dl); and HDL levels ≥35 mg/dl. We modified NCEP TG goals for persons with very high TG because we thought it would be unrealistic to expect a complete response based upon the known
potency of lipid-lowering agents used in this trial. The overall objective was to achieve the NCEP goal for LDL, TG, and HDL, hereafter termed the composite goal.
Study design
A randomized, open-label, 48-week clinical trial was conducted on HIV-infected persons with combined dyslipidemia (LDL ≥130 mg/dl and TG ≥200 mg/dl) on potent antiretroviral regimens for 6 months. Subjects were recruited from 45
Adult AIDS Clinical Trials Group (AACTG) sites. Enrollement was stratified by gender, TG level (TG 200-800 mg/dl versus TG >800 mg/dl), and cardiovascular risk factors (at least two cardiovascular risk factors versus fewer than two). Stratification for TG levels was based upon our assumption that individuals with very high TG levels were unlikely to achieve the NCEP goal of having a TG <200 mg/dl. Cardiovascular risk factors included age (men ≥45 years, women ≥55 years or premature menopause without estrogen replacement therapy); family
history of CHD (first-degree male relative with CHD before 55 years of age or first-degree female relative with CHD before 65 years of age); current cigarette smoking (defined as anyone who smokes or smoked at least five cigarettes per week at any time during the last 3 months); hypertension defined as untreated
systolic blood pressure (SBP) >140 mm Hg or diastolic blood pressure (DBP) >90 mm Hg from an average of two or more readings on two or more occasions or requiring antihypertensive treatment (which includes medication, diet, and/or
exercise); and low HDL cholesterol at the time of screening (<35 mg/dl). In the presence of high HDL cholesterol (≥60 mg/dl), one risk factor is subtracted.
Subjects were randomized to receive either micronized fenofibrate 200 mg po daily or pravastatin 40 mg po daily (Fig. 1). Subjects who had failed to reach the composite goal by week 12 were required to receive both pravastatin and fenofibrate beginning at week 16 and were followed for a total of 48 weeks.
Subjects who were not willing to start dual-agent therapy were required to discontinue study medications at week 16, were considered off-drug/on-study, and were followed for the full duration of 48 weeks. Subjects were requested to continue antiretroviral therapy without change throughout the duration of the
study. The study was designed as a noninferiority trial with a noninferiority margin of 10%, to determine if fenofibrate was noninferior to pravastatin with respect to achieving the composite goal. Enrollment was suspended prior to targeted accrual (630) after the initial review by a Data and Safety Monitoring
Board concluded that since an insufficient proportion of subjects in both single-agent arms met the composite goal at week 12, all subjects should be offered dual-agent therapy.
Criteria for inclusion and exclusion
Eligible subjects had documented HIV-1 infection. Subjects must have been on a lipid-lowering diet and have exercised for at least 30 days prior to screening; have fasting TG 200 mg/dl and fasting LDL 130 mg/dl (direct by ultracentrifugation). Of the 660 subjects screened for entry 90% of screen failures were due to failure to meet the lipid level entry criteria of the study. This may have occurred, in part, because study sites were unfamiliar with dyslipidemia and screened many persons with very high TG levels that had normal LDL values on direct measurement. In addition, many subjects did not have previously documented adequate fasting lipid levels prior to study screening. Subjects were required to be on stable treatment with potent antiretroviral therapy for >6 months that provided adequate viral suppression as determined by the subject's primary care physician. Structured treatment interruptions were not allowed.
All subjects were ≥18 years of age, with Karnofsky performance status ≥70 and able and willing to give written informed consent. Women of reproductive potential were required to have a negative pregnancy test. All subjects had to use appropriate contraceptive measures. Subjects were excluded for any of the
following laboratory criteria: platelet count 65,000/mm3; hemoglobin 9.1 gm/dl for men and 8.9 gm/dl for women; AST and ALT >3 x upper limit of normal; creatine kinase (CK) >4.0 x upper limit of normal; serum creatinine >1.5 x upper limit of normal or calculated creatinine clearance <50 ml/min; absolute neutrophil count 850/mm3; and fasting glucose >126 mg/dl; lipase . 1.5 x upper limit of normal. Receipt of any experimental therapy (not FDA approved) within 14 days prior to study entry or receipt of any prescription or nonprescription lipid-lowering agents within 14 days prior to screening,
including niacin in doses ≥1 g/day, were not allowed. Subjects must not have been on any lipid-lowering agent for 24 weeks in the past. Subjects who had failed previous statin or fibrate therapy (after 24 weeks of treatment) were also excluded.
Other exclusion criteria were receipt of drugs that may increase the risk of myopathy or rhabdomyolysis within 14 days prior to entry; receipt of creatine monophosphate or immunomodulating therapy within 30 days prior to entry; current receipt of systemic chemotherapy for B cell lymphoma or malignancies
other than Kaposi's sarcoma; or allergy/sensitivity to study drug(s) or any medication related to statins and/or fibrates. Subjects with active drug or alcohol abuse or dependence that, in the opinion of the investigator, would interfere
with adherence to study requirements were excluded. Subjects were also excluded if they had any known history of CHD including a prior history of angina, myocardial infarction, angioplasty, or bypass surgery; or uncontrolled hypertension within 4 weeks of entry defined as SBP >180 mm Hg or DBP >115
mm Hg; diabetes mellitus requiring medical therapy; current receipt of antidiabetic medications; history of rhabdomyolysis; history of severe hepatic dysfunction; active or symptomatic gallbladder disease within 3 months of entry; hypothyroidism; pregnancy and breast-feeding; or testosterone doses exceeding
those for physiologic replacement therapy. Hormone replacement therapy for postmenopausal women, for birth control, and for transgendered subjects was allowed.
End points
The primary end points were the achievement of the composite goal based on absolute values for LDL, TG, and HDL at week 12, and safety/tolerability of lipid-lowering therapy at week 12. Secondary end points were these values at weeks 28 and 48. Lipids were measured by lipoprotein fractionation by ultracentrifugation at the AACTG Central Metabolic Laboratory (Quest Diagnostics, Baltimore, MD).
Approval, support, and conduct of the study
The institutional review boards of the participating institutions approved the study and each study subject gave written informed consent. Fenofibrate was donated by Abbott Laboratories and pravastatin was donated by Bristol-Myers Squibb.
Subject visits and laboratory assays were supported by the AACTG funded by the National Institute of Allergy and Infectious Diseases (NIAID). The study was approved by the scientific committees of the AACTG and the NIAID Division of
AIDS Clinical Science Review Committee.
Statistical analysis
The statistical analyses consisted of descriptive statistics and confidence intervals. Exact tests were used to explore associations between categorical variables. Kruskal-Wallis tests were used to compare distributions of continuous variables between groups. Wilcoxon signed rank tests were utilized to explore the significance of within-group changes.
All significance testing was performed at the 0.05 level with no adjustment for multiple testing. All reported p-values are two-sided. All analyses were conducted in an intention-to-treat manner.
Data from subjects who discontinued therapy but remained on study were used in all analyses; in the case of missing data at an analysis time point, results from the preceding visit were carried forward, if available. Some subjects were lost to follow-up and lacked appropriate data to carry forward for some analyses at later time points; however, these subjects were included in analyses at earlier time points if their data were available.
AFFILIATIONS of Study Investigators
1New York University, New York, New York 10016.
2Harvard School of Public Health, Boston, Massachusetts 02115.
3Social and Scientific Systems, Inc., Silver Spring, Maryland 20910.
4National Institute of Allergy and Infectious Diseases, Bethesda, Maryland 20892.
5University of Minnesota, Minneapolis, Minnesota 55455.
6Weill Medical College of Cornell University, New York, New York 10021.
7University of California, San Diego, La Jolla, California 92093.
8Frontier Science and Technology Research Foundation, Chestnut Hill, Massachusetts 02467.
9University of Cincinnati, Cincinnati, Ohio 45221.
Present address: National Institute of Allergy and Infectious Diseases, Bethesda, Maryland 20892.
Present address: Wyeth Laboratories, Madison, New Jersey.
Study group members are listed in the Acknowledgments.
|
|
| |
| |
|
|
|