 |
 |
 |
| |
In vitro antiviral effects of combinations of Abbott HCV polymerase inhibitors with IFN or NS3/4A protease inhibitors
|
| |
| |
Reported by Jules Levin
AASLD, Oct 27-31, 2006, Boston, MA
Gennadiy Koev, T. Dekhtyar, L. Han, P. Yan, J. Beyer, T. Ng, T. Lin, D. Larson, T. Bosse, H-J. Chen, K. McDaniel, L. Klein, R. Wagner, W. Kati, D. Kempf, H. Mo, and A. Molla. Antiviral Research, Global Pharmaceutical Research and Development, Abbott Laboratories, Abbott Park, IL, USA
INTRODUCTION
HCV is currently one of the major health care problems in the world. In search of an alternative to the standard IFN-RBV treatment of HCV infection, we have discovered a series of potent inhibitors of the HCV NS5B RNA polymerase. A-837093 and A-848837 are two representative compounds from this series. While monotherapy often leads to resistance development (See posters 406, 432 talk 128), drug combinations have proven to deliver superior results in HIV therapy, and are almost certain to be most effective in treatment of HCV (Koev et al., 2006, Mo et al., 2005). In this study, we examined the short-term and long-term antiviral activity of Abbott HCV polymerase inhibitors A-837093 and A-848837 alone and in combination with IFN and the NS3 protease inhibitors BILN 2061 and SCH 503034. Our results indicate a great potential for successful combinations of our polymerase inhibitors with other drug classes.
AUTHOR CONCLUSIONS
- The HCV polymerase inhibitor exhibited additive and synergistic interactions with the protease inhibitor BILN 2061 and IFN in vitro.
- Long-term treatment of 1b-N replicon with either HCV polymerase inhibitors (A-837093, A-848837) or protease inhibitors (BILN 2061, SCH 503034) alone resulted in multi-log reductions of replicon RNA levels but did not clear RNA due to development of resistance mutations.
- Long-term treatments of 1b-N replicon cells with A-837093 or A-848837 in combination with IFN exhibited an enhanced antiviral effect, compared to single compound treatments.
- Long-term combinations of A-837093 or A-848837 with the HCV protease inhibitors resulted in complete elimination of HCV replicon RNA within 12 days.
- Our findings suggest that Abbott benzothiadiazine inhibitors of HCV RNA polymerase in combination with drugs of different classes, are likely to be effective in treating HCV infection in vivo.
METHODS
- The antiviral compounds were synthesized in-house at Abbott Laboratories. Their structures and anti-replicon activities are presented in Fig.1.
- The 1b-N subgenomic replicon containing the SEAP reporter (Ikeda et al., 2002; Yi et al., 2002) was obtained from Dr. Stanley Lemon (UTMB, Galveston, TX). Replicon-containing cells were maintained in DMEM supplemented with 10% FBS and Pen/Strep solution.
- The antiviral activity of HCV inhibitors was determined by monitoring the levels of HCV RNA and/or SEAP activity in HCV replicon cells.
- In the drug combination assay, each compound was serially diluted two-fold, and six dilutions of one compound were combined with six dilutions of another compound in a checkerboard manner. Drug interactions were analyzed using Loewe additivity and Bliss independence models (Greco et al., 1995, Thisted, 1998).
- For the long-term replicon clearance assay, replicon cells (106 cells) were plated in a T75 flask in 15 ml of supplemented DMEM without G418. Compounds were added to specified concentrations, and the cells were grown to -95% confluency (4 days). At each passage, cells were trypsinized, 106 cells were frozen in 350 ml of RLT RNA lysis buffer (QIAGEN), and additional 106 cells were passed into another T75 flask with fresh media and inhibitors
- RNA was extracted from 106 cells using QIAGEN RNeasy spin kit. Total RNA was eluted in 50 ml of nuclease-free water. RNA representing 105 cells (5 ml) was analyzed in a Real-Time RT PCR assay (Applied Biosystems). The NS3 and NS5B coding regions were amplified and sequenced.
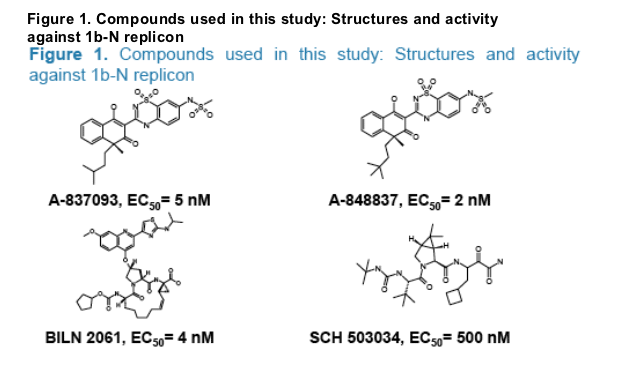
RESULTS
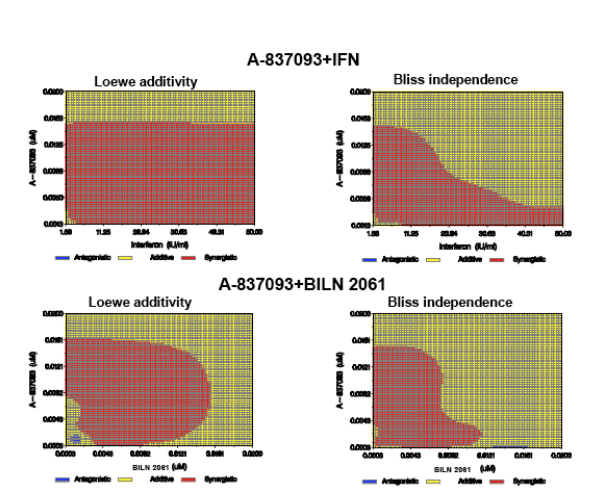
1. To assess the nature of drug interactions in the replicon, combinations of A-837093 with either BILN 2061or IFN were tested over a range of concentrations near their EC50, in a checkerboard fashion.
Statistical analysis of the inhibition data showed addtitive to synergistic interactions at the majority of concentrations tested between A-837093 and IFN as well as A-837093 and BILN 2061 (Fig. 2).
Figure 2. Additive and synergistic interactions of A-837093 with IFN and BILN 2061. Red areas represent synergy, yellow - additivity, and blue - antagonism
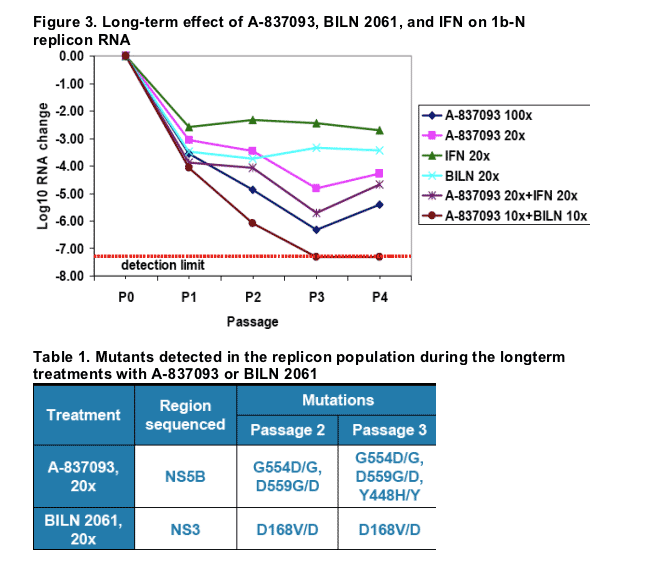
2. To determine the long-term effect of the compounds on the replication of HCV replicon, A-837093, BILN 2061, and IFN were tested in a long-term replicon clearance assay, alone (20xEC50) and in combinations (20xEC50+20xEC50 and 10xEC50+10xEC50).
None of the compounds alone resulted in a complete elimination of the replicon RNA due to development of resistant mutants (Table 1). The combination of A-837093 with BILN 2061 resulted in undetectable HCV RNA within 12 days of treatment.
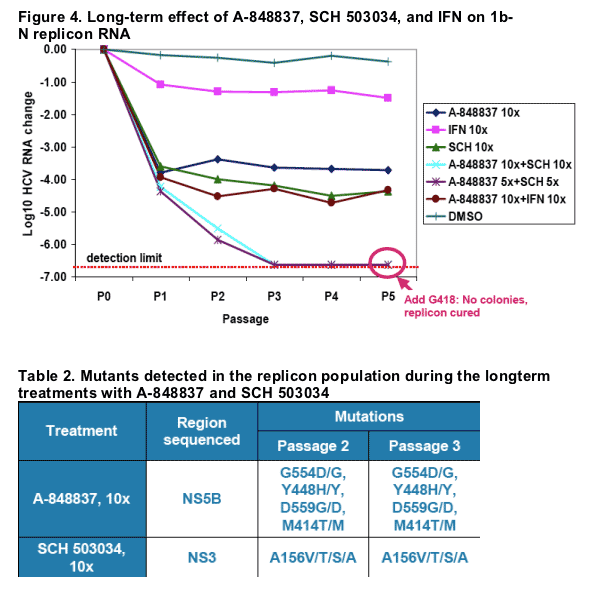
3. To determine the long-term effect of A-848837, SCH 503034, and IFN on the HCV replicon, the compounds were tested in a long-term replicon clearance assay, alone and in combinations. As in the previous experiment, none of the compounds alone resulted in a complete elimination of the replicon RNA due to development of resistant mutants (Table 2). The combination of A-848837 with IFN showed inhibition effect greater than that achieved by either drug alone. The
combination of A-848837 with SCH 503034 resulted in undetectable HCV RNA within 12 days of treatment. After passage 5, cells were grown in the presence of G418 (0.2 mg/m) for two weeks. No colonies were observed in cells treated with the combinations of A-848837 and SCH503034, reconfirming clearance of replicon RNA by the combination treatments.
REFERENCES
Greco W.R., G. Bravo, J.C. Parsons. 1995. The search for synergy: a critical review from a response surface perspective. Pharmacol Rev 47(2): 331-85.
Ikeda M, Yi M, Li K, Lemon SM, 2002. Selectable subgenomic and genome-length dicistronic RNAs derived from an infectious molecular clone of the HCV-N strain of hepatitis C virus replicate efficiently in cultured Huh7 cells. J Virol 76(6): 2997-3006.
Koev, G., T. Dekhtyar, L. Han, P. Yan, T. I. Ng, C. T. Lin, H. Mo and A. Molla. 2006. Antiviral interactions of an HCV polymerase inhibitor with an HCV protease inhibitor or interferon in vitro. Antiviral Res. (In press).
Mo H, Lu L, Pilot-Matias T, Pithawalla R, Mondal R, Masse S, Dekhtyar T, Ng T, Koev G, Stoll V, Stewart KD, Pratt J, Donner P, Rockway T, Maring C, Molla A., 2005. Mutations conferring resistance to a hepatitis C virus (HCV) RNA-dependent RNA polymerase inhibitor alone or in combination with an HCV serine protease inhibitor in vitro. Antimicrob Agents Chemother 49(10): 4305-14.
Thisted, R. A. 1988. Elements of Statistical Computing: Numerical Computation. London, Chapman & Hall.
|
| |
|
 |
 |
|
|