 |
 |
 |
| |
Tenofovir+FTC+Efavrienz vs Combivir+EFV:
48 Week Final Results Study 934
|
| |
| |
Reported by Jules Levin
Intl AIDS Society Conference
Rio de Janeiro
July 24-27, 2005
"Superior Outcome for Tenofovir DF, Emtricitabine and Efavirenz Compared to Fixed Dose Zidovudine/ Lamivudine and EFV in Antiretroviral Naive Patients"
AL Pozniak1, JE Gallant2,E DeJesus3, R Campo4, B Gazzard1, JR Arribas5,
B Lu6, D McColl6, J Enejosa6, and A Cheng6 for the Study 934 Team
1Chelsea and Westminster Hospital, London, UK;
2Johns Hopkins Univ School of Medicine, Baltimore, Maryland, USA;
3Orlando Immunology Center, Orlando, Florida, USA;
4Univ Miami, Miami, Florida, USA; 5Univ Hospital La Paz, Madrid, Spain;
6Gilead Sciences, Inc., Foster City, California, USA
Poznial presented in an oral talk at the final day of the meeting in Rio the final 48 week results from Study 934 which reports results of using tenofovir plus FTC compared to AZT/3TC, a standard nuke component of HAART. The primary study endpoint is 48 weeks. ART-naive patients were randomized to TDF/FTC/EFV, all once daily, or AZT/3TC (Combivir) twice daily plus EFV once daily. The fixed dose formulation of TDF/FTC was not used in the study, Pozniak said.
POZNIAK SUMMARY: superior overall response to FTC/TDF compared to Combivir arm. No patient developed K65R. Significantly more patients receiving Combivir discontinued due to adverse events (9% vs 4%). Limb fat was lower in patients receiving Combivir at week 48 (8.9 vs 6.8). Less lipid abnormalities (cholesterol, triglycerides) were experienced by patients receiving FTC/TDF compared to Combivir (AZT/3TC).
STATISTICAL ANALYSIS
Non inferiority Trial
Primary Endpoint < 400 c/mL at Week 48 -Time to Loss of Virologic
Response (TLOVR)
- FDA-required endpoint
- Similar to ITT Missing = Failure, Switch = Failure
- Requires confirmation for success
- Used by FDA for presentation in U.S. Prescribing Information of newly approved antiretrovirals
RESULTS
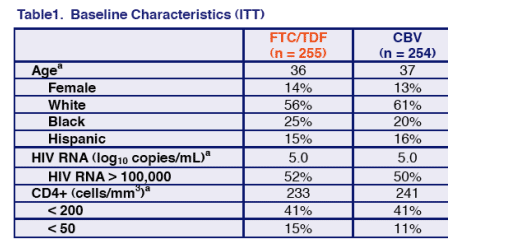
60% of study patients were white, 20-25% black & 15% Hispanic. 50% of patients had >100,000 copies/ml viral load. Average HIV RNA was 100,000 c/ml for patients. Average Cd4 count was 235, and 41% of patients had <200 CD4s & 11-15% had <50 CD4s. So the patients in the study were relativele advanced in the HIV stage of disease.
BASELINE NNRTI Resistance (ITT)
22 patients (11 FTC/TDF vs. 11 CBV) had NNRTI resistance before the study started
-Investigators notified if affected
-FDA recommended excluding these patients for Week 48 primary endpoint analysis (n = 487)
-Primary Efficacy Endpoint (HIV RNA < 400 c/mL) at Week 48 analyzed for both populations, excluding NNRTI-R (n = 487) and ITT
(n = 509)
84% of patients receiving TDF/FTC/EFV were responders vs 73% receiving Combivir/EFV (p=0.002).
Discounted due to adverse event: 4% receiving TDF/FTC/EFV vs 9% receiving Combivir/EFV.
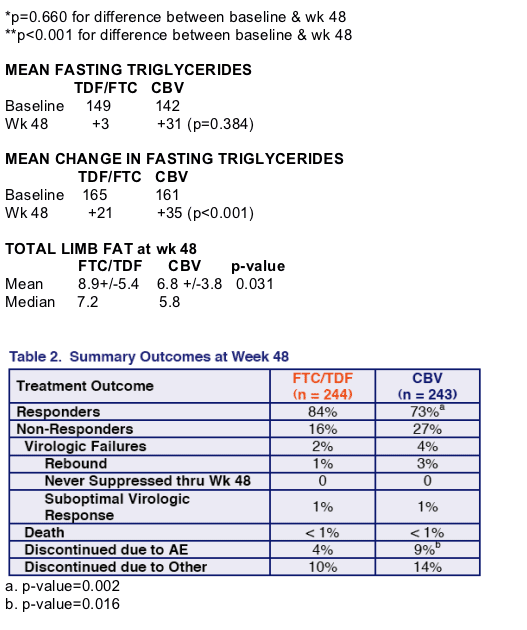
Proportion <400 c/ml (TLOVR) n=509: 81% receiving FTC/TDF vs 70% recxeiving Combivir (p=0.005) (95% CI +3.4%, +18.1%). If you exclude NNRTI resistant n=487: 84% vs 73% (p=0.002) (+4.3%, 18.6%).
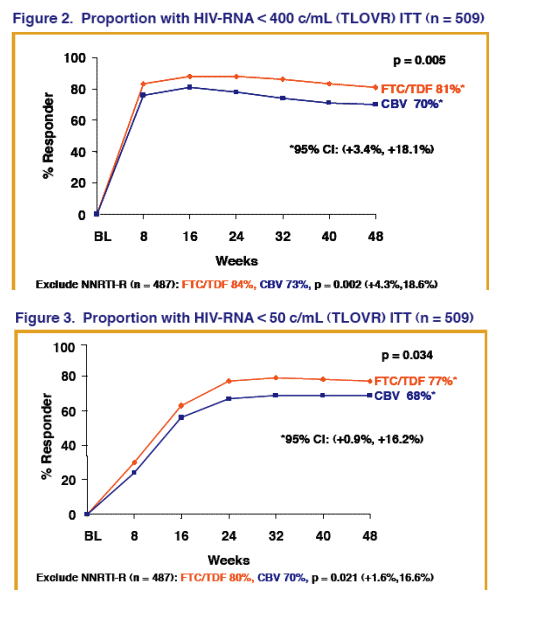
Proportion <50 c/ml HIV RNA (TLOVR) ITT n=509: 77% receiving FTC/TDF vs 68% for Combivir (p=0.034, 95% CI: +0.9%, +16.2%).
CD4 count increase at week 48 was +190 for FTC/TDF & 158 for Combivir.

|
|
| |
|
 |
 |
|
|