 |
 |
 |
| |
Switch to Reyataz: lipids, viral load
|
| |
| |
"Efficacy of Atazanavir (ATV) Based HAART in Patients Switched From a Stable PI or Boosted PI (PI/r) Treatment. Planned Week 24 Analysis of a Phase IIIb 48-Week Multicenter, Open-label, Randomized, Prospective Study: The SWAN Study"
J. Gatell1, D. Salmon-Ceron2, A. Lazzarin3, E. Van Wijngaerden4, F. Antunes5, C. Leen6, A. Horban7, V. Wirtz8, L. Odeshoo8, C. Gruber9 and E. Ledesma8 for the SWAN Study Group
1Hospital Clinic i Provincial, Barcelona, ES, 2Group Hospitalier Cochin, Paris, FR, 3Ospedale San Raffaele, Milano, IT, 4Univ Ziekenhuizen Leuven, Leuven, BE,
5Hospital de Santa Maria, Lisboa, PO, 6Western General Hospital, Edinburgh, UK, 7Wojewodzki Szpital Zakany, Warsaw, PL,
8Bristol-Myers Squibb, Pharmaceutical Research Institute, Wallingford, CT, USA, 9Bristol-Myers Squibb, Pharmaceutical Research Institute, Braine l'Alleud, BE
Reported by Jules Levin
IAS Conference
Rio de Janeiro
July 24-27, 2005
SUMMARY:
In a study population with significant ARV experience, but no known previous PI virologic failures, a switch to ATV (Reyataz) from a stable PI regimen demonstrated a statistically significant reduction in the risk of Viral Rebound at week 24 (3% vs 8%). The other study patients continued on current regimen.
Fifty-five percent of subjects in the ATV arm switched from a RTV-boosted PI at study entry, with 36% of all subjects switching from LPV/RTV. Of note patients who switched received unboosted 400 mg Reyataz unless they were also taking tenofovir in which case boosted (300/100) Reyataz was used. According to poster 8% of patients were taking tenofovir in the previous regimen prior to the study.
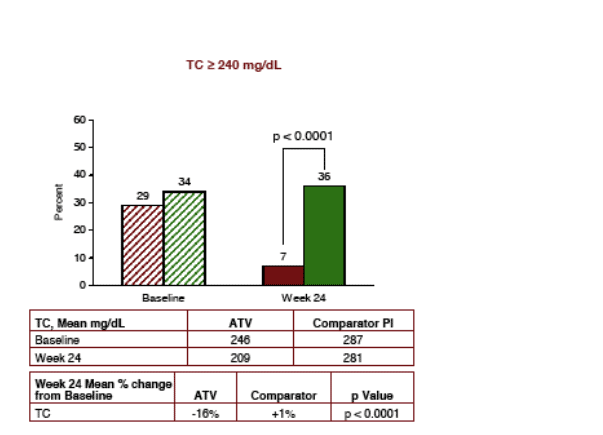
LIPIDS: Total Cholesterol declined by 16% after switch to ATV after 24 weeks, LDL-C declined by 16%, triglycerides declined by 38%, and there were benefits regarding HDL-C (27% on Reyataz vs 41% on CPI had <40 HDL-C), and there were benefits regarding Total/HDL-C Ratio (78% >3 or more for Reyataz vs 91% for Comparator PI, and LDL/HDL Ratio of >3 or more (30% for Reyataz vs 61% for comparator PI). All these differences were statistically significant.
On-treatment emergent AEs of any grade related to gastrointestinal toxicity (diarrhea, nausea, vomiting or abdominal pain) were higher on the Comparator PI arm than in subjects switching to ATV: 15/133 (11%) in the Comparator PI arm vs 17/274 (6%) in the ATV arm (p = NS).
No relevant differences were observed for SAEs (other than deaths) or AEs leading to discontinuation. Disct rate was 6-8% in study so far.
Hyperbilirubinemia was frequent in the ATV arm, 1% of subjects discontinued due to ocular icterus or jaundice
ALT and AST elevations were infrequent in those switching to ATV and were comparable to the control arm (4-5%), also within those with HBV/HCV co-infection (11-14%).
The use of lipid-lowering agents was 8-9% at baseline and 8% for Reyataz & 13% for comparator PI group at week 24, but difference was not statistically significant. The use of anti-diarrheal agents for subjects who switched to ATV was significantly lower than for the Comparator PI arm (2% vs 7%, p < 0.05).
STUDY DETAILS
Patients with <50 HIV RNA viral load were switched to Reyataz once daily regimen or remained on the same regimen and followed for 48 weeks in this study. This is a 24 week interim analysis. 278 patients received Reyataz. Of note, patients received unboosted Reyataz 400 mg once daily and only received Reyataz boosted by 100mg of ritonavir when tenofovir was also used. 30% of patients had AIDS, baseline median CD4 was 490, baseline HIV RNA 1.69 log copies/ml (<50 c/ml). 70% of patients had 1 or more baseline cardiovascular risk factors. 417 patients were randomized in this study, 274 to Reyataz and 133 to remaining on their current regimen.
--Years of prior PI therapy: 3.4
--38% of patients were on Kaletra.
--Years on prior NRTI therapy: 3.8
--8% of patients were on tenofovir at baseline.
--Years on prior NNRTI therapy: 1.3; none were on NNRTIs at baseline.
--Mean time on study therapy at this interim analysis: 29 weeks
--More subjects in the Comparator PI arm discontinued before starting treatment after learning their random assignment to continue unmodified therapy
RESULTS
The efficacy analysis at week 24 demonstrated that the proportion of subjects with Viral Rebound at week 24 was significantly lower in the ATV group (3%; 8/278) than in the Comparator PI group (8%; 11/141) (pairwise difference ATV- Comparator PI, -4.9; 95% CI: -9.1, -0.7; p < 0.05). Viral Rebound = 2 consecutive HIV RNA levels (or last one only before discontinuation) ≥ 50 copies/mL.
Hazard ratios (95% CI) for Viral Rebound [ATV:Comparator PI] in the unboosted PI and boosted PI strata were 0.21 (0.05, 0.81) and 0.73 (0.21, 2.59), respectively.
Changes in CD4 counts were similar across treatment groups with a mean change of +12 cells/mm3 in the ATV arm and +15 cells/mm3 in the Comparator PI arm.
LIPIDS
For subjects initiating lipid-lowering therapy, subsequent lipid data were censored from the lipid analyses
Subjects in the ATV arm experienced significant reduction of all measured atherogenic lipid fractions through week 24; the difference in mean percent change from baseline for non-HDL-C was -22% in the ATV arm vs 0% in the Comparator PI arm (p<0.0001).
The percentage of subjects on lipid-lowering therapy prior to the study was comparable between the ATV and Comparator PI arms (8% vs 9%, respectively); however at week 24 the use of lipid-lowering agents was lower in subjects switching to ATV (8% vs 13%, p = NS)
Median fasting glucose levels were comparable between the ATV and Comparator PI groups at baseline (89 mg/dL in the ATV group and 87 mg/dL in the Comparator PI group). No change in mean fasting glucose was observed for those who switched to ATV or remaining on unmodified therapy at week 24.
The bar with red stripes was the baseline total cholesterol for the patients who remained on the comparator PI group and the green stripes was the bar reflecting baseline total cholesterol for patients who switched to Reyataz. The green solid bar was the % of patients on the comparator Pi who had >240 total cholesterol after 24 weeks and the red solid bar was the % of patients with >240 total cholesterol (the level set by NCEP Guidelines as high for Total Cholesterol) who switched to Reyataz (7% for Reaytaz vs 36% for comparator PI (P<0.0001).
TOTAL CHOLESTEROL
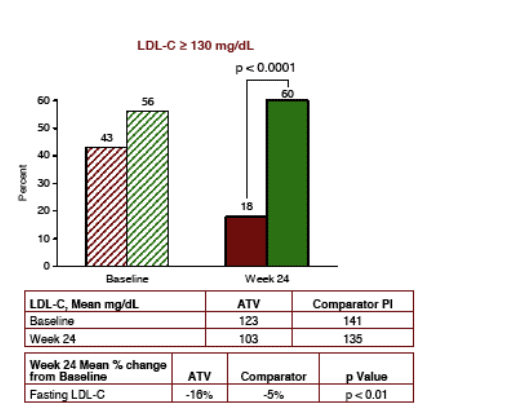
LDL Cholesterol declined for patients switching to Reyataz by 16% vs a 5% decline for patients remaining on comparator PI (p<0.01). 60% of patients staying on comparator arm vs 18% who switched to reyataz had LDL-C above the NCEP Guidelines for high LDL-C (p<0.0001).
LDL-CHOLESTEROL
TRIGLYCERIDES
After 24 weeks, 27% of patients remaining on compatator PI had high triglycerides >250 (NCEP Guidelines) compared to 8% who switched to Reyataz (p<0.001). TG declined from mean of 204 to 125 in Reyataz group and was 201 for comparator group at baseline and 191 at week 24. TG declined by 38% for Reyataz group and increased by 1% for comparator PI (p<0.0001).
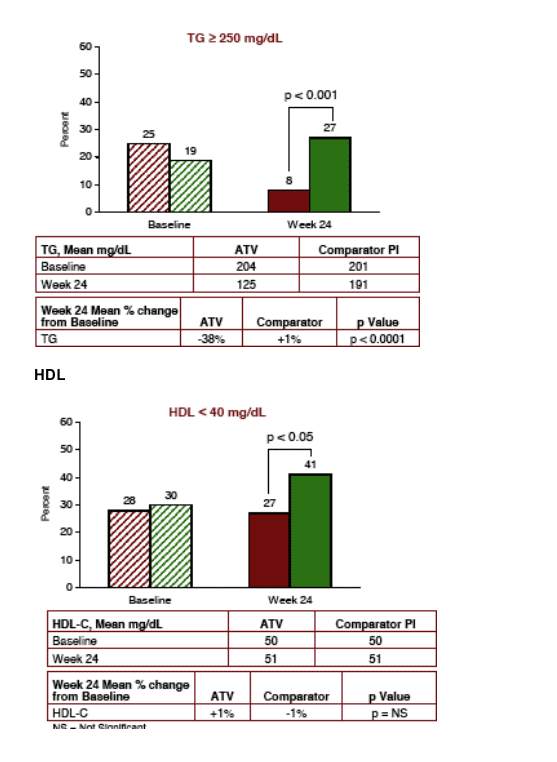
HDL
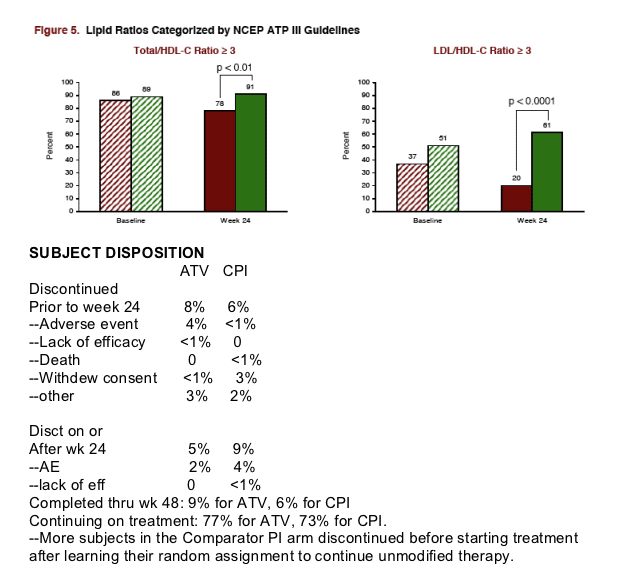
Continuing on treatment: 77% for ATV, 73% for CPI.
--More subjects in the Comparator PI arm discontinued before starting treatment after learning their random assignment to continue unmodified therapy.
To account for the higher post-randomization pre-treatment dropout rate for those subjects assigned to continue on unmodified therapy, two sensitivity analyses were performed for the primary endpoint. The first analysis assumed no VReb for subjects leaving the Comparator PI arm but not for subjects in the ATV arm. The second analysis was conducted on the treated (as opposed to randomized) population. Both analyses confirmed the efficacy of the switch to ATV.
SAFETY
The switch to ATV (vs Comparator PI) did not lead to an increase in the discontinuation rates due to AEs (6% vs 5%), or the incidence of SAEs (8% vs 6%) or deaths (0% vs 4%).
On-treatment emergent AEs of any grade related to gastrointestinal toxicity (diarrhea, nausea, vomiting or abdominal pain) were higher on the Comparator PI arm than in subjects switching to ATV: 15/133 (11%) in the Comparator PI arm vs 17/274 (6%) in the ATV arm (p = NS).
The use of anti-diarrheal agents for subjects who switched to ATV was significantly lower than for the Comparator PI arm (2% vs 7%, p < 0.05).
Grade 3-4 ALT or AST elevations occurred with similar frequency in both arms (4-5% for ALT, 2% for AST). A higher incidence of ALT and AST elevations occurred in co-infected subjects, with similar frequency across treatment groups 14% for Reyataz group vs 11% for comparator PI group. For AST 6% vs 8%.
Grade 3-4 elevations in total bilirubin were reported in 38% of the subjects on ATV and in 2% of the subjects on the Comparator PI. Elevations in total bilirubin for subjects with ALT or AST elevations were experienced by 3% of subjects in the ATV arm and no subjects in the Comparator PI arm
Jaundice or ocular icterus of any grade (27/274 [10%]) was reported for subjects on ATV, with only 3 cases (1%) of grade 3-4 jaundice or ocular icterus. One percent of subjects in the ATV arm discontinued due to ocular icterus or jaundice. Ocular iceterus: 3% for Reyataz group vs 0 for Comparator PI. Jaundice: 2% for Reyataz vs 0 for CPI group. Diarrhea was 0 for Reytaz vs 2% for CPI group.
BACKGROUND
Atazanavir (ATV) is a potent, well-tolerated, once-daily (QD) HIV protease inhibitor (PI) extensively studied in naive and experienced patients
In a Phase IIIB trial (AI424-044), virologic suppression was maintained or improved after switching patients with low viral load from the PI nelfinavir to QD ATV. In addition, improvement in lipid parameters (total cholesterol [TC], fasting low-density lipoprotein cholesterol [LDL-C], fasting triglycerides [TG]) was demonstrated in clinical trials and cohort studies for the patients switched to ATV-containing HAART. Similar findings were observed in trial AI424-067, where patients with hyperlipidemia but virologically controlled had the PI component in a variety of non-ATV containing PI-based regimens switched to ATV.
Limitations of other PI-based regimens include a higher frequency of gastrointestinal side effects, dyslipidemia, greater pill burden and the potential for increased polypharmacy due to the frequent necessity of concomitant anti-diarrheal or lipid lowering Medications.
Regimen simplification may lead to improved adherence, a reduced risk of antiretroviral-resistance, and less potential for drug interactions
The current trial (AI424-097) was designed to evaluate the efficacy and safety of a switch to ATV (or ATV/ritonavir [RTV] if in association with tenofovir disoproxil fumarate [TDF]) in HIV-infected patients experiencing virologic suppression but seeking regimen simplification.
STUDY OBJECTIVES
Primary: to Compare the proportion of subjects with virologic rebound (VReb) ≥ 50 copies/mL through 48 weeks between subjects who switch to an ATV-containing HAART regimen and those who remain on an unmodified PI-containing HAART regimen
Secondary
- Time to VReb for subjects with HIV RNA < 50 copies/mL at baseline
- Magnitude of change from baseline in CD4 count
- Frequency and severity of all clinical and laboratory adverse events (AEs), and discontinuations for AEs
- Percentage of subjects with viral load > 1,000 copies/mL
- Changes from baseline in fasting lipids
TC
LDL-C
HDL Cholesterol (HDL-C)
Non-HDL Cholesterol (non-HDL-C)
TG
Changes in fasting glucose
STUDY DESIGN
Screening Criteria
--Stable PI-containing regimen with or without RTV
--First or second HAART treatment
--No previous virological failure while on a PI-based HAART
--Current PI component at least BID and/or ≥3 pills/day
--Suppressed viral load (< 50 c/ml)
Patients switched to Reyataz or continued their regimen for 48 weeks with a 24-week interim analysis.
Comparator PI(s) (with or without RTV at a dose of 200 mg or less) + backbone
ATV 400 mg QD + unchanged backbone. Only when TDF was part of the backbone, ATV/RTV 300/100 mg QD was used.
Statistical Analyses
The study was designed with 90% power to demonstrate non-inferior rates of VReb between the two regimens for the primary analysis, comparing the proportions of subjects with VReb at week 48. The ATV regimen was declared to be noninferior to the Comparator PI regimen if the upper limit of the 95% CI for the difference in proportions was < 12%. A minimum of 372 subjects randomized in a 2:1 ratio provided at least 90% power to demonstrate non-inferiority for the
primary analysis. The present report includes results of the planned week 24 interim analysis
Study population was stratified by use of boosted vs unboosted PI at randomization.
Efficacy and lipid analyses included data through 24 weeks for all randomized subjects. Mean percent changes from baseline and standard errors for lipid parameters were computed on the log scale and back transformed.
Safety analyses included all data available through time of analysis for all treated subjects until last patient reached 24 weeks. Serious AEs (SAEs) and deaths were included without regard to treatment status at time of onset for enrolled
subjects
|
|
| |
|
 |
 |
|
|