 |
 |
 |
| |
Consensus Interferon + IFN Gamma in PegIFN/RBV Non-Responders
|
| |
| |
Reported by Jules Levin
DDW, Chicago, May 2005
"Response of Chronic Hepatitis C PEG IFN-alpha-2 + Ribavirin Nonresponders to Treatment with IFN Alfacon-1 (15 μg) and IFN Gamma-1b (50 μg)"
Carroll Leevy1, Chris Chalmers2, Lawrence M. Blatt2
1The New Jersey School of Dentistry and Medicine, Newark, New Jersey
2InterMune, Inc., Brisbane, CA
Leevy & Intermune reported these study results & information in a poster at DDW.
ABSTRACT
Introduction: The elimination of serum HCV RNA following IFN-based therapies displays biphasic kinetics with the first order attributed to the direct antiviral effects of IFN-a and the second order attributed to an immune-mediated clearance of infected cells by induction of TH1 cytokines, primarily IFN-£^ (gamma). Patients who have not had >=2 log10 reduction in serum HCV RNA by wk 12 have a 97–100% chance of not responding.
It has been demonstrated that antiviral effects and TH1 responses are enhanced by combining IFN-£^ (gamma) and IFN-a in in vitro systems. Given these data, we conducted a retrospective study of nonresponders to PEG IFN-a-2 + ribavirin (RBV) who were re-treated with IFN alfacon-1 and IFN-£^ (gamma)1b without RBV.
Methods: All patients (N = 50) received PEG IFN-£\-2 and RBV for 12 wk and did not achieve ≥2 log10 drop in HCV RNA. With no washout, patients were retreated with IFN alfacon-1 15 μg SQ daily, and IFN-£^ (gamma)1b 50 μg SQ TIW for 48 wk. Serum HCV RNA was assessed at wk 8, 12, 24, 48 (EOT), and 60 (12 wk posttreatment) to determine virologic response on- and off-treatment. Serum HCV RNA was also assessed at wk 72 to determine SVR.
Consecutive patients from one institution with chronic HCV infection who did not respond to PEG IFN-£\-2/RBV therapy and who agreed to undergo a course of therapy with IFN alfacon-1/IFN-£^ (gamma) 1b.
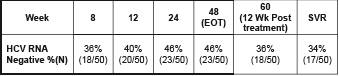
Results: Virologic responses are shown below (Amplicore qualitative assay, Roche Diagnostics):
34% (17/50) SVR
36% (18/50) at week 60 (12 wk post treatment)
48 weeks End of treatment response: 46% (23/50)
week 8: 36% (18/50)
week 12: 40% (20/50)
week 24: 46% (23/50)
One patient interrupted therapy due to constitutional symptoms while all others tolerated therapy well. By wk 48, 13 patients (26%) required filgrastim for reductions in ANC to below 0.75x109/L. After 12 wk of PEG IFN-a-2 and RBV therapy the mean hemoglobin level was 11.6 °” 0.7 g/dL. By wk 8 all IFN alfacon-1 and IFN-gamma 1b patients recovered hemoglobin levels to normal with no use of erythopoeitin.
Only one patient had dosing temporarily interrupted; there were no dose reductions for other patients.
Reductions in ANC were well controlled with filgrastim.
Conclusions: Retreatment of PEG IFN-a-2 + RBV nonresponders with the combination of IFN alfacon-1 and IFN-£^ (gamma) 1b is well tolerated, and analysis of posttreatment virologic response suggests that this treatment may be of potential benefit in these difficult-to-treat patients. This combination did not interfere with hemoglobin recovery, indicating a differential safety profile with respect to RBV-containing regimens. Hemoglobin levels returned
to normal during treatment. A larger, dose-finding study of the combination of IFN alfacon-1 and IFN-£^ (gamma) 1b is ongoing.
Graph. Quantitative changes in mean HCV RNA (copies/mL) after treatment
with IFN alfacon-1 + IFN-£^ 1b in this retrospective analysis (N = 50)
In contrast to previous reports of IFN-a–containing regimens, the combination of IFN alfacon-1 + IFN-gamma 1b therapy led to prolonged viral suppression in all patients after cessation of therapy (P < 0.01, t-test).
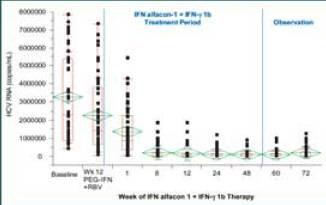
Graph. Quantitative changes in HCV RNA (log10 copies/mL) after treatment with IFN alfacon-1 + IFN-gamma 1b in patients who did not clear virus by
end of treatment (N = 27)
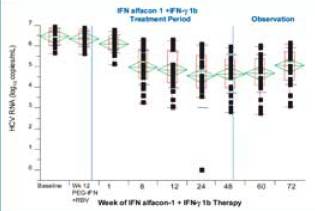
There was a statistically significant reduction in HCV RNA concentration
from baseline to end of observation (P < 0.001, t-test). The mean at baseline was
6.47 ± 0.41 compared with the mean at Week 72, which was 5.04 ± 0.88).
STUDY OBJECTIVE
This retrospective case series examined the safety and efficacy of re-treating patients who did not respond to PEG IFN-a-2 + RBV therapy with a combination of a bioengineered type I IFN (IFN alfacon-1) and a recombinant type II IFN (IFN-£^ (gamma) 1b).
RATIONALE
• Chronic hepatitis C is a disease of T-cell hyporesponsiveness
• Clearance of acute HCV infection requires a potent TH1 response mediated by production of IFN-gamma-4
• Clearance of HCV in chronically infected patients treated with IFN-a requires a shift from a dominant TH2 to a TH1 response (mediated by IFN-gamma)
• IFN-gamma 1b and IFN alfacon-1 display synergistic antiviral activity when combined in vitro5
Combining type I and type II interferons holds promise for the treatment of chronic HCV:
INTRODUCTION
Current treatment of chronic hepatitis C is problematic:
--PEG IFN-a-2 and RBV is effective in ~50% of treatment-naïve patients1,2
--Retreatment of patients who fail Rebetron therapy with PEG IFN-a-2 is
effective in only 4-12% of patients3 (some studies show slightly higher rates of SVR after re-treatment (e.g. 19%+ in Pegasys Canadian EAP).
--Side-effect profile of combination therapies is significant HCV patients who
do not respond to treatment with PEG IFN-a-2 + RBV are a growing public health
concern.
IFN alfacon-1 (Consensus Interferon, Infergen)
• Type I interferon
• Approved for the treatment of HCV
• Binds and signals through type I receptor
• Bioengineered: consists of the most common amino acids in naturally
occurring IFN-£\ subtypes
• 10- to 100-fold higher biological potency in vitro compared with IFN-£\-2b/2a6
• Promising data in PEG IFN-a-2/ RBV nonresponders7
IFN-£^ (gamma)1b
• Type II interferon
• Approved for the treatment of chronic granulomatous disease and
osteopetrosis
• Binds and signals through type II receptor
• Has a spectrum of biologic effects
- Antiviral
- TH1 cytokine
- Antiproliferative
- Immunomodulatory
SAFETY
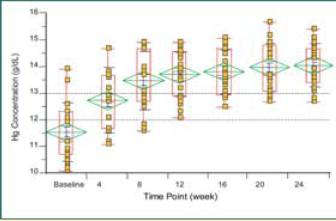
Hemoglobin levels in all patients returned to within normal range while
patients were receiving IFN alfacon-1 and IFN-£^ 1b. No growth factors were
used.
Graph. Hemoglobin recovery in patients receiving IFN alfacon-1
(15 μ}g daily) + IFN-£^ 1b (50 μ}g TIW) after failing to respond
to treatment with PEG IFN-a-2 + RBV (N = 50)
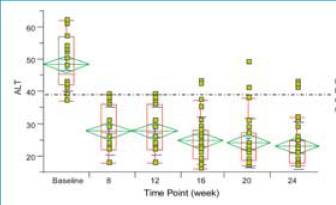
Patients had rapid reduction in ALT after initiation of therapy.
Graph. Reductions in serum ALT in patients receiving IFN alfacon-1
(15 μ}g daily) + IFN-£^ 1b (50 μ}g TIW) (N = 50)
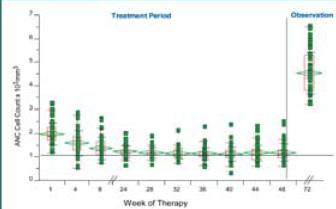
14 patients (28%) had reductions in WBC (ANC < 0.75 x 109) that
required use of G-CSF.
• Dosing was temporarily interrupted for one patient; there were no
dosing reductions for other patients
• Neutropenia was manageable with G-CSF and did not require
dose reductions
graph. Changes in ANC after treatment with IFN alfacon-1 +
IFN-£^ (gamma) 1b (N = 50)
References
1 Fried M, et al. 2001. Pegylated (40kDa) Interferon Alfa-2a (PEGASYS) in Combination With Ribavirin: Effi cacy And Safety Results From a Phase III, Randomized, Actively Controlled, Multicenter Study. Gastroenterology. 2001;120,
A-55.
2 Manns MP, et al. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis c: A randomised trial. Lancet. 2001;358:958-965.
3 Shiffman ML, et al. Retreatment of chronic hepatitis C virus infection in patients who failed to achieve sustained virologic response. Minerva Gastroenterol Dietol.
2004;50(1):37-49.
4 Thimme R, et al. Viral and immunological determinants of hepatitis C virus clearance, persistence, and disease. Proc Nat Acad Sci. 2002;99:15661-15668.
5 Blatt LM, et al. Synergistic Effects of Type 1 (Infergen) and Type 2 (Actimmune) Interferons in Preclinical Models of HCV: Demonstration of Potential Efficacy. Hepatology. 2003;38 (Suppl 1):80A (AASLD 2003).
6 Blatt LM, et al. The Biologic Activity and Molecular Characterization of a Novel Synthetic Interferon-Alpha Species, Consensus Interferon. J Interferon Cytokine Res 1996;16:489-499.
7 Kaiser S, et al. Successful retreatment of peginterferon nonresponder patients with chronic hepatitis C with high dose consensus interferon induction therapy.
Gastroenterology. 2003;124(4-Suppl 1): pA-700.
8 Tong MJ, et al. Prediction of response during interferon alfa 2b therapy in chronic hepatitis C patients using viral and biochemical characteristics: a comparison. Hepatology. 1997;26(6):1640-1645.
|
|
| |
| |
|
 |
 |
|
|