 |
 |
 |
| |
"Evaluation of Hyperpigmentation in HIV-Infected Patients Receiving
Emtricitabine (FTC, Emtriva)"
|
| |
| |
Reported by Jules Levin
B Rashbaum reported this poster at IAS-Rio (July 2005)
Capital Medical Associates, P.C., Washington, DC, USA
AUTHOR CONCLUSIONS
--In our patient population, the incidence of increased SP observed with FTC was uncommon (2%), and of low grade severity, and similar to that observed in pivotal Phase III trials
--SP was observed only in African American individuals and the incidence (8%) was similar to that seen in other studies
--There was no apparent relationship between SP and prior use of AZT
--No patient discontinued FTC treatment due to SP
INTRODUCTION
--Increased skin pigmentation (SP) has been associated with various
antiretroviral (ARV) agents, most notably zidovudine (AZT)
--FTC has been reported to be associated with SP, which appears after
weeks to months of therapy
--In clinical trials with FTC the lesions were reported to be asymptomatic
and usually Grade 1 in severity. No patient in any of the studies had
therapy discontinued due to SP.
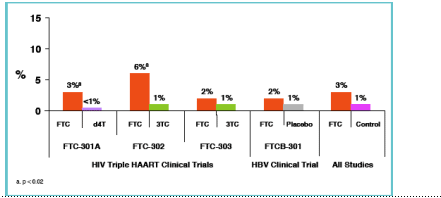
FTC-associated Skin Pigmentation (SP) in Phase 3 Clinical Trials1
--SP occurred mainly on the palms and soles
--Severity: Mild, 29/33 (88%); moderate, 4/33 (12%) of cases
--Caucasians < 1%, Hispanics 3%, Asians 4%, Blacks 8%
Incidence of Skin Discoloration in Phase 3 Trials
STUDY OBJECTIVE
To evaluate the frequency of development of increased skin
pigmentation and its clinical characteristics in patients taking
FTC 200 mg PO once-daily for HIV infection in an urban practice
METHODS
--Observational analysis
--100 HIV-positive infected males treated at Capital Medical Associates,
P.C., Washington, D.C.
--Patients were receiving FTC-containing HAART regimen for at least
3 months
--All patients were evaluated (data collection form) for any appearance of pigmentation
changes on palms, soles, oral mucosa, and other body parts
periodically over 12 months
RESULTS
--Baseline characteristics:
-Gender: all males
-Race: 67% Caucasian, 26% African American, and 7% of
Hispanic descent
-Mean (range) time on FTC: 11 (1.5 -18) months
-Prior AZT therapy: 54%
Overall findings:
-2 cases of hyperpigmentation were observed
-Both cases occurred in individuals of African American descent
Neither had a prior history of AZT use
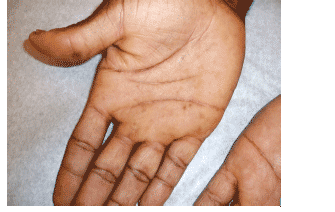
-Clinical manifestations:
--Patient #378: palms, soles and oral mucosa (Grade 2)
--Patient #929: soles only (Grade 1)
-Incidence of 2% overall; 8% in African American patients
-SP lesions were not found to progress in either patient even with
continued use of FTC over >6 months of follow-up
REFERENCE
1. Mondou E. et al. Incidence of Skin Discoloration Across Phase 3
Clinical Trials of Emtricitabine (FTC) in Adults, XV International AIDS
Conference, Bangkok,Thailand, July 11-16, 2004; Poster No. #5916.
|
| |
|
 |
 |
|
|