 |
 |
 |
| |
HBV DNA Elevations & Risk for Liver Cancer
|
| |
| |
"Persistent Elevation of Serum HBV DNA Level is a Risk Factor for Hepatocellular Carcinoma: The R.E.V.E.A.L.-HBV Study"
.....the study reports that patients with persistent HBV DNA (viral load) >100,000 copies/ml have a 9 fold greater risk for developing liver cancer. The average time between first & followup HBV DNA test was 10 years for these patients.....
Reported by Jules Levin
Chien-Jen Chen1, Hwai-I Yang1, Jun Su2, Chin-Lan Jen1, Edward Kuo3, San-Lin You1, Uchenna H Iloeje2
1National Taiwan University of Taiwan, Taiwan; 2Bristol-Myers Squibb Co., Wallingford, CT, USA; 3Private Medical Practice, previously Bristol-Myers Squibb Co., Taipei, Taiwan
Digestive Disease Week
Chicago, IL, May 14-19, 2005
Poster Number M917
The study authors reported these data & information.
AUTHOR'S CONCLUSIONS:
-The incidence of HCC increases with increasing HBV DNA across a
biological gradient
-Elevated serum HBV DNA level is a strong risk predictor for HCC, independent of HBeAg status, chronic HCV infection, and elevated serum ALT level
-Persistent elevation of serum HBV DNA level was associated with the greatest risk of HCC
Subgroup Analyses: Persistent HBV DNA Elevation and HCC Risk
-1,678 men and women had a serum HBV DNA level of ≥104 copies/mL (10,000 copies/ml) at cohort entry
-The serum samples collected at the last follow-up health examination or at the
follow-up examination preceding the HCC diagnosis (for cases) were tested for serum HBV DNA level
-1,376 of 1,678 (82%) had serum samples collected at follow-up health examination available for HBV DNA testing
- Samples from subjects with entry HBV DNA level <104 copies/mL were
not re-tested; this was the reference group
PERSISTENTLY ELEVATED HBV DNA (viral load) OVER 10 YRS INCREASES RISK FOR LIVER CANCER
The table immediately below summarizes multivariate-adlusted relative risk of HCC (liver cancer) by HBV DNA (viral load) level in blood samples collected at study beginning & last followup visit by patient.
Patients with 10,000 to 100,000 copies/ml at baseline & <10,000 at followup exam had 6 HCC cases& have adjusted relative risk of 1.4 (range: 0.6-3.4) compared (time between tests: 10.7 yrs) to the 1.0 risk established as a reference for patients with <10,000 at baseline & not tested after that first visit.
Patients with 10,000 to 100,000 at baseline & also at followup visit: 2 cases of HCC, adj-RR: 0.9 (9 yrs between tests) (range: 0.2-3.7)
Patients with 10,000 to 100,000 at baseline & >=100,000 at followup visit: 5 HCC cases; adj-RR: 3.1 (range: 1.2-7.9) (9.8 yrs between tests)
Patients with >=100,000 at baseline & <10,000 at followup visit: 9 cases of HCC; adj-RR: 3.6 (range: 1.7-7.6) (11 yrs between tests)
Patients with >=100,000 at baseline & 10,000 to 100,000 at followup visit: 11 cases of HCC; adj-RR: 6.9 (range: 3.4-13.8) (10.5 yrs between tests)
Patients with >=100,000 at baseline & >=100,000 at followup visit: 56 HCC cases; adj-RR: 9.1 (range: 5.8-14.1) (9.8 yrs between HBV DNA tests)
Table 4: Persistent HBV DNA Elevation and HCC Risk
Multivariate-adjusted relative risk of HCC by HBV DNA level in serum samples collected at recruitment and last follow-up examinations
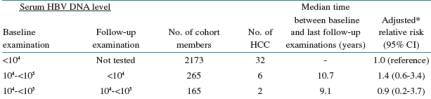


*Adjusted for gender, age, anti-HCV antibody status, cigarette smoking, and alcohol consumption. p=0.01 for the trend among those who had baseline serum DNA ≥105 copies/mL.
†p<0.01. ‡p<0.001.
Table 3 reports the incidence of liver cancer developed by patients just based on HBV DNA levels after one test, the baseline test; not based on persistent elevations.
Patients with <300 copies/ml )n=951) were used as reference with 1.0 adj-RR (risk); this group had 16 cases of HCC, liver cancer. Incidence rate per 100,000: 145.2
Patients with 300 to 1,000 copies/ml: n=1,222; HCC cases: 16; incidence rate 112.5 per 100,000; adj-RR: 0.9 (range: 0.5-1.9)
Patients with 1,000 to 10,000 copies/ml: n=664; 24 cases of HCC; incidence rate: 314 per 100,000; adj-RR: 2.4 (range: 1.3-4.5)
10,000 to 100,000 copies/ml: n=362; 38 HCC cases; incidence rate: 952 per 100,000; adj-RR: 7.2 (range: 4-12.9)
>=1 million: n=652; 82 HCC cases; incidence ratio: 1,149 per 100,000; adj-RR: 11.6 (range: 6.7-19.9)
Table 3: Incidence of HCC by Cohort Entry HBV DNA Level (N=3851)
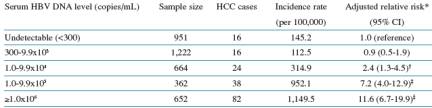
*Adjusted for gender, age, anti-HCV antibody status, cigarette smoking, and alcohol consumption. p<0.001 for the trend.
†p<0.008. ‡p<0.001.
Table 6: Incidence of HCC After Removing HCV Coinfected Subjects
After removing patients with HCV coinfection rates are higher.
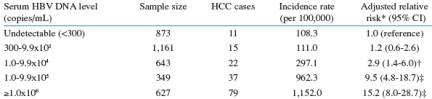
*Adjusted for gender, age, anti-HCV antibody status, cigarette smoking, and alcohol consumption.
p=0.01 for the trend among those who had baseline serum DNA ≥105 copies/mL.
†p<0.01. ‡p<0.001.
Introduction
-Chronic HBV infection has been established as one of the most important risk
factors for HCC1
-Reduction in circulating HBV DNA level is a marker of efficacy for antiviral treatment of chronic HBV infection
-However, there is little data on the prospective dose-response relationship between HBV DNA serum levels and HCC2
Study Objectives
-To determine the independent effects of serum HBV DNA on the development of HCC adjusting for HBeAg status and serum ALT level
-To determine whether increasing levels of HBV DNA are associated with increasing risk of HCC across a biological gradient
-To determine if persistent elevation of HBV DNA is associated with the highest risk of developing HCC
Study Design/Methods
-Community-based prospective cohort study
-7 townships in Taiwan (Sanchih, Chutung, Potzu, Kaoshu on Taiwan Island;
Makung, Huhsi, Paisha on Penghu Islets)
-All 89,293 (47,079 male and 42,214 female) residents between ages of 30 and
65 years were invited to participate between 1991 and 1992
-23,820 (11,973 males; 11,847 females) who gave consent and were free of HCC by ultrasound screening enrolled in this cohort
-Interviews using a standard questionnaire were conducted by trained public health nurses
-Blood samples collected at baseline and follow-up visits were separated and frozen at -70°c on the day of collection
-HBsAg, HBeAg, serum ALT tests were conducted on the baseline serum samples
-All HBsAg-positive subjects had an ultrasound at baseline and at least every 12 months
-Cirrhotic patients had ultrasounds at least every 6 months
HCC Case Ascertainment
-The occurrence of HCC was detected by
-Regular follow-up health examination with ultrasonography
-For complete ascertainment additional data linkage was performed
-To the National Cancer Registry in Taiwan from January 1, 1991 through
June 30, 2004
-To the National Death Certification and National Health Insurance profiles
-Verification of all cases was based on chart review based on the EASL criteria3
-A histopathological examination
-A positive lesion detected by at least two different imaging techniques (including
abdominal ultrasonography, angiogram, or computed tomography)
-One positive imaging study with an elevated serum level of _-fetoprotein >400 ng/mL
RESULTS
HCC Analyses
-Of the 4,155 HBsAg-positive subjects, 3,851 (92.7%) had enough baseline serum sample for HBV DNA testing
-These 3,851 subjects contributed 43,993 person-years as of June 30, 2004
-All HBV DNA analyses done using COBAS Amplicor Roche Diagnostics,
(Indianapolis IN)
- LOQ 300 copies/mL
-Cox proportional hazards model was used for all analyses of the association between HBV DNA and HCC risk
-HCC analyses repeated after removing those with evidence of HCV infection
Subject Demographiics n=3851
Gender
Male, n (%) 2,367 (61.5)
Age, yr
--30-39, n (%): 1,254 (32.6)
--40-49, n (%): 1,058 (27.5)
--50-59, n (%): 1,138 (29.6)
60-65, n (%) 401 (10.4)
Serum ALT level, IU
--Median (min-max): 12.0 (2-765)
--Mean (SD): 18.9 (26.8)
HBeAg
Positive, n (%) 581 (15.1)
Negative, n (%) 3,270 (84.9)
References
1. Chen CJ, et al. J Gastroenterol Hepatol. 1997;12:S294-S308.
2. Yang HI, et al. N Engl J Med. 2002;347:168-174.
3. Bruix J, et al. J Hepatol. 2001;35:421.
|
|
| |
| |
|
 |
 |
|
|