 |
 |
 |
| |
Simplification to Kaletra Monotherapy
|
| |
| |
Reported by Jules Levin
The concept of simplification to Kaletra monotherapy from triple-drug HAART continues to receive much attention. Large studies examining Kaletra monotherapy are ongoing and enrolled. As well, Reyataz/r monotherapy is being studied. The results of small pilot studies show that full viral suppression with this approach can be attained. Until we have results from large studies and understand the type of patients who can successfully use this approach, it remains experimental and use of the recommended Standard of Care is reliable.
There were 3 related studies presented at Rio on simplification of 3-drug HAART to Kaletra monotherapy. These pilot studies are exploring if you can simplify therapy to Kaletra monotherapy after full suppression <50 copies/ml on a standard HAART regimen. The studies reported 48-week results, risk factors for the patients who lost virologic suppression on Kaletra monotherapy, and an evaluation of viremia levels using a sensitive viral load assay. Three patients lost viral suppression (>500 copies/ml twice 2 weeks apart) on Kaletra simplification (n=21) vs none who remained on triple-HAART (n=21). Of note, the 3 viral failures resuppressed virus upon adding back 2 nukes. Non-adherence, missed doses, and a significantly shorter time with HIV RNA <50 copies/ml before simiplification (40 vs 132 months) were the two risk factors identified for the patients who lost virologic suppression. The study authors added however, although suboptimal adherence seems to facilitate loss of virologic suppression while receiving LPV/r maintenance, it should be noted that perfect adherence does not appear to be an absolute requirement for success of LPV/r maintenance therapy. Actually, half of the patients who did not lose virologic suppression had drug refill scores of 70-94% (see explanation of 'drug refill scores' below).
JE McKinnon reported for his research group, which included John Mellors (oral WeOa0203), on levels of persistent viremia after simplification using an experimental Roche assay with a limit of 3 copies/ml. For patients who remained suppressed <50 copies/ml the levels of virus using the sensitive assay were the same, median 3-6 copies/ml, throughout the 48 weeks. So patients who simplified to Kaletra monotherapy and maintained <50 copies/ml did not have different levels of persistent viremia when using this sensitive assay. Patients on Kaletra monotherapy had the same percent of undetectable (30%) viremia as patients remaining on triple-HAART through 48 weeks. However, when comparing patients on Kaletra monotherapy who lost viral suppression to those who did not lose viral suppression: at baseline there was no difference in median viral load, but at week 8 median viral load was higher (not significant) among the 4 failure patients compared to the patients who remained suppressed. At week 12 the difference in median viral load between failures and non-failures appeared a little wider, and by week 24 the failures (n=3) had a statistically significant higher median viral load (about 3 logs) compared to patients who retained viral suppression.
ADDITIONAL SUMMARY:
In the OK Study (poster WePe 12.3C05), conducted in Spain, 42 patients receiving Kaletra plus 2 nukes and <50 copies/ml for at least 6 months were randomized to remain on HAART or simplify to Kaletra monotherapy. 21 patients were randomized to either simplifuication or remaining on triple-HAART. Patients who simplified had been <50 c/ml for 28 months, CD4 before switch was 662, nadir CD4 was 139, and viral load previously was 100,000 c/ml before HAART. Patients who remained on triple-HAART had been <50 c/ml for 15 months, CD4 was 585, nadir CD4 was 90, and viral load previous to HAART was 100,000 c/ml. Patients in both groups were a mix of IVDUs and MSM.
At week 48, 81% on Kaletra monotherapy and 95% on triple-HAART had <50 copies/ml (ITT, MD=F). In the triple-HAART there was 1 discontinuation due to hyperlipidemia, and on Kaletra simplification there were 3 patients who lost viral suppression and 1 lost to followup. Viral suppression was defined as 2 viral loads >500 copies/ml 2 weeks apart. All 3 patients with loss of viral suppression added nukes (either AZT/3TC or d4T/3TC) and re-suppressed viral load <50 copies/ml. Lipid values were normal before simplification and there was no change at week 48.
Preliminary data show that failure of these 3 patients was not associated with the development of primary resistance mutations. One patient had no genotypic mutations; the second patient developed L63P & V77I; the 3rd patient developed V77I.
For two patients Arribas reported lopinavir drug level troughs while <50 c/ml. For 1 patient troughs were 5.78 ug/mL at week 1 and 4.57 at week 16. But drug levels were not reported after that. For the 2nd failure trough was 9.4 ug/ml at week 1 and 5.7 ug/ml at week 24, but no more drug levels were reported.
RISK FACTORS for loss of virologic suppression for patients on Kaletra monotherapy:
The persistent residual viremia level (replication below 50 copies/ml) was not different between viral failures and non-failures, and this was the subject of an oral presentation which is discussed below. As potential risk factors for loss of viral suppression the study considered: AIDS diagnosis, pre-HAART HIV RNA, CD4 count (baseline & nadir), time with HIV RNA <50 c/ml, time on Kaletra prior to simplification, use of Kaletra as first PI, adherence by drug refill score [(number of days of antiretroviral dispensation / number of total days until next dispensation) x 100], and adherence by self-report using the GEEMA adherence questionnaire.
The GEEMA adherence questionnaire has 6 questions:
1. did you ever forget to take your medicine?
2. are you careless at times about taking your medicine?
3. sometimes if you feel worse, do you stop taking your medicines?
4. thinking about last week, how often have you not taken your medicine?
5. did you not take any of your medicine over the past weekend?
6. over the past 3 months, how many days have you not taken any medicine at all?
Responses were quantified to questions 4 and 6. Patients were classified as adherent or non-adherent when there was a positive response to any of the 4 qualitative questions included in the questionnaire.
Patients with loss of virologic suppression had:
1. significantly shorter time with HIV RNA <50 c/ml before starting Kaletra (median 40 [range 30-84] vs 132 months [range 40-331], p=0.02.
2. significantly lower adherence as measured by GEEMA questionnaire. Total days without medication was significant (p=0.008): 0 for patients who maintained suppression & median of 3 days for patients who lost suppression. Total missed doses in week prior to the study visit was significant (p=0.013): 0 for patients who maintained suppression, and median of 3 missed doses in the week for patients who lost suppression.
3 out of 4 patients who lost viral suppression had adherence rates (by drug refill scores) that could justify the outcome (59%, 60%, 79%). Only 1 out of 10 patients with drug refill scores between 70 & 95% lost viral control (drug refill score=79%). In one patient, loss of viral suppression was observed despite good adherence (drug refill score=100%).
PERSISTENT VIREMIA SUB-STUDY
Plasma samples from each participant in the OK Study were analyzed from screening baseline and at weeks 4, 8, 12, 24, and 48 after randomization. They used a modified Roche Amplicor HIV RNA-1 assay (v1.5). The assay's protocol was modified to allow for quantification of about 3 copies/ml of viral RNA. Four HIV RNA controls (12.5, 6.25, 3.125 and 0 copies/ml) were run with each assay. Longitudinal samples from a given subject were batch tested to eliminate inter-assay variability. A p-value of 0.05 was used as the cutoff for statistical significance.
Using this experimental viral load assay about 30% of the samples tested had undetectable viremia (<3 copies/ml). Among the patients who remained suppressed throughout the 48 weeks the median level of virus was about the same, 3-6 copies/ml at each time-point they checked. Of note, there was no difference in the median level of viremia at baseline for the subjects in the LPV/r alone arm between failures and non-failures. But, in the LPV/r alone arm, a trend towards increasing viremia was found in the failure samples at week 8 as compared to the non-failures (p=0.087) (ed note: the number of patient samples was small so perhaps that's why a statistically significant difference was not seen), with a statistically significant difference in median viremia at week 24 (p=0.007).
DETAILS OF OK STUDY
Jose Arribas (Madrid, Spain) reported 48-week results from the OK Study: "Lopinavir/ritonavir as a single-drug therapy for maintenance of HIV-1 viral suppression. A randomized, controlled, open-label, pilot clinical trial". Arribas said LPV/r is an appropriate candidate for single-drug HAART because of its high potency, high genetic and pharmacological barriers to resistance, extremely low risk of resistance in antiretroviral-naive patients, and non-controlled experiences (studies) suggest a possible use of lopinavir/r as a single-drug HAART (Pierone, Gathe).
The primary goal of the OK Study was to determine the feasibility of maintaining virological control with lopinavir/ritonavir monotherapy in patients who have had undetectable viral load for 6 months. 42 patients receiving lopinavir/r (Kaletra) + 2 NRTIs (or 1 NRTI + TDF) were randomized 1:1 to continue or to stop the NRTIs. Patients had to be on continuous ART during at least the prior 6 months; HIV viral load <50 copies/ml for at least 6 months prior to study entry; receiving Kaletra + NRTIs for 4 weeks; no history of virologic failure while receiving a PI; change of PIs for adverse events or other reasons is allowed if changes had been made while viral load was <50 copies/ml; HBsAg-negative.
Definition of loss of viral suppression per protocol analysis (ITT-MD=F):
2 viral loads >500 c/ml 2 weeks apart, or
change in randomized therapy, or
treatment discontinuation, or
lost to followup.
Baseline characteristics:
About 5 log HIV viral load; median number of months with <50 c/ml was 28 for patients who simplified and 15 for those who stayed on triple drug therapy but the poster said this difference was not significant; CD4 at baseline, about 600-650; CD4 nadir 139 in simplification arm & 90 in triple arm, not significant. 48% of patients in both arms had HCV. Patients were on LPV/r for 13 months. For 30% it was the first PI; for 47-67% it was 2nd PI.
CHANGE IN LIPIDS
Lipids on HAART before simplification:
total chol: 180;
LDL chol: 90;
HDL chol: 44;
trig: 190.
MEAN CHANGE IN FASTING SERUM LIPIDS AT WEEK 48 (mg/dL):
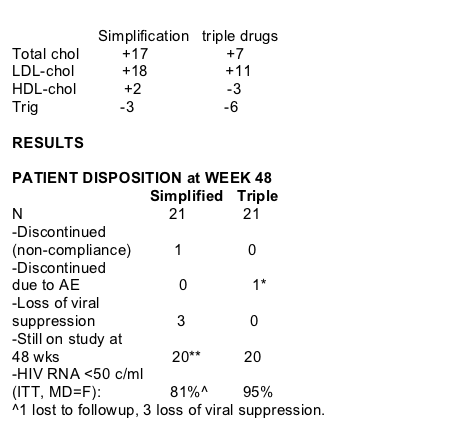
*hyperlipidemia associated with multiple cardiovascular risk factors and despite treatment with a lipid-lowering drug. At week 24 his fasting Total Cholesterol was 282, LDL-chol. 164, and serum TG 421.
**3 patients with loss of viral suppression are still actively followed.
|
|
| |
|
 |
 |
|
|