 |
 |
 |
| |
Characterization of Anemia in HIV+ Subjects Treated with ART With and Without Zidovudine in 54 Clinical Trials
|
| |
| |
Reported by Jules Levin
IAS-Rio July 2005
Mark T. Edwards, Wayne Burkle, Amy Cutrell, Qiming Liao, Cindy Brothers and Jaime E. Hernandez
GlaxoSmithKline R&D, RTP, NC, USA
Poster Number TuFo0106
All GSK-sponsored clinical trials with at least 24 weeks of ART finished from
January 1995 to January 2004 and with an authorized database were analyzed.
......The incidence of grade 2/4 anemia in ART-naive subjects was similar in those receiving ZDV (1.5%) vs. those not receiving ZDV (1.1%). In ART-experienced subjects, the incidence of anemia was higher, (p<0.01), in those receiving ZDV (1.8%) vs. those not receiving ZDV (0.6%). Severe anemia (grade
3/4) was seen in 1.0% of ART-naïve and in 0.9% of ART-experienced subjects
taking ZDV vs. 0.6% and 0.3% of subjects not taking ZDV....several risk factors were identified that could be useful when considering ZDV-containing HAART....
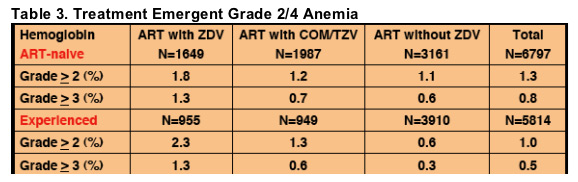
Table 3. Treatment Emergent Grade 2/4 Anemia
AUTHOR CONCLUSIONS
--The overall incidence of anemia in this cohort was low (1%).
--Subjects treated with ZDV had a higher incidence of anemia compared to subjects not treated with ZDV.
--This incidence, however, was also low and appeared to be lower when COM/TZV were used.
--Multivariable logistic regression models selected ZDV use, high baseline viral load, and male gender as determinants of increased risk.
--As expected, higher hemoglobin values at baseline generally suggest a decreased risk of developing anemia.
INTRODUCTION
Hematologic abnormalities have been described with HIV infection. The most common abnormality is anemia, which has been associated with advanced HIV disease and reported in 40% of untreated subjects and in 60 to 95% of treated
subjects (1-5). Anemia has also been identified as a predictor of decreased HIV
disease survival (6). Although HAART has been reported to improve HIV-related
anemia, this complication remains relatively common and is associated with
fatigue and decreased quality of life; with some studies showing improvement of
these parameters after appropriate treatment.
Hematologic toxicity (mainly anemia and neutropenia) has been reported in HIV+ subjects, including those treated with ZDV. This ZDV-associated toxicity may be
dose related. Approximately 3% of subjects have reported moderate to severe
anemia (DAIDS 1992 grades 2 to 4) at the currently approved dose. To better
characterize the incidence, severity and determinants of anemia in patients
treated with HAART, we performed an analysis of data from 54 trials including
more than 12,000 subjects treated with ART with and without ZDV.
METHODS
All GSK-sponsored clinical trials with at least 24 weeks of ART finished from
January 1995 to January 2004 and with an authorized database were analyzed.
Anemia was identified by hemoglobin (Hb) levels below normal, and classified
with the DAIDS 1992 scale.
Only subjects with hemoglobin data and a treatment start date were included in
this investigation. Descriptive statistics were summarized for naïve and
experienced subjects treated with and without ZDV.
A retrospective analysis of risk factors for anemia in HIV clinical trials was also
conducted. Univariate logistic regression models were fit to understand the
individual predictive ability of each potential risk factor. Clinical characteristics
used as potential risk factors were: BL demographics, BL CDC Class, BL
plasma HIV-1 RNA, BL CD4+ cell count, presence/absence of ZDV in the
regimen, BL hemoglobin, BL weight, BL ART status (ART-naïve or ARTexperienced), and geographic region (US, Europe, Latin America, or rest of
world [ROW]). Post BL hemoglobin results were also classified using the
DAIDS 1992 scale; anemia was defined as a hemoglobin value that resulted in
a grade 2 or higher classification. Two multivariable logistic regression models
were finally constructed using stepwise selection.
RESULTS
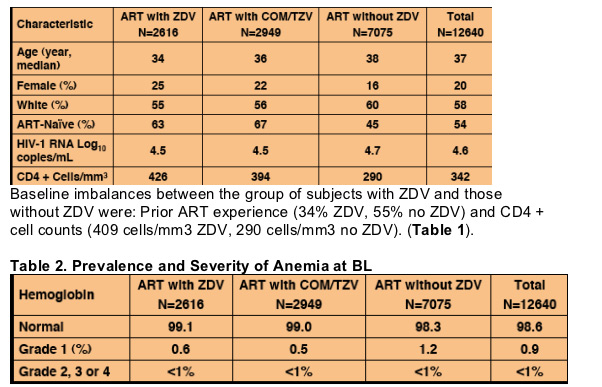
Baseline demographics and characteristics were comparable between subjects
treated with (5565 subjects) and without (7075 subjects) ZDV. 65% and 45%
of subjects respectively were ART-naïve at BL (Tables 1, 2).
Table 1. Baseline Demographics and Characteristics
The incidence of grade 2/4 anemia in ART-naive subjects was similar in those
receiving ZDV (1.5%, 95%CI [1.1%,1.9%]) vs. those not receiving ZDV (1.1%,
95%CI [0.7%,1.5%]). In ART-experienced subjects, the incidence of anemia
was higher, (p<0.01), in those receiving ZDV (1.8%, 95%CI [1.2%,2.4%]) vs.
those not receiving ZDV (0.6%, 95%CI [0.4%,0.9%]). Severe anemia (grade
3/4) was seen in 1.0% of ART-naïve and in 0.9% of ART-experienced subjects
taking ZDV vs. 0.6% and 0.3% of subjects not taking ZDV (Table 3).
When subjects receiving ZDV were classified as receiving ZDV as a separate
drug vs. as part of a fixed combination tablet (Combivir [COM, ZDV 300mg/
Lamivudine 150mg] or Trizivir [TZV, ABC 300mg/ Lamivudine 150mg/ZDV
300mg]) incidences of anemia were 2.0% (95%CI [1.5%,2.5%]) for ZDV and
1.2% (95%CI [0.8%,1.6%]) for COM/TZV (Table 3).
Among the 12,640 subjects with hemoglobin data and treatment information,
80% were male, 99% had normal hemoglobin values at baseline, and 46%
were ART-experienced (Tables 1 and 2). Global distribution was 49% US,
19% Europe, 3% Latin America, 30% ROW.
There were 147 (1.2%) cases of post baseline grade 2-4 anemia as
determined by the DAIDS 1992 scale; 12493 subjects served as controls.
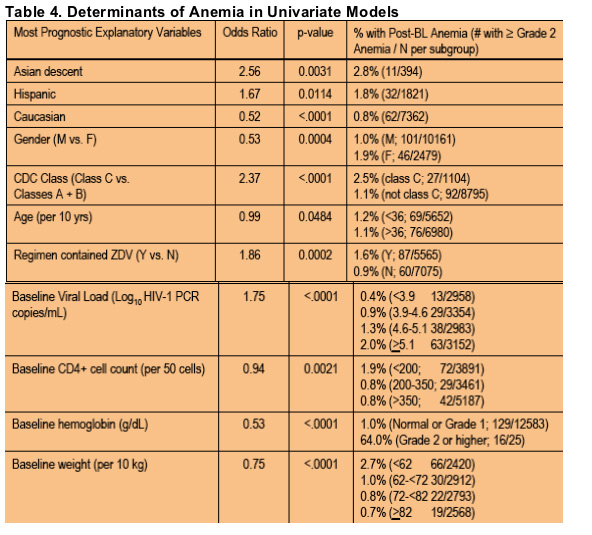
The results of the univariate logistic regression analysis are shown in Table 4
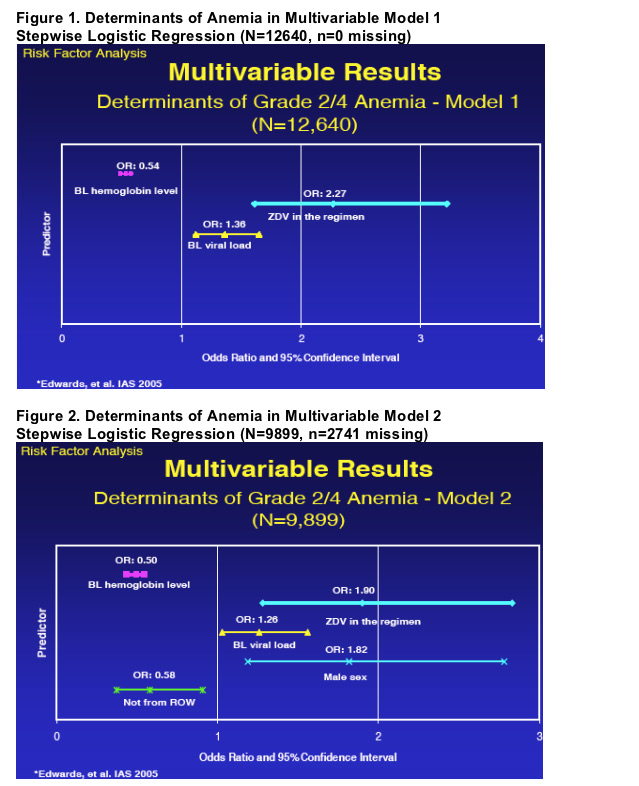
while the results of the 2 multivariable models are shown in Figures 1 and 2.
AUTHOR DISCUSSION
Despite significant advances in the treatment of HIV infection, the need for drugs with better long term tolerability and toxicity has been identified. Although effective HAART has been associated with improvement of hematologic complications including anemia, this toxicity remains relatively common. Therefore, comparative safety data in patients treated with HAART with and without ZDV and identification of risk factors for the development of anemia are particularly pertinent.
In this analysis using data from more than 12,000 patients participating in 54 controlled clinical trials and with varied characteristics, we were able to demonstrate a low prevalence of anemia at BL and a relatively low incidence of HAART-associated anemia. Although the risk of developing anemia was higher for subjects receiving ZDV-containing HAART, this incidence was also low and appeared lower when COM or TZV were used. Reasons for this finding are not known but may include variability in the BL characteristics that were identified as risk factors for the development of anemia.
To identify potential risk factors for the development of anemia in this large cohort, two multivariable models correcting for confounders were constructed. The first model was designed to maximize the number of subjects in the model while the second maximized the number of variables for consideration in the model. Interestingly, both models identified the presence of anemia and a higher viral load at BL as risk factors for subsequent development of anemia regardless of the use of ZDV or not. Ssali et al, recently reported risk factors for the development of severe anemia after ZDV treatment in patients in Africa (7) and also describe BL Hb levels as a determinant; one of our multivariable models identified sex as a potential risk factor but unlike the DART analysis, the risk was higher for males in our cohort. This may be explained by another finding in the same model, namely that subjects from areas similar to those analyzed in DART had a higher risk of anemia vs. other geographic areas.
In conclusion, the vast majority of patients treated with HAART +/- ZDV in this cohort did not develop anemia; several risk factors were identified that could be useful when considering ZDV-containing HAART.
|
| |
|
 |
 |
|
|