 |
 |
 |
| |
Once-Daily vs.Twice-Daily Lopinavir/ritonavir in
Antiretroviral-Naive Patients: 96-Week Results
|
| |
| |
Reported by Jules Levin
3rd IAS Conference on HIV Pathogenesis and Treatment
24-27 July 2005, Rio de Janeiro, Brazil
poster WePe12.3C12
AUTHOR CONCLUSIONS:
The study population had relatively advanced HIV disease, as approximately 45% of subjects had baseline CD4 count below 200 cells/mm3, including 16%
with CD4 count below 50 cells/mm3, and 38% had baseline HIV-1 RNA above 100,000 copies/mL.
-At Week 96, by intent-to-treat analysis with noncompleters considered failures, 57% of subjects in the QD LPV/r+TDF+FTC regimen demonstrated HIV-1
RNA <50 copies/mL, compared to 53% for the regimen of BID LPV/r with QD TDF + FTC (p=0.58).
-Through 96 weeks, noninferiority of the LPV/r QD regimen compared to LPV/r BID-based regimen (ITT NC=F) was confirmed by the 95% confidence
interval for the difference (QD minus BID) in response proportions (-10% to 19%).
-No subject developed LPV or TDF resistance through 96 weeks.
-In this study with the soft gel capsule formulation of LPV/r, gastrointestinal events were the most common adverse events, with a higher rate of diarrhea in
the QD arm.
The new tablet formulation of Kaletra is on an accelerated fast track approval by the FDA. You can read about the presentation on the pk of single doses of this new tablet in a report from the Rio Conference on the NATAP website. Pk on multiple doses is expected to be presented soon. The data presented at Rio suggested there might be less diarrhea & GI upset associated with the new tablet formulation.
See tables below regarding detailed resistance, safety and lipid profile.
Authors: JM Molina1, A Wilkin2, P Domingo3, R Myers4, J Hairrell5, C Naylor5,T Podsadecki5, M King5, G Hanna5
1Hôpital Saint Louis, Paris, France; 2Wake Forest University School of Medicine,Winston-Salem, NC;
3Hospital de la Santa Creu i Sant Pau, Barcelona, Spain; 4Phoenix Body Positive, Phoenix, AZ; 5Abbott Laboratories, Abbott Park, IL
BACKGROUND
Lopinavir (LPV) is an HIV protease inhibitor (PI) that is co-formulated with ritonavir (r), which functions as a pharmacokinetic enhancer. LPV/r is marketed as Kaletra. The approved adult dose of LPV/r is 400/100 mg twice-daily (BID). In the U.S., adult dose of LPV/r 800/200 mg once-daily (QD) is also approved for
antiretroviral-naive patients. Antiviral activity of LPV/r has been demonstrated in antiretroviral-naive and PI-experienced patients.
A once-daily antiretroviral regimen including LPV/r may offer an advantage with regard to convenience while maintaining antiviral potency in antiretroviralnaive
patients. In a pilot study (Study 056), antiretroviral-naive, HIV-1-infected adults (N=38) received LPV/r 800/200 mg QD or 400/100 mg BID with stavudine and lamivudine given BID.1,2 LPV/r 800/200 mg QD produced similar Cmax and AUC, and lower and more variable Ctrough compared to 400/100 mg
BID. However, virologic response through 72 weeks was similar.2 Further, the inhibitory quotient (IQ; Ctrough/IC50 for wild-type HIV-1) achieved with once-daily LPV/r compares favorably to that of other QD PIs.3
Based on these pilot results, Study 418 was initiated to further assess the pharmacokinetics, antiviral activity and safety of a once-daily dosing regimen for
LPV/r in antiretroviral-naive patients. In Study 418, subjects received LPV/r with tenofovir disoproxil fumarate (TDF) 300 mg and emtricitabine (FTC)
200 mg once-daily. Subjects receiving LPV/r 800/200 mg QD demonstrated slightly higher lopinavir Cmax, similar AUC, and lower Ctrough compared to
400/100 mg BID.4 The median IQ was 49 for QD and 94 for BID.
Analysis of the comparative safety and efficacy through 48 weeks showed noninferiority of the LPV/r QD regimen compared to the LPV/r BID regimen, with a higher rate of diarrhea in the LPV/r QD arm.5 This analysis presents the comparative safety and efficacy through 96 weeks.
METHODS
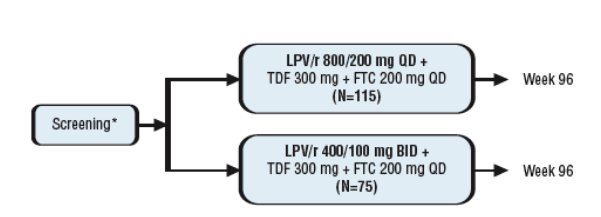
Study 418 is the first trial of an entirely once-daily LPV/r-based regimen (Figure 1).
-Randomized, open-label, multi-center, international study.
-Subjects were antiretroviral-naive, with plasma HIV-1 RNA >1,000 copies/mL and any CD4 count.
-190 subjects were randomized 3:2 to LPV/r 800/200 mg QD (n=115) or 400/100 mg BID (n=75).
-All subjects also received TDF 300 mg and FTC 200 mg QD.
Analysis
-Plasma HIV-1 RNA levels were assessed using Roche Amplicor HIV-1 Monitor Ultrasensitive Quantitative PCR Assay, Version 1.5 (limit of quantitation,
50 copies/mL).
-The proportion of subjects with HIV-1 RNA below 50 copies/mL was assessed using an intent-to-treat, noncompleter=failure (ITT NC=F) method, in which
missing values were considered failure unless the immediately preceding and following values were below 50 copies/mL. An observed data (OD) method
was also used, in which missing values were excluded from the analysis.
-Analysis was also conducted using the U.S. FDA time-to-loss of virologic response (TLOVR) algorithm, in which subjects are considered responders if they achieve two consecutive HIV-1 RNA levels <50 copies/mL and maintain the levels through the timepoint of interest. Subjects who subsequently
demonstrate two consecutive rebound HIV-1 RNA levels >50 copies/mL or who discontinue from the study are considered non-responders.
-For each HIV-1 RNA result above 500 copies/mL between Weeks 12-96, isolates were submitted for genotypic resistance testing, as were corresponding
baseline isolates for each subject. Resistance to lopinavir was defined as the emergence of any mutation at protease amino acid 8, 30, 32, 46, 47, 48, 50,
82, 84 or 90 with corresponding phenotypic lopinavir resistance of at least 2.5-fold versus wild-type. Resistance to TDF was defined by the development of
any new mutation at amino acid 41, 65, 67, 70, 210, 215, 219 or the 69 insertion mutation in reverse transcriptase. Resistance to FTC was defined by the
development of the M184V/I/T mutation in reverse transcriptase.
-Cumulative incidence of adverse events through 96 weeks was summarized.
Baseline Characteristics
-Demographics and baseline disease characteristics were similar between treatment groups, with over 20% female and about 45% non-Caucasian subjects

-The study population had relatively advanced HIV disease, as approximately 45% of subjects had baseline CD4 count below 200 cells/mm3, including 16%
with CD4 count below 50 cells/mm3, and 38% had baseline HIV-1 RNA above 100,000 copies/mL.
-The overall mean baseline viral load was approximately 65,000 copies/mL.
Efficacy
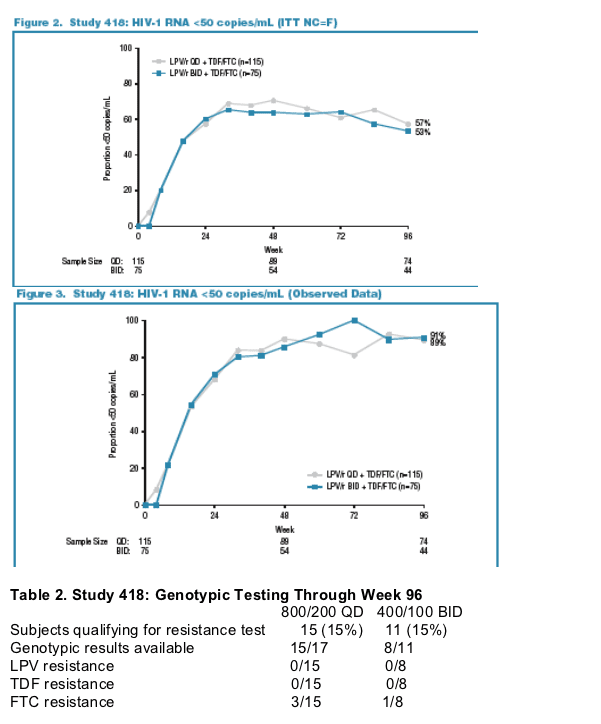
-In the ITT (NC=F) analysis (Figure 2) and the OD analysis (Figure 3), a similar proportion of subjects achieved HIV-1 RNA below 50 copies/mL through
96 weeks.
-By FDA TLOVR analysis, Week 96 response rates were 57% QD and 55% BID. On treatment: 90% < 50 copies/ml.
-Based on the ITT (NC=F) analysis, the 95% confidence interval for the difference (QD minus BID) in Week 96 response proportions was (-10%, 19%).
-Results of genotypic testing were available for 23 subjects with HIV-1 RNA >500 copies/mL occurring at any time during Weeks 12-96. No subject
demonstrated LPV or TDF resistance, and only 4 subjects demonstrated FTC resistance (Table 2).
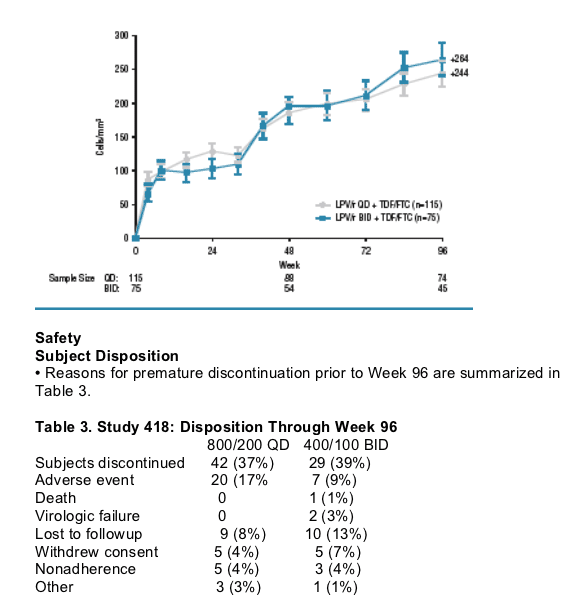
-CD4 cell count mean increases (+250) from baseline were comparable between treatment groups (Figure 4).
Figure 4. Study 418: CD4 Cell Count Mean (±SE) Change from Baseline
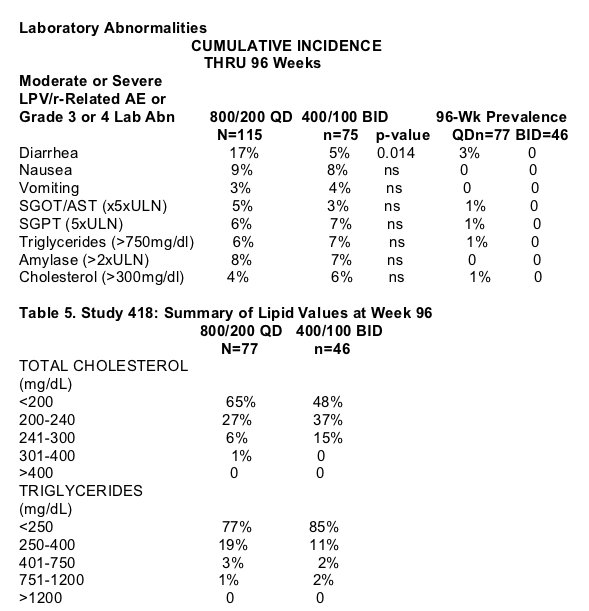
-A higher rate of discontinuations due to adverse events was observed in the QD group, while higher rates of loss to follow-up were observed in the BID group.
-Adverse events resulting in discontinuation were generally gastrointestinal in nature. One subject in the BID group on chronic prednisone therapy for
myositis died of multi-organ failure after 6 weeks on study, following a diagnosis of adenocarcinoma. The event was considered unrelated to study drugs.
Adverse Events/Laboratory Abnormalities
-Moderate or severe, drug-related adverse events and grade 3 or 4 lab abnormalities occurring in >3% of patients in either treatment group are shown in Table 4.
Table 4. Study 418: Most Common Adverse Events and Grade 3/4 Laboratory Abnormalities
Overall for 98% of subjects, the maximum creatinine value was 21.5 mg/dL. One subject in each group demonstrated creatinine >3.0 mg/dL (acute renal
failure - ARF).
-ARF occurred at Week 38 in a 54-year-old male subject who required temporary hemodialysis. Renal biopsy demonstrated tubulointerstitial nephritis. The subject discontinued the study and creatinine levels returned to near normal.
-ARF occurred at Week 34 in a 75-year-old male subject with a baseline creatinine clearance of 40 mL/min who was begun on full dose TDF. The subject
received one hemodialysis session. Renal biopsy demonstrated non-specific changes with focal degenerative signs (cytoplasmic vacuolization) in some
renal tubules. LPV/r was restarted but TDF was replaced by stavudine 30 mg BID and the dose of emtricitabine was reduced to 200 mg every 72 hours. His
creatinine level continued to decrease. TDF dosing recommendations implemented after initiation of this study indicate that every other day dosing of TDF would have been most appropriate for this subject based on creatinine clearance.
-No subject demonstrated a new creatinine elevation >1.5 mg/dL after Week 48.
REFERENCES
1. Bertz R, Foit X, Ye L, et al. Pharmacology of Antiretroviral Chemotherapeutic Agents: Pharmacokinetics and Therapeutic Drug Monitoring, 9th Conference on
Retrovirus and Opportunistic Infections, Seattle, 2002 (oral #126).
2. Feinberg J, Bernstein B, King M, et al. Once Daily vs. Twice Daily Kaletra (lopinavir/ritonavir) in Antiretroviral-naive HIV+ patients: 72-week follow-up. XIV
International AIDS Conference, Barcelona, Spain, 2002 (Abstract TUPEB4445).
3. Stevens RC, Kakuda TN, Bertz R, et al. Inhibitory Quotient of Protease Inhibitors Using a Standardized Determination of IC50, 4th International Workshop on Clinical Pharmacology of HIV Therapy, Cannes, France, 2003 (poster 4.2).
4. Chui Y-L, Foit C, Gathe J, et al. Multiple-Dose Pharmacokinetics and Initial Antiviral Effect of Once Daily Lopinavir/ritonavir (LPV/r) in Combination with Tenofovir (TDF) and Emtricitabine (FTC) in HIV-Infected Antiretroviral-Naive Subjects (Study 418), Second IAS Conference on HIV Pathogenesis and Treatment, Paris, France, 2003 (abstract #839).
5. Gathe J, Podzamczer D, Johnson M, et al. Once-Daily vs. Twice-Daily Lopinavir/ritonavir in Antiretroviral-Naive Patients: 48 Week Results. 11th Conference on Retrovirus and Opportunistic Infections, San Francisco, CA, 2004 (Poster 570).
|
|
| |
|
 |
 |
|
|