 |
 |
 |
| |
Tipranavir & TDM
|
| |
| |
Reported by Jules Levin
3rd IAS Conference
July 24-27, 2005
Rio de Janeiro
Boehringer Ingelheim reported these study results in an oral presentation at IAS.
"Tipranavir/Ritonavir (TPV/r) 500 mg/200 mg BID drives week 24 viral load (VL) below 400 copies/mL when combined with a second active drug (T-20) in highly protease inhibitor-experienced HIV+ patients"
H Valdez, MD; SM McCallister, MD; DB Hall, PhD; J Scherer, MD; VM Kohlbrenner, MD; DL Mayers, MD Boehringer Ingelheim Pharmaceuticals
Ridgefield, CT
Background provided by presenter.
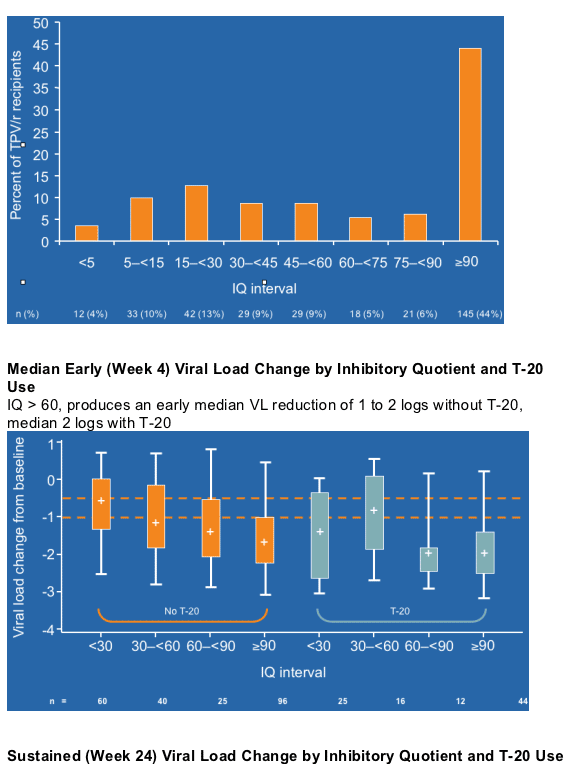
--Studies have shown that for treatment-experienced patients receiving a PI, the magnitude of the inhibitory quotient (IQ) correlates with the magnitude of week 24 viral load reduction.
--Therapeutic drug monitoring (TDM) has been proposed as a tool to achieve adequate IQs and minimize toxicity in highly treatment-experienced (HTE) patients.
--It is unknown whether, with the levels achieved with ritonavir-boosted protease inhibitors, TDM is necessary for the management of HTE patients.
--To ascertain the proportion of TPV/r HTE recipients (500 mg/200 mg BID) attaining an adequate IQ and to describe the magnitude of virologic response with a given TPV IQ, with and without a second active agent, a randomly selected sample of TPV/r recipients in the RESIST 1 and 2 trials who had their isolates phenotyped was studied.
AUTHOR CONCLUSIONS:
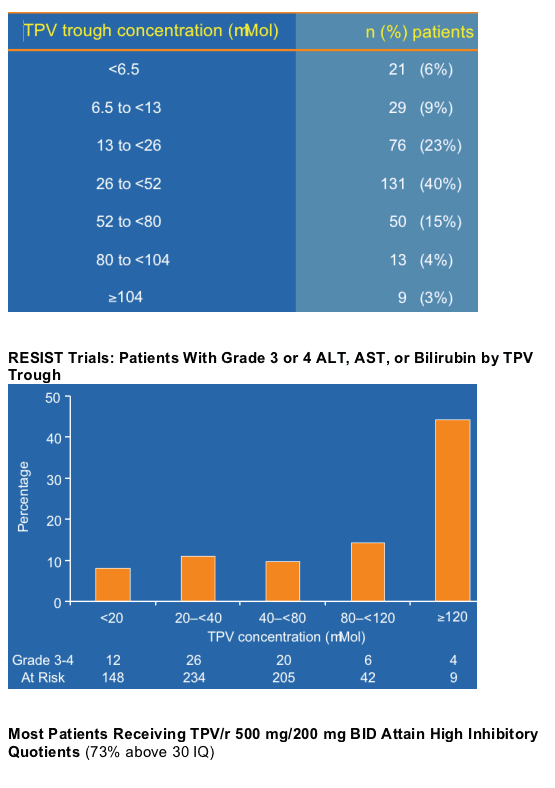
--Most HTE patients who receive TPV/r at a dose of 500 mg/200 mg BID achieve TPV trough concentrations >60-fold in excess of the TPV IC50 of their isolates.
In this patient population, there was often a paucity of agents available to construct a viable background.
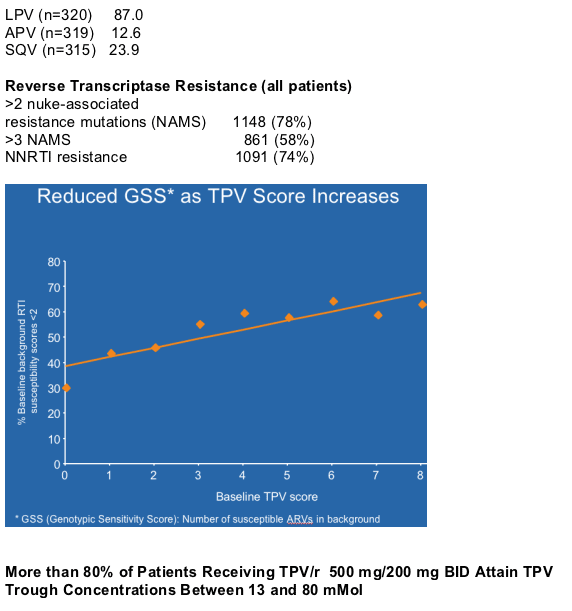
--TPV IQ of 30 or more produces large (>1 log10) early (4 weeks) VL reductions, but these are sometimes not sustained because of lack of background regimens.
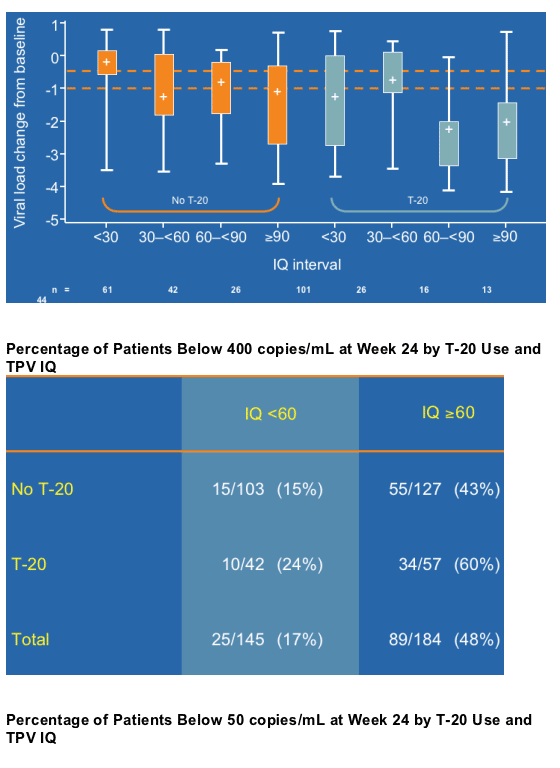
IQ > 60, produces an early median VL reduction (at week 4) of 1 to 2 logs without T-20, median 2 logs with T-20; 73% of patients had above 30 IQ, in this analysis from RESIST.
--When used with another active drug such as T-20, TVP IQ of 30 or more achieves VL below the limit of detection (<400 copies/mL) in the majority of hard-to-treat patients at week 24.
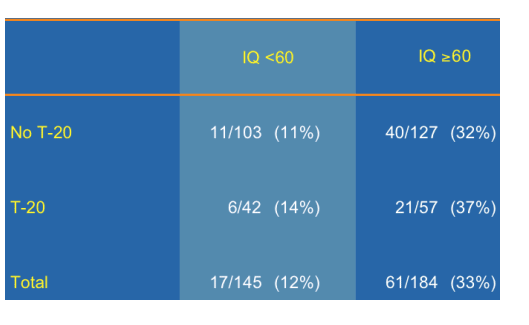
--One third of these hard-to-treat patients with TPV IQs ≥60 achieve a VL below 50 copies/mL at week 24. (43% achieved <400 c/ml without T20, 60% achieved <400 c/ml with T20; see pix below).
--The TPV concentrations achieved in most patients are not associated with increased levels of hepatotoxicity. The analysis presented found that most people who develop G3 or greater ALT with TPV do not do so with very high levels but TPV concentrations between 20 and 80, within the target range.
--TDM does not appear necessary when the TPV/r dose of 500 mg/200 mg BID is used among highly treatment experienced patients.
See pictures of tables below.
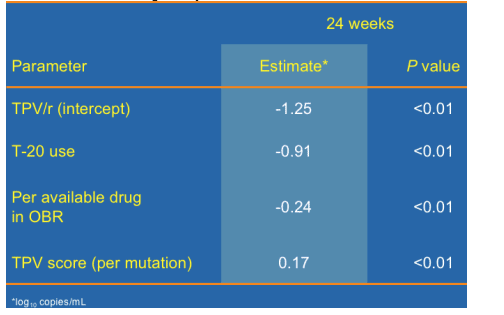
Predictors of TPV/r Antiviral Response
Multiple Regression Model (*log10 copies/mL)
--Looking at predictors of the 24-week viral load response, the use of TPV, use of T-20 and number of active drugs in the OBR all add to produce the viral load reduction while the number of TPV score mutations reduces the response. Specifically, use of TPV/r is associated with a 1.25 log response, addition of T-20 adds an additional .91 logs, and each genotypically available RT inhibitor adds an additional quarter of a log of activity. Each TPV mutation reduces the response by 0.17 logs such that 7 or more TPV mutations would eliminate the TPV effect, correlating nicely with earlier TPV score results.
STUDY METHODS
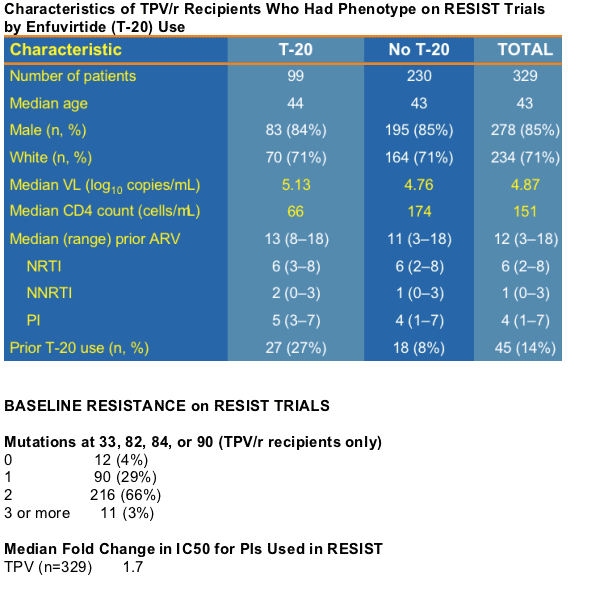
--Phenotypic resistance testing of baseline HIV-1 isolates for 400 TPV/r recipients participating in RESIST trials was performed at VIRCO.
--Trough TPV concentrations were obtained in RESIST 1 and 2 trial participants at weeks 2, 4, and 16 or 24
--IQ was calculated using:
Geometric mean TPV Cmin
(divided by)
Fold wild-type change x 0.058 (wt IC50) x 3.75
Characteristics of TPV/r Recipients Who Had Phenotype on RESIST Trials by Enfuvirtide (T-20) Use
|
|
| |
|
 |
 |
|
|