 |
 |
 |
| |
Valopicitabine (NM283) plus Peg-Interferon in Treatment-Naive Hepatitis C Patients with HCV Genotype-1 Infection: HCV RNA Clearance During 24 Weeks of Treatment
|
| |
| |
E Lawitz, T Nguyen, Z Younes, J Santoro, N Gitlin, D McEniry, R Chasen, J Goff, S Knox, K Kleber, B Belanger, N Brown, D Dieterich, and the 006 Investigator Group
Doug Dieterich reported the NM283 study results at AASLD in the Monday morning 8am Presidential Plenary.
NM283 was ahead of all new HCV drugs in development but development was delayed due to adverse events, see Protocol Amedment below. The FDA requested data on a lower dose to see if this resolved the adverse events. This presentation at AASLD reports the results at the lower dose of 200 mg compared to the higher dose used previously, 800 mg. Immediately below is the summary of viral load responses and the safety information reported, but they did not report a breakdown on the percent of patients with specific adverse events such as percent with diarrhea, GI side effects, and elevations in lipase/amylase.
The authors concluded:
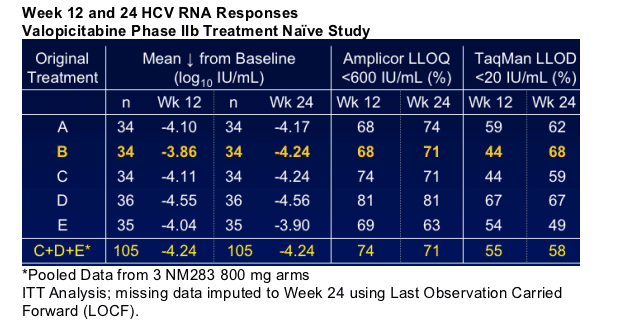
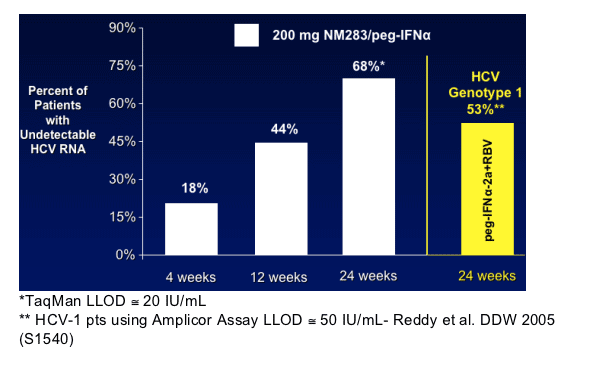
--By Week 24 the number of patients with HCV RNA < 20 IU/mL is similar across treatment arms.
--200 mg valopicitabine/peg-IFN_ compares favorably with historical results for peg-IFN_-2a/RBV in HCV-1 pts at Week 24 (68% vs. 53%)
-- GI side effects with 200 mg valopicitabine/peg-IFN_ less frequent and less severe compared to higher valopicitabine dose regimens: By Week 24, no d/c's for GI-related AEs in patients reduced to 200 mg
-- No incremental hematologic toxicity of 200 mg valopicitabine/peg-IFN_ versus peg-IFN_ monotherapy
-- For Phase III trials in rx-naive patients, valopicitabine 200 mg/d is the preferred dose for use with peg-IFNa w/wo RBV
Safety Summary
Valopicitabine Phase IIb Treatment Naive Study
Most frequent AEs were GI-related (nausea, vomiting, loose stools):
-- Usually mild/moderate; lessen within 1-2 wks in most pts, typically manageable with continued treatment
--In 800 mg arms, -20% of pts with persistent moderate/severe GI symptoms
-- GI AEs less frequent and less severe with 200 mg/day
29/173 (17%) pts d/c'd for AEs, mostly GI
-- Only 3/34 (9%) pts d/c'd for GI-related AEs from 200 mg arm
-- In pts dose reduced to 200 mg, no d/c's for GI-related AEs by Week 24
15 SAEs reported by Week 24
5 attributed to valopicitabine or valopicitabine/peg-IFN_, mostly GI (1 in 200 mg arm)
- No GI related drug-attributed SAEs since dose reduction
- Only 1 drug-attributed SAE (TB reactivation) post dose reduction (400 mg/peg-IFN)
Lab abnormalities
- Decreases in WBC, ANC & platelets in all treatment arms consistent with peg-IFN_
- No grade 3/4 hemoglobin decreases in any treatment arm
- Sporadic grade 3/4 elevations in AST, amylase, lipase (predominantly 800 mg arms)
In the 200 mg arm
- No grade 1-4 hemoglobin decreases
- No grade 3/4 amylase/lipase elevations
- Only 1 pt with grade 3 ALT/AST increase
Breakthrough/Resistance Evaluations
-- S282T is signature mutation for valopicitabine resistance in vitro.
-- HCV RNA sequencing undertaken for all patients with viral breakthrough or with detectable viremia as study progresses.
-- No resistance to date in treatment-naive study. One breakthrough due to non-compliance.
-- In Phase IIb non-responder trial, S282T confirmed as breakthrough mutation.
Results consistent with relatively durable effects for nuc-type HCV Pol inhibitors, (analogous to HIV NRTIs).
DETAILED STUDY RESULTS REPORTED
Objectives & Key Eligibility Criteria
Valopicitabine Phase IIb Treatment Naive Study
Valopicitabine (NM283): nucleoside-type HCV polymerase inhibitor, evaluated in > 400 patients in Phase II trials
Objectives
Identify optimal valopicitabine dose for use in combination regimen(s) with peg-IFN_ (+/- RBV) for Phase III registration studies
Efficacy objectives
Antiviral efficacy vs peg-IFNa alone at Week 4
Clearance to non-detectable HCV RNA during treatment with
valopicitabine/peg-IFNa regimens
Sustained virologic response (SVR) rates
Safety/tolerance objectives
Safety/tolerance for valopicitabine/peg-IFNa regimens
Key Eligibility Criteria
HCV genotype 1, treatment naive, compensated liver disease
Candidate for interferon therapy
Baseline HCV RNA ≥5 log10 IU/mL (100,000)
Baseline ALT >1.0 x ULN and <5 x ULN
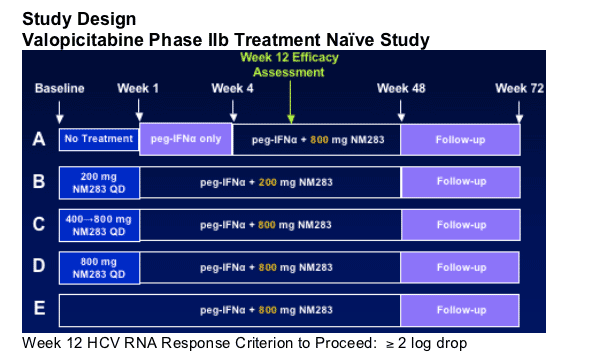
Protocol Amendment: Dose Modification
Valopicitabine Phase IIb Treatment Naive Study
GI tolerance issues for some patients at 800 mg/d valopicitabine dose level. A dose reduction was implemented in late March 2006:
Patients receiving 800 mg valopicitabine/peg-IFN continued treatment at reduced dose, randomly assigned (1:1) to:
- 200 mg valopicitabine/peg-IFN_ or
- 400 mg valopicitabine/peg-IFN_
At time of protocol amendment:
-- 12 patients with HCV RNA ≥ 600 IU/mL, all discontinued
-- RNA data included in efficacy analyses, LOCF from discontinuation
68% of patients had completed study Week 12
Patients in Group B (200 mg valopicitabine/pegIFN) continued study treatment unchanged.
Data displayed according to original treatment assignment
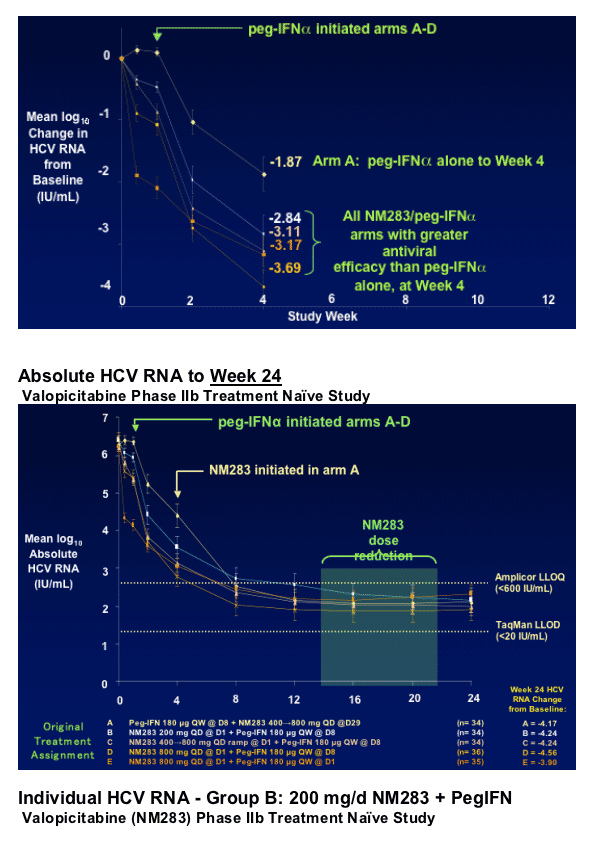
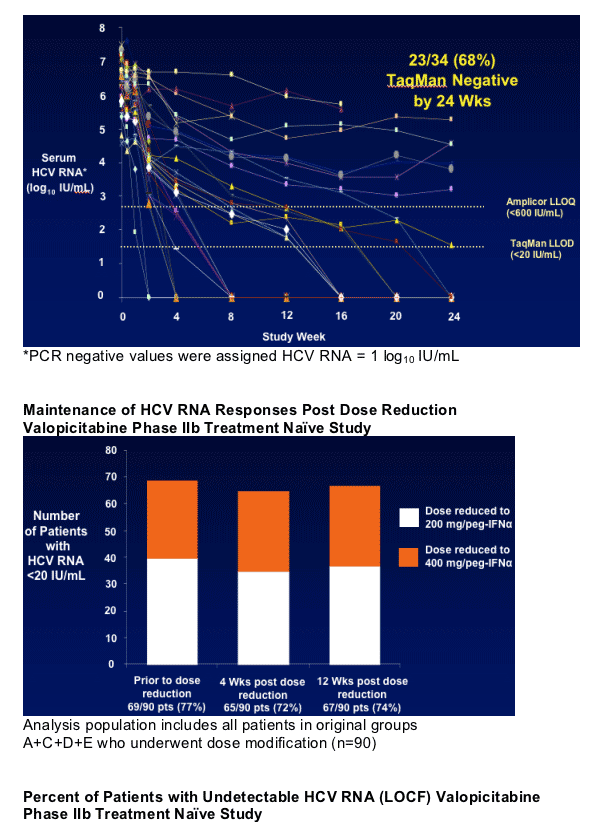
|
| |
|
 |
 |
|
|