| |
New NNRTI GW695634: safety & antiviral activity; 7-day monotherapy study
|
| |
| |
Reported by Jules Levin
"Antiviral Activity and Safety of GW695634, A Novel Next Generation NNRTI in NNRTI-resistant HIV-1 Infected Patients"
Reported by Jules Levin
S Becker1, J Lelezari2, C Walworth3, P Kumar4, J Cade5, J Ng-Cashin6, Y Kim6, J Scott6, M St. Clair6, L Jones6, and W Symonds6 On Behalf of the NN210005 Study Team
1pacific Horizon MedicalGgroup, San Francisco, CA; 2 Quest Clinical Research, San Francisco, CA; 3 Orange County Center for Special Immunology, Fountain Valley, CA; 4 Georgetown University Hospital, Washington, DC; 5HIV Wellness Center, Las Vegas, NV; 6GlaxoSmithKline, RTP, NC.
This poster was presented at IAS-Rio July 2005.
GW695634 is a novel next generation NNRTI in early development for the treatment of HIV-1. In vitro, GW678248 potently inhibits both wild-type HIV-1 and NNRTI-resistant mutants, including virus with K103N and Y181C mutations.
This study investigated the safety and tolerabilility of GW695634 and GW678248 in NNRTI-experienced HIV-1 infected subjects over a 7-day monotherapy treatment period, and evaluating the initial antiviral & immunological activity of GW678248 in NNRTI-experienced subjects. Single & repeat dose PK in NNRTI-experienced subjects was studied and data will be presented at future meeting in 2005.
BRIEF SUMMARY: After 7 days of monotherapy viral load reductions were -1.1 log to -1.6 logs. 5/9 patients receiving highest dose of 400mg bid and 2/9 patients receiving 300mg bid in this study experienced rash, but only 1/10 receiving 200mg bid experienced rash. See below for additional safety and antiviral activity data.
This was a multi-center,double-blind, randomized, parallel group, placebo-controlled, dose-ranging study comparing 4 doses of GW695634 or placebo administered as monotherapy for 7 days:
100 mg bid (n=10)
200mg bid (n=10)
300 mg bid (n=10)
400 mg bid (n=10)
or placebo (n=10)
At screening patients were required to be:
NNRTI resistant by current genotype or historical evidence
CD4 <100
HIV RNA >2000 c/ml
No ARTs were permitted for 4 weeks prior to study entry
Plasma HIV RNA, PK, and safety parameters were monitored daily.
AUTHOR CONCLUSIONS
--GW695634, the pro-drug of GW678248, demonstrated potent antiviral activity (-1.1 to -1.6) log copies/ml plasma decline) across all doses studied in this NNRTI-experienced population following 7 days of monotherapy.
--3 subjects harbored viruses which were generally resistant to first-generation NNRTIs, but responded to GW695634.
--GW695634 was generally well tolerated over 7 days.
These data provide "proof-of-concept" which supports further clinical development of GW695634 for use in subjects failing currently marketed NNRTIs.
RESULTS
--46 subjects were enrolled with a median of 2 NNRTI-associated primary mutations (range 1-4), including L100I, K103N (55%), V108I, Y108I, Y181C/I (30%), Y188C/L/H, G190S/A, and P225H. The baseline NNRTI resistance data are summarized:
--17 (39%) of subjects had historical evidence of prior NNRTI resistance mutations, but were wild-type by population sequencing at study entry.
--of the 44 subjects with baseline genotype information, 27 (61%) had at least 1 NNRTI mutation ate baseline: 9 (20%) had single mutations, 10 (23%) had 2 mutations, and 8 (18%) had 3 mutations.
5% had L100I
40% K103N
5% V106A/M/I
25% V108I
20% Y181C
10% Y188C/L/H
10% G190S/A
10% P225H
In vitro data in poster showed 20 fold resistance of GW678248 to mutant NNRTI virus with Y188L, and several fold resistance to mutants K103N/G190A and V106I//Y181C.
--44 NRTI-experienced HIV-1 infected men & women >18 yrs old completed the study: 1 patient discontinued the study early due to a non-serious AE at day 6 and 1 was lost to followup on day 1.
--the 5 treatment groups had median baseline plasma viral load values ranging from 4.4 to 4.6 log c/ml and median CD4 counts ranging from 230-345. Average age was 41-46. 2 females in study. 8 blacks, 3 Hispanics, 34 Whites.
--On day 8, median plasma HIV RNA-1 reductions were -1.2 log c/ml (100mg), -1.1 log c/ml (200mg), -1.6 (300mg), -1.3 (400mg), and +0.14 (placebo).
--significant changes in day 8 VL compared to placebo were seen across all GW695634 dose groups (p-value range <0.0001 to 0.005).
--GW695634 was generally well tolerated over 7 days. As summarized, the most common frequent drug-related adverse events were diarrhea, nausea, and rash. The majority of the AEs were mild to moderate in intensity.
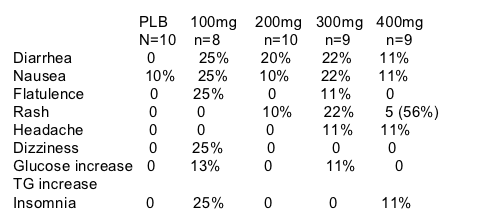
CLINICAL SAFETY DATA
In total 289 patients and healthy volunteers have received GW695634 in 6 Phase 1 studies. 155 patients/volunteers have received GW695634G in multiple dose studies: 39 (25%) developed drug-related rash; diffuse maculopapular rash (grade 2) in majority (n=35); additional isolated, local, oral mucosal involvement in 4 cases (grade 3); 6 withdrawals for asymptomatic serum transaminase elevations.
|
|
| |
| |
|
|
|