 |
 |
 |
| |
Resistance to Adefovir (Hepsera) Can Be Associated with Significant Virologic Breakthough and Fatal Hepatic Decompensation
|
| |
| |
Reported by Jules Levin
DDW Conference
May 2005
Authors: S Fung, M Hussain, A Andreone, S Han, R Reddy, A Regev, E keefe, J Marrero, P Richtmyer, A Lok
Abstract M921
Background: Adefovir resistance occurs less frequently and later in the course of treatment compared to lamivudine (3TC). Susceptibility of ADV-resistant hepatitis B virus (HBV) mutants is only reduced by 3-10 fold, suggesting that virologic breakthrough and clinical deterioration are unlikely. However, there are very little data regarding the clinical course and response to salvage therapy in ADV-resistant patients.
AIMS: To describe the clinical course of hepatitis B patients found to have ADV-resistant mutations.
<
Surveillance for ADV-resistant mutation was performed on patients receiving ADV, who had a suboptimal response (<2 log decrease in HBV DNA or HBV DNA >4 log after 1 year of treatment), and in those with virologic breakthrough (>1 log increase in HBV DNA after initial suppression).
<
HBV DNA was quantified using a PCR assay. ADV-resistant mutation was detected using a line probe assay and confirmed by direct sequencing of the HBV polymerase gene.
RESULTS
Seven male patients, mean age 49 (+/-13) years, were found to have ADV resistance between 9/03 and 9/04.
All patients had prior treatment with lamivudine for 27 (+/-14) months and breakthrough infection or genotypic resistance to lamivudine was confirmed in 6 patients.
<
At the start of ADV, median ALT was 75 IU/L (range, 27-1161); mean HBV DNA was 7 (+/-1) log copies/ml;
4 patients were HBeAg+ve; and 2 had liver transplants.
Six patients were switched to ADV with <1 month overlap with lamivudine; 1 patient continued to receive lamivudine.
During treatment, 6 patients had a suboptimal virologic response.
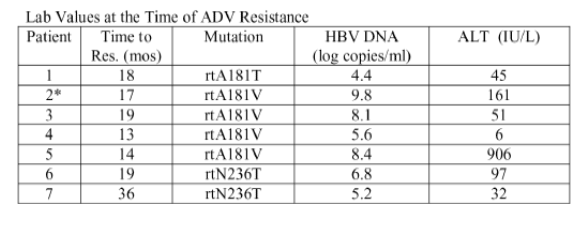
At the time of ADV resistance, which occurred at a mean of 19 ± 8 months after treatment initiation, median ALT was 51 IU/L (range, 6-906) and HBV DNA increased to over 6 log copies/ml in 6 patients.
<
After detection of ADV resistance, hepatic decompensation occurred in 2 patients, 1 of whom died.
Salvage therapy with lamivudine, entecavir or tenofovir was given to 6 patients; 3 of 5 patients with > 6 months of follow-up had > 3 log reduction in HBV DNA.
AUTHOR CONCLUSIONS
Resistance to ADV is not rare and may be associated with substantial increases in HBV DNA level and fatal hepatic decompensation. Surveillance for ADV resistance and institution of salvage therapy should be considered in immunosuppressed or cirrhotic patients.
|
|
| |
| |
|
 |
 |
|
|