 |
 |
 |
| |
New Kaletra Tablet PK Study
|
| |
| |
Reported by Jules Levin
Intl AIDS Society Conf
Rio de Janeiro
July 24-27, 2005
"Significantly Reduced Food Effect and Pharmacokinetic Variability with a Novel Liponavir/Ritonavir Tablet Formulation"
There were several highlights from the Rio IAS Conference & these include the new Kaletra tablet formulation, final results from Study 934 of EFV/TDF/FTC, phase II data of the new NRTI for resistance Reverset, study results from phase II study of the new PI TMC114/r for drug resistance. Other highlights include new NNRTI & PI from GSK, new NNRTIs from Tibotec TMC125 & TMC 278. Boerhinger Ingleheim researchers presented a tipranavir study examining whether TDM is needed or useful in evaluating drug levels to see whether patient levels fall within the desired range & not too low or too high, and to see if hepatoxicity is related to drug levels. The study researchers concluded that most patients (80%) appear to achieve TPV levels within target levels, and the TPV/r concentrations achieved in most patients are not associated with increased risk of hepatoxicity. Interesting Reyataz studies include a switch study to unboosted Reyataz, and a retrospective review of ALT/AST from the Spanish Reyataz EAP. Further details of these studies & others will be reported. HIV entry inhibitors received quite a bit of attention at the conference. The GSK, Pfizer, and Schering CCR5 inhibitors all were highlighted. There was quite a bit of education regarding the mechanisms of action of these drugs and reviews of data so far collected, as well as the potential usefulness and difficulties for this new class of drugs. In all, there is quite a bit of ongoing research into finding new drugs for HIV as well as for hepatitis C.
The oral presentation at Rio was the first for the new Kaletra tablet and presented single-dose PK study results in healthy volunteers and compared to the currently used soft-gel capsule study results showed there will be (1) a reduced pill count, (2) no refrigeration needed,(3) lack of a significant food effect, (4) less PK variability, (5) less diarrhea compared to the soft-gel capsule.
At Rio, George Hanna from Abbott reported for the first time publicly regarding the new tablet formulation of Kaletra at an oral session on the last day Wednesday of the conference. The purpose of the tablet development program: To develop a LPV/r tablet formulation to reduce pill count, allow for room temperature storage and maintain bioavailability similar to the SGC formulation under fed conditions.
Hanna reported:
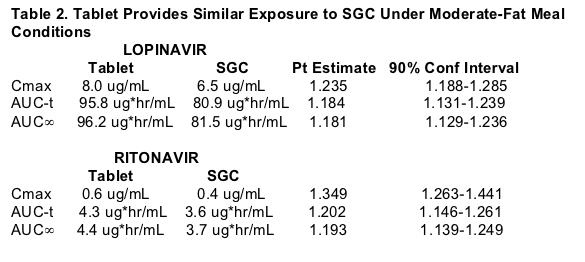
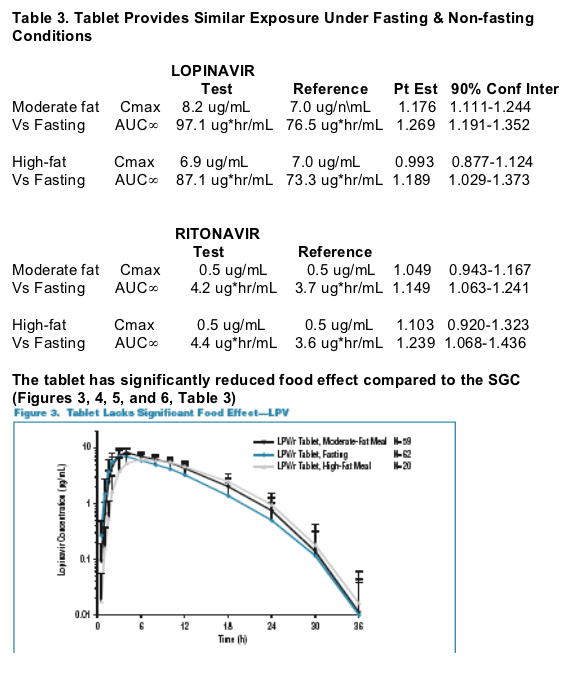
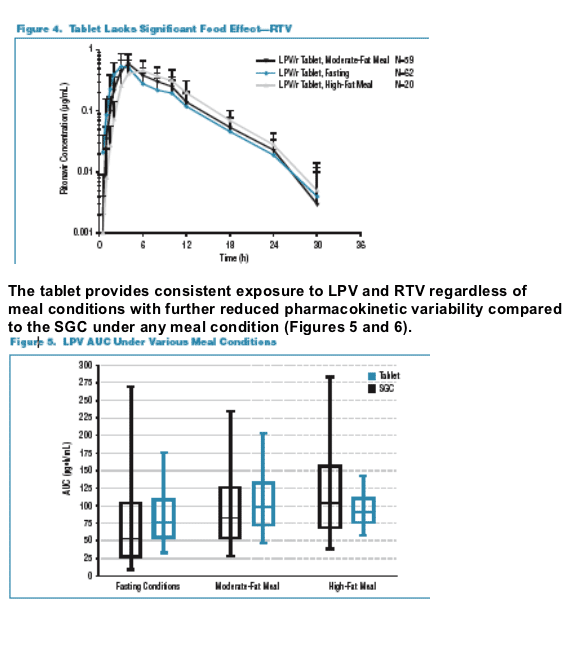
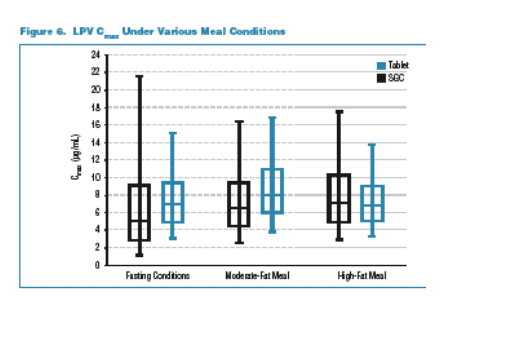
Following a single dose of LPV/r 400/100, the tablet provides similar exposure lopinavir and ritonavir exposures to the soft-gelatin capsule (SGC) under moderate fat meal conditions. The tablet met bioequivalence criteria with respect to lopinavir (LPV) and ritonavir (RTV) relative to the SGC at a dose of 400/100 under the moderate-fat meal conditions. The tablet lacks significant food effect for LPV & RTV. The tablet provides similar exposure under fasting and non-fasting conditions. The tablet provides consistent exposure to LPV and RTV regardless of meal conditions and the tablet has reduced pharmacokinetic (PK) variability compared to the SGC under any meal condition. 138 healthy adults received the tablet formulation, and the vast majority of adverse events were mild. No subject discontinued due to a drug related adverse event. The frequency of moderate or severe adverse events after a single-dose administration (400/100mg): diarrhea 0.3% for the tablet vs 1.1% for the SGC; any adverse event, moderate or severe 0.7% for tablet vs 1.1% for SGC. Hanna reported that multiple-dose administration of the tablet was similarly safe and well tolerated. In summary, Hanna concluded the benefits of the tablet formulation include:
1. reduced pill count
2. room temperature storage
3. lack of significant food effect
4. lower variability of lopinavir levels compared to the soft-gel capsule
5. less diarrhea and other adverse events.
The 90% confidence interval for the LPV AUC of the tablet to the SGC was within 0.80-1.25 and the Cmax extended slightly above 1.25 to 1.285. The tablet formulation shows an approximate 18% increase in LPV bioavailability compared to the SGC. Despite this modest increase in average bioavailability the tablet is expected to produce narrower ranges of concentrations (less PK variability) than the SGC with fewer patients likely to be exposed to high or low LPV concentrations. The tablet is expected to provide more consistent LPV and RTV levels day to day as meal conditions vary. The tablet formulation was safe and well tolerated and appeared to result in lower incidence of GI effects compared to the SGC in healthy subjects receiving single doses. In the Fall 2005 the results of study of multiple doses and study results from patients are expected to be presented at future meetings. The FDA has granted accelerated fast track designation for the Kaletra tablet.
BACKGROUND/INTRODUCTION
Kaletra is currently available as 133.3/33.3 (LPV/RTV) mg soft gelatin capsule (SGC) or 80/20 mg/mL liquid formulation, requires refrigerated storage prior to dispensing, and is recommended to be taken with food in order to optimize lopinavir exposure. A novel melt extrusion technology was used to reduce pill count from 6 SGC per day to 4 tablets per day. In addition, this 200/50 mg tablet formulation of LPV/r does not require refrigeration.
Melt Extrusion Technology (Meltrex)
--LPV/r is a low solubility/low permeability drug (Biopharmaceutics Classification System Class 4).
--Historically, solid formulations of LPV/r showed poor bioavailability.
-Unformulated solid fails to provide bioavailability (<5%).
-Incorporation of surfactants, acids or other wetting agents with traditional technologies failed to provide adequate bioavailability for solid formulations.
-In vitro dissolution did not necessarily correlate with in vivo bioavailability.
Melt extrusion technology (Meltrex) has overcome these challenges.
-Meltrex significantly improves the bioavailability of poorly soluble compounds like LPV/r by dissolving drug in polymer in a solvent-free environment.
The drug remains in a state of molecular dispersion as the polymer hardens.
-This extruded material can be shaped as tablets, granules, pellets, sheets, sticks, or powder which can be further processed into conventional tablets.
STUDY METHODS
The tablet was evaluated by one pilot single and multiple dose study and two studies using production scale lots. The single-dose PK, relative bioavailability of a LPV/r 400/100 mg tablet dose compared to SGC under moderate fat meal conditions since the SGC is recommended to be taken with food. In addition, the relative bioavailability of the LPV/r 400/100 as the tablet was assess under various meal conditions.
Healthy subjects (n=141 single dose, of whom 23 also received multiple dose) were enrolled intp these phase I, open-label, randomized, cross-over studies if they met these following criteria:
--general good health
--no concomitant medication
--body mass index 18 to 27 kg/m2
Subjects were equally randomized to each regimen sequence in each study.
Single doses were separated by at least 5 days. LPV/r was administered under fasting conditions, after a moderate fat breakfast (500-600 kCal, 20-30% from fat), or after a high fat breakfast (about 1000 kCal, 50% from fat).
Pharmacokinetic Analysis
Blood samples were collected for LPV and RTV assays as follows:
-Pre-dose (0 hour) and at 0.5, 1, 1.5, 2, 3, 4, 6, 8, 12, 18, 24, 30 and 36 hours following a single dose
Drug concentrations were measured by validated LC/MS/MS methods:
-LPV limit of quantitation (LOQ) = 5 ng/mL
-RTV LOQ = 1 ng/mL
LPV and RTV PK parameters were calculated with standard non-compartmental analysis using WINNONLIN v. 4.1 software (Pharsight Corp., Mountain View, CA) to estimate the maximum observed concentration (Cmax), area under the plasma concentration time curve (AUC) to the last measured concentration (AUCt) and to infinity (AUC°), and terminal phase half-life (t1/2).
Statistical Analysis
The bioavailability of the tablet relative to the SGC was assessed by a two one-sided test procedure via 90% confidence intervals obtained from the analysis of the natural logarithms of Cmax, AUCt and AUC° within the framework of the ANOVA model using the SAS system v. 6.12 software (SAS Institute, Cary, NC).
Safety Analysis
Safety and tolerability were assessed throughout the study based on reported adverse events, vital signs, electrocardiograms, and clinical laboratory measurements.
RESULTS
DEMOGRAPHICS of Subjects Receiving Single Doses of Tablet Formulation
Subjects: healthy adults
Sex: 105 males (76%), 33 females (24%)
Race/ethnicity: 99 whites (72%), 23 blacks (17%). 16 Hispanic (12%)
Age (yrs): 35 +/-11 (19-55)
Weight (KG): 77 +/-10 (55-101)
Height (cm): 176 +/-9 (147-192)
|
|
| |
|
 |
 |
|
|