 |
 |
 |
| |
VX-950: HCV PROTEASE INHIBITOR
INITIAL RESULTS OF A PHASE 1b, MULTIPLE DOSE STUDY OF VX-950, A HEPATITIS C VIRUS PROTEASE INIHIBITOR
|
| |
| |
Reported by Jules Levin
Perhaps the most exciting presentation at this DDW conference (Chicago, May 13-19, 2005) was today's presentation at the morning HCV Oral Session of the first study results of this HCV protease Inhibitor in HCV-positive genotype 1 patients presented by HW Reesnik from the academic Medical Center, Amsterdam, the Netherlands and by Vertex Pharmaceuticals who is the drug sponsor & developer.
BACKGROUND
VX-950 is an orally available HCV protease inhibitor.
--reversible & tight binding to HCV protease active site with slow off-rate
--Ki=7nM (formation of stable enzyme-inhibitor complex)
--active in replicon assays, replication assays & in animal models
--single-dose healthy subjects study completed in 2004
VX04-950-101 CLINICAL STUDY DESIGN
Dose escalation study, Interim Safety Review prior to each cohort
--primary objective: assessment of safety
--secondary objective: assessment of pharmacokinetics & viral kinetics
--study drug dosed as oral suspension (in asking company about formulation, they said a tablet formulation is in development)
--PART A: healthy subjects
--450mg, 750mg, & 1250mg, or placebo 3x/day for 5 days
--6 VX-950, 2 placebo in each cohort
PART B: 36 patients with genotype 1 HCV
--450mg or 750mg 3x/day, 1250mg wx/day, or placebo for 14 days
--10 VX-950, 2 placebo in each cohort
VX04-950-101 Interim Analysis
Part A: healthy subject dosing & follow-up, complete unblended preliminary analysis available.
PART B (patients with HCV) to completion of dosing (Day 14) only
--results unblended only by treatment group (not by patient)
--HCV RNA data available first, complete preliminary analysis available
--safety (adverse events) & lab data not yet unlocked & unblended
no summary statistics or counts of adverse events are available
Authors reported VX-950 Was Well Tolerated by Healthy Subjects
--no Seroius advserse events
--no changes in vital signs, physical exams, or ECGs
--most common adverse events in VX-950 groups considered possibly related to study drug (all mild in severity):
Number of Subjects
Headache: 5 (28%)
Diarrhea:3 (17%)
Nausea: 2 (11%)
frequent urination: 2 (11%)
sleepiness/drowsiness: 2 (11%)
PHARMACOKINETCS
Trough levels are positively correlated with increasing dose. Investigator said trough levels for 750mg dose were 45% greater than other doses. Steady state is reached by Day 3 of dosing.
BASELINE STATUS FOR PATIENTS WITH HCV
Placebo(n=6), 450mg q8h(n=10), 750mg q8h(n=8), 1250mg q12h(n=10)
Male/female: 3/3; 8/2; 3/5; 8/2
Median age: 53, 47, 52, 44
Median wt (kg): 77, 78, 75, 70
Treatment-naïve: 2, 1, 3, 3
Median HCV-RNA (log)*: 6.3, 6.45, 6.1, 6.48
*IU/mL. Cobas Taqman HCV RNA assay, Roche
PRELIMINARY SAFETY IN PATIENTS WITH HCV (VX04-950-101)
--no SAEs
--no discontinuations
--no elevations of ALT/AST or other clinical chemistry findings reported
--no events in Interim Safety Reviews which delayed or prevented dose escalation
--analysis of adverse event incidence pending
VIRAL LOAD REDUCTIONS
Viral load reductions were shown in graphs. All doses showed steep viral load reductions during the first phase kinetics, the first 3-4 days. The 750mg dose viral load reduction was a steady slow incremental decline following the first phase decline. The other two doses showed plateauing of viral load decline after the first phase & after about 7 days the viral levels started to rebound back towards baseline. The speaker said they are conducting resistance analysis.
UNDETECTABLE HCV RNA ACHIEVED at DAY 14 with VX-950
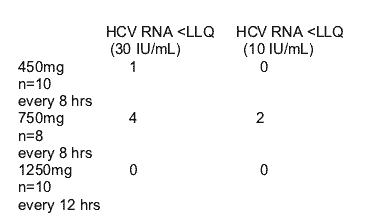
MEDIAN CHANGE from Baseline HCV RNA
-4 log with 750 mg every 8 hr dose
They showed a graph of the individuals patients & their viral load response: the range of viral load reductions were from declines of just over 3 logs to slightly more than 6 logs from baseline. One patient showed similar viral load reductions after days 3-4 but viral load rebounded back to baseline. This patient had the lowest VX-950 exposure in the dose group.
The authors reported that after VX-950 dosing stopped the return towards baseline was slow in patients who achieved HCV RNA <30 IU/mL; it took 28 days to return to baseline levels.
PENDING DATA ANALYSIS
--full unblinded clinical safety & lab data evaluation, pst-dosing data (Part B)
--viral sequence analysis
--viral kinetic analysis
--neopterin as marker of endogenous interferon response
AUTHOR'S CONCLUSIONS
--Multiple dosing of VX-950 for 5-14 days was well tolerated in healthy subjects & patients with HCV
--4 log median reductions in HCV RNA & undetectable HCV RNA achieved
with 14 days of VX-950
--rapdity of viral load decline, undetectable HCV RNA & slow return of HCV RNA post-dosing suggest that VX950 should be explored as a monotherapy
--combination therapy wih other antiviral agents or IFN may be required to obtain optimal responses
--treatment duration with HCV PI therapy may be shorter than the current standard of care
|
|
| |
| |
|
 |
 |
|
|