| |
Predictors of Body Changes
|
| |
| |
"CD4+ Cell Count, Viral Load, and Highly Active Antiretroviral Therapy Use Are Independent Predictors of Body Composition Alterations in HIV-Infected Adults: A Longitudinal Study"
Clinical Infectious Diseases Dec 1, 2005;41:000
Ann Yelmokas McDermott,1 Norma Terrin,2 Christine Wanke,2,3 Sally Skinner,3 Eric Tchetgen,4 and Abby H. Shevitz,2,3,a
1Jean Mayer US Department of Agriculture Human Nutrition Research Center on Aging at Tufts University, 2TuftsNew England Medical Center, 3Department of Public Health and Family Medicine/Nutrition Infectious Disease Unit, Tufts University School of Medicine, and 4Harvard School of Public Health, Boston, Massachusetts
....disease status was associated with regional body changes, because a lower baseline CD4+ cell count (P <.006) or a higher baseline viral load (P <.03) predicted a greater loss of trunk and limb fat....
....women were more likely to report insufficient calcium intake (69.1% did not meet the dietary reference index value, compared with 34.6% of men; P < .001) and insufficient energy intake (i.e., <35 kcal/kg of body weight) (69.1% of women vs. 30.9% of men; P < .001)...
....The current longitudinal study confirms that ongoing improvement in CD4+ cell count was concurrent with gains in extremity LM, perhaps because the latter reflects improved health, nutrition, and mobility that may occur with an improvement in CD4+ cell count.... (?? I'm not sure I understand ths, they appear to say that increased CD4 count on HAART was associated with increase in limb fat. If so, I don't know how or why they had this finding, perhaps it relates to the ART drugs patients were taking overall in this study)
These findings.....support the findings from our group and others that disease status is involved...
ABSTRACT
Background. To understand the concurrent effects of human immunodeficiency virus (HIV) infection, the immune system, and antiretroviral therapy on body composition alterations, we examined annualized composition changes in HIV-infected adults who were receiving stable antiretroviral therapy.
Methods. With use of data from the Nutrition For Healthy Living Study, we performed multivariate analyses using longitudinal models to evaluate the relationship of CD4+ cell count, viral load, and highly active antiretroviral therapy (HAART) or antiretroviral therapy (ART) with changes in trunk and extremity composition for 110 men and 42 women who provided data relating to 194 study intervals (i.e., intervals of time between 2 assessment visits). Of these intervals, 165 involved HAART use (89.7% involved protease inhibitorbased regimens), and 29 did not involve HAART use. Patients receiving HAART or ART (who had continuous use during the interval) were compared with HAART- or ART-naive subjects.
Results.
The median length of intervals between visits was 12.9 months (interquartile range, 12.117.6 months).
In models adjusted for HAART or ART use, baseline CD4+ cell count was positively associated with increased trunk fat (mean increase per year, 2.3% per 100 cells/mm3; 95% confidence interval [CI], 0.7%3.9%]) and, in men, with increased extremity fat (mean increase per year, 1.8% per 100 cells/mm3; 95% CI, 0.6%3.0%).
Increase in CD4+ cell count predicted increased extremity lean mass (mean increase per year, 0.6% per 100 cells/mm3; 95% CI, 0.05%1.1%).
Higher baseline viral load predicted fat loss (trunk fat loss per year, -5.0% per log10 copies/mL; 95% CI, -9.4% to -0.7%; extremity fat loss per year, -3.4% per log10 copies/mL; 95% CI, -6.1% to -0.6%), as did zidovudine use (trunk fat loss per year, -10.8%; 95% CI, -20.4% to -1.4%; extremity fat loss per year, -4.9%; 95% CI, -9.8% to -0.01%).
HAART use independently predicted decreased bone mineral content (extremity bone mineral content loss per year, -1.6%; 95% CI, -3.1% to -0.08%) but did not predict changes in fat or lean mass.
Receipt of protease inhibitor-based HAART predicted a -1.9% decrease in extremity bone mineral content per year (95% CI, -3.6% to -0.2%), and zidovudine use predicted a -2.6% decrease in trunk bone mineral content per year (95% CI, -4.4% to -0.8%).
Conclusions. Baseline viral load, CD4+ cell count, and change in CD4+ cell count predicted alterations in trunk fat, extremity fat, and lean mass. HAART use and zidovudine use were associated with bone loss, and zidovudine use was associated with fat loss, but HAART use was not associated with fat mass changes.
DISCUSSION
With the advent of new and effective antiretroviral therapies, immune restoration and viral suppression are now possible in HIV-infected adults in the United States. As a result, it has been difficult to determine which medication and host factors are associated with the varying aspects of body composition alterations. Many previously reported analyses did not use continuous or objective measures of change, have been cross-sectional, have not included HAART-naive persons for comparison, did not look directly at the effects of CD4+ cell counts or viral loads after adjustment, or did not consider such behaviors as dietary intake and smoking [6, 7, 14, 22]. Although case reports and initiation studies have implicated the introduction of HAART in rapidly inducing body-shape changes in some people, it remains unclear whether these changes continue to evolve with continuous HAART or antiretroviral therapy use. We offer exploratory findings from a longitudinal examination of regional body composition changes in a large cohort of HAART or antiretroviral therapyexperienced and naive men and women, and we demonstrate ongoing changes in individuals receiving stable therapies.
This analysis confirms an average annual loss of extremity fat in the cohort. DEXA cannot distinguish between upper and lower trunk fat, nor can it distinguish between the visceral and subcutaneous compartments of the abdomen, but total trunk fat changes were not found. Most importantly, disease status was associated with regional body changes, because a lower baseline CD4+ cell count (P <.006) or a higher baseline viral load (P <.03) predicted a greater loss of trunk and limb fat.
HAART use or its duration did not contribute to change. Of interest, zidovudine but not stavudine was associated with loss of limb and trunk fat. Because of the time frame in which data were collected, we were unable to determine the effect of some individual medications, including abacavir and the NNRTIs. These findings agree with the current literature that suggests lipoatrophy is the unique physical manifestation of HIV infection, and they support the findings from our group and others that disease status is involved [4, 6, 19, 2325]. However, it remains impossible to separate the individual effects of antiretroviral therapy agents, because they are used in combination.
Changes in LM have been less well investigated in the HAART era. An early report from our group found that, after HAART initiation, weight increased, and LM did not [8], but our later reports found greater LM in individuals receiving HAART than in others [5]. The current longitudinal study confirms that ongoing improvement in CD4+ cell count was concurrent with gains in extremity LM, perhaps because the latter reflects improved health, nutrition, and mobility that may occur with an improvement in CD4+ cell count.
It appears that both medication and modifiable lifestyle factors altered bone health in our cohort. HAART use and PI use remained associated with bone mineral loss, even after adjustment for the strong effects of numerous well-known osteopenia cofactors. Other significant risk factors included cigarette smoking and dietary practices. The literature has been conflicted about the prevalence of predictors of BMC changes [12, 2629]. Our analysis supports the involvement of antiretroviral therapy, as well as known risk factors, reinforcing the need for additional longitudinal studies that account for diet, cigarette smoking, and medications concurrently.
Although a cohort such as this does not allow us to examine the changes induced by the introduction of HAART (as would be possible in a randomized control trial), our data do assist in understanding the real-world situation for many patients who experience ongoing body changes over time while receiving treatment. Though our numbers were small for some analyses, statistical differences were apparent. These findings suggest the importance of peak viral load and nadir CD4+ cell count on the subsequent loss of fat, a concurrence in the improvements in CD4+ cell count and LM, and the primary influences of medications and traditional risk factors in bone demineralization. Many details about HIV-associated body composition alterations remain unanswered, but this work confirms the growing body of evidence that such changes are multifactorial.
Table 2. Baseline values and changes in regional body composition as assessed by dual-energy x-ray absorptiometry after the first study interval for 152 HIV-infected adults enrolled in a longitudinal study of body composition alterations.
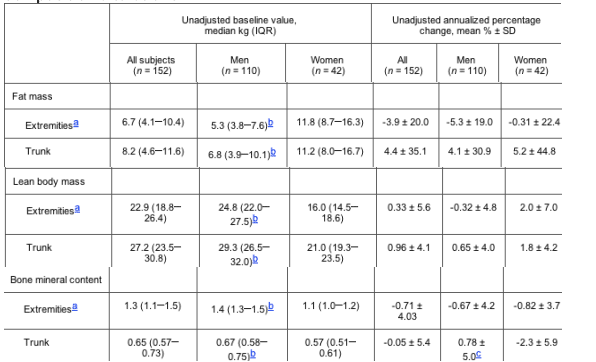
a Determined by the sum of arms plus legs to as defined by dual-energy x-ray absorptiometry scan.
b Statistically significant difference between men and women (P < .001).
c Statistically significant difference between men and women (P = .003).
RESULTS
Subjects.
One hundred ten men and 42 women (a total of 152 subject) with a total of 194 intervals met inclusion criteria for this analysis. Seventy-six percent of individuals provided 1 interval for analysis, 20% provided 2 intervals, and 4% provided 3 intervals. Table 1 presents the baseline assessments for the participants. In subjects using HAART at baseline, the median time of continuous HAART use prior to the initial assessment visit was 15.3 months (interquartile range, 7.026.7 months). Median interval length was 12.9 months (interquartile range, 12.117.6 months). Of the 165 HAART-use intervals, 89.7% of these involved regimens that were PI-based HAART; 29 were non-HAART intervals.
Table 1 shows differences between men and women in the cohort. On the basis of 3-day food record from their initial visit, women were more likely to report insufficient calcium intake (69.1% did not meet the dietary reference index value, compared with 34.6% of men; P < .001) and insufficient energy intake (i.e., <35 kcal/kg of body weight) (69.1% of women vs. 30.9% of men; P < .001) (data not shown). There were no statistically significant differences by sex, except that women were less likely to use HAART (76.2% vs. 90.9%; P = .02), more likely to use non-HAART antiretroviral therapies (23.8% vs. 8.2%; P = .009) (data not shown), and had been receiving HAART for a shorter duration (median duration, 6.8 vs. 15.2 months; P = .02) than were men. Baseline regional body composition differed by sex (table 2); women had nearly double the FM, two-thirds the LM, and less BMC than men (P < .001 for all). No differences in annualized percentage change in regional body composition were observed between men and women, except for trunk BMC (P = .003) (table 2).
Predictors of Regional Body Composition Changes
Examining all 194 intervals (unadjusted interval data not shown), the cohort experienced an average annual loss of 3.6% of extremity FM (P = .011), with greater changes seen in men. Increases in trunk FM were not significant (P = .66). Trunk LM (nonfat, nonbone mass) increased 0.9% per year (P = .002), whereas extremity LM did not change. Small decreases in extremity BMC were not statistically significant. Women had a statistically significant annual loss of trunk BMC (mean unadjusted change [±SD] in women, -1.5% ± 5.6%; mean unadjusted change [±SD] in men, 0.94% ± 5.0%; P = .004 for sex interaction).
Baseline CD4+ cell count.
Figures 2-4 illustrate adjusted models examining CD4+ cell count and viral load effects on regional changes. A positive linear relationship existed between change in trunk fat and baseline CD4+ cell count, with a 2.3% annualized mean increase in trunk fat per 100 CD4+ cells/mm3 (95% CI, 0.7%3.9%; P = .006). The relationship between extremity FM and baseline CD4+ cell count varied by sex (P = .02 for sex interaction). Baseline CD4+ cell count predicted a 1.8% annualized mean increase in extremity FM per 100 CD4+ cells/mm3 in men only (95% CI, 0.6%3.0%; P = .005); for women, the association was not statistically significant (P = .18). Baseline CD4+ cell count was not associated with changes in LM or BMC.
Change in CD4+ cell count.
Extremity LM increased with changes in CD4+ cell count from baseline (mean increase per year, 0.59% per 100 cells/mm3; 95% CI, 0.05%1.1%; P = .03).
Baseline viral load.
Higher viral load at baseline predicted a subsequent loss of extremity FM (mean decrease per year, -3.4% per log10 copies/mL; 95% CI, -6.1% to -0.6%; P = .02) and trunk FM (mean decrease per year, -5.0% per log10 copies/mL; 95% CI, -9.4% to -0.7%; P = .03).
Change in viral load.
No association was found between change in viral load and regional body composition changes.
Use of HAART or antiretroviral therapy.
Table 3 shows the association between use of specific antiretroviral therapy and HAART regimens and body composition changes in adjusted models. HAART use (found in 146 intervals) was associated with a mean annual 1.6% loss of extremity BMC in both sexes (95% CI, -3.1% to -0.08%; P = .04) and a mean 2.5% annualized LM increase in women (95% CI, 0.5%4.6%; P = .02) but with no other body composition changes.
Increased extremity LM (mean increase per year, 2.8%; 95% CI, 0.1%5.6%; P = .04) was associated with NNRTI-based HAART regimens, but the number of intervals analyzed was very small (16 of 169 intervals analyzed involved NNRTI-based HAART regimens). PI-based HAART (used in 92 of 169 intervals) was associated with extremity BMC loss (mean decrease per year, -1.9%; 95% CI, -3.6% to -0.2%; P = .03), but no other HAART regimen had this association.
Zidovudine use (found in 60 of 103 intervals analyzed) was associated with an annual decrease in extremity FM (mean decrease per year, -4.9%; 95% CI, -9.8% to -0.01%; P = .049) and trunk FM (mean decrease per year, -10.8%; 95% CI, -20.4% to -1.4%; P = .02). Zalcitabine use was associated with a decrease in trunk FM, and stavudine use was not. Use of lamivudine (found in 117 of 127 intervals) was associated with increased trunk LM (mean increase per year, 3.0%; 95% CI, 0.1%5.8%; P = .04). Although few individuals were receiving didanosine (found in 27 of 137 intervals), use of the agent was associated with a decrease in trunk LM (mean decrease per year, -6.4%; 95% CI, -10.3% to -2.5%; P = .002) in women only. Zidovudine use (found in 60 of 103 intervals) was associated with an annual decrease in trunk BMC (mean decrease per year, -2.6%; 95% CI, -4.4% to -0.8%; P = .005). The use of stavudine (found in 69 of 130 intervals) was associated with an increase in trunk BMC (mean increase per year, 2.0%; 95% CI, 0.4%3.6%; P = .01).
Adjusted models found no effect on regional FM attributable to use of any individual PI (table 3), but saquinavir use (found in 28 of 91 intervals) and ritonavir use (found in 24 of 92 intervals) were each associated an annualized decrease in extremity LM (mean decrease per year, -1.4% [95% CI, -2.7% to -0.7%] and -1.8% [95% CI, -3.2% to -0.4%], respectively; P = .04 and P = .01, respectively). Nelfinavir use (found in 43 of 98 intervals) was associated with a 2.2% annualized increase in extremity BMC (95% CI, 1.2%3.2%; P < .001).
Low energy intake predicted a large loss of trunk fat (coefficient range, -7.8% to -17.1% per year; P < .05 for all) in models examining the effect of viral load change, HAART use, and the PIs ritonavir, indinavir, nelfinavir, and saquinavir and was retained in these models. Cigarette smoking was associated with annual decreases in BMC in the trunk (coefficient range, -1.4% to -2.3%) and extremities (coefficient range, -1.1% to -3.2%). In the HAART model, each year of age predicted a more rapid decrease in trunk LM (mean decrease per year, -0.05%; 95% CI, -0.09% to -0.006%; P = .03).
|
|
| |
| |
|
|
|