 |
 |
 |
| |
Pioglitazone Improves Fatty Liver
|
| |
| |
Following this introduction are the study results presented in an oral session yesterday showing improvements in fatty liver & histopathology in individuals with NASH (fatty liver) from a pilot study.
Fatty Liver, HIV
Hello from DDW (Digestive Disease Week, Chicago, May 14-19). At this annual meeting important information & studies related to liver disease are presented in between presentations and abstracts focused more on digestive diseases including GI cancers, diarrhea, small bowel motility disorders and colonoscopies. Of note from today symposiums were talks on hepatitis B and a talk on non-alcoholic fatty liver disease (also called fatty liver, NAFLD, NASH). Before reporting on HBV, some important information about fatty liver and HIV. In both HIV positive & negative individuals these serious complications can occur. Fatty liver is the accumulation of fat in the liver. Fatty liver can lead to cirrhosis & liver disease. The deposition of fat or triglycerides in the liver can be causative. Mitochondrial abnormalities can lead to fatty liver. Insulin resistance is associated with fatty liver. Accumulation of fat in the belly, central fat distribution, is also causative of fatty liver. Metabolic syndrome is associated with insulin resaistance, diabetes, and fatty liver. Visceral (in the belly) adipose tissue (fat tissue) is particularly resistant to insulin. Thus, metabolic abnormalities in HIV are not only associated with a potential risk for heart disease, but appear to increase the risk for liver disease and advanced fibrosis. Although there are no large scale randomized clinical trials for the treatmentof fatty liver (nonalcoholic fatty liver disease, or NASH- non-alcoholic steatohepatitis) there are available approaches based on the pathophysiologic principles & data supporting these options: weight loss; even a modest 5-10% decrease in body weight can result in marked improvement in insulin sensitivity; overweight &visceral (belly) fat accumuluation are associated with insulin resistance & fatty liver. Diet: diet high in refined sugars, saturated fats & low in fiber promote insulin resistance. Exercise has been shown to increase the oxidative capacity of muscle cells & utilization of fatty acids for oxidation. This decreases fatty acid & triglyceride accumulation in the myocytes & thereby improve insulin sensitivity. The degree of improvement in insulin sensitivity is related to the intensity of exercise. In animal model with steatohepatitis, metformin treatment improved hepatic steatosis and inflammation. In a recent study, Metformin was associated with an improvement in serum aminotransferase activities. However, no histologic data were provided. The use of the drug remains experimental. Several studies of pioglitazone demonstrate improved insulin resistance. UDCA (ursodeoxycholate) is a hyderophilic bile acid which is associated with hepatoprotective properties. In one study UDCA produced improved liver enzymes & a decrease in hepatic steatosis, but long term benefits & optimal dose remain to be established. A study is underway. There is one published study on Vitamin E in a series of 11 pediatric patients with NASH who received 400 IU/day vitamin E (diatocopherol) orally. 400-1200IU of vitamin E were given & resulted in improved ALT in all cases. No followup liver histology was available. At EASL a study was presented demonstrating benefit from 400 IU vitamin E.
"A Double-Blind, Placebo-Controlled Trial of Pioglitazone in the Treatment of Non-Alcoholic Steatohepatitis (NASH)"
Stephen Harrison (Brooke Army Medical Center) presented these study data at the Digestive Disease Week conference in Chicago May 15, 2005.
Note from Jules Levin: Fatty liver may result from elevations of lipids in HIV+ individuals although this has not been studied. As well, fatty liver may reduce response rates to HCV therapy with peginterferon+RBV, but this also has not yet been studied in HIV. Fatty liver can also have the affect of advancing liver disease by causing fibrosis. HIV+ individuals who don't have hepatitis can develop fatty liver & of course individuals with viral hepatitis can develop fatty liver, as well as individuals who don't have HIV or viral hepatitis.
BACKGROUND
The prevalence of obesity & co-existent insulin resistance is rising dramatically. NASH (fatty liver) has been shown to be associated with insulin resistance. A number of small studies have shown that PPAR-gamma ligands (e.g. pioglitazone) may improve insulinb resistance & NASH, but these studies are not placebo controlled. This study is the first trial assessing the benefit of diet+pioglitazone compared with diet+placebo in the treatment of NASH.
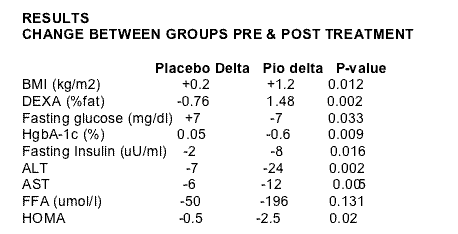
Patients were excluded if evidence of other liver disease or currently on insulin sensitizing medication or insulin injections. Patients received pioglitazone 30mg/day for 8 weeks then 45mg/day for a total treatment period of 24 weeks. 70 patients were randomized & screened. Baseline characteristics were comparable between the placebo & pioglitazone groups, except for gender. Preliminary results are presented for 40 patients:
Placebo-Pioglitazone
Age: 50-51
Gender: m/f, 11/11 PLBO; 6/12 PIO
BMI (kg/m2): 33.2-33.3
DEXA (%fat): 35.5-34
Fasting glucose (mg/dl): 113-110
HbA-1c (%): 6.1-5.9
Fasting insulin (uU/ml): 20-18
ALT: 53-52
AST: 41-37
FFA (umol/l): 749-758
HOMA: 5.1-4.9
AUTHOR'S SUMMARY
Placebo & diet alone improved some histopathologic parameters attesting to the importance of a placebo control in studies of therapeutic modalities for NASH.
Use of pioglitazone markedly improved:
--glucose metabolism & insulin sensitivity, but not for placebo study
participants
--lipid metabolism (decreased FFA, increased adiponectin)
--hepatic fat (NMR spectroscopy), but not for placebo
--liver enzymes
Effect of pioglitazone on histopathology:
--marked decrease in liver fat
--supportive beneficial changes in ballooning necrosis
--composite index of inflammation, ballooning necrosis, steatosis & fibrosis was significantly improved between groups
--inflammation significantly improved in placebo & pio groups, but not between groups
--fibrosis improved significantly in pioglitazone group (p=0.04) but not in the placebo group
The authors concluded that this relatively small group of patients with NASH treated for 6 months with pioglitazone+diet was associated with favorable metabolic & histologic outcome when compared with diet alone. These encouraging results need to be corroborated in larger trials with longer treatment intervals.
EFFECT OF PIOGLITAZONE ON ADIPONECTIN LEVELS
After 12 weeks in study there was no change in placebo group but levels increased significantly in the pioglitazone group (p=0.001) from about 5 to 10 ug/ml.
|
|
| |
| |
|
 |
 |
|
|