 |
 |
 |
| |
Infergen (Consensus Interferon) Phase III DIRECT Study End of Treatment Results
|
| |
| |
Reported by Jules Levin
AASLD, Oct 27-31, 2006, Boston, MA
"USE OF DAILY INTERFERON ALFACON-1 (INFERGEN) PLUS RIBAVIRIN IN PATIENTS INFECTED WITH HEPATITIS C VIRUS WHO ARE NONRESPONDERS TO PREVIOUS PEGYLATED INTERFERON PLUS RIBAVIRIN THERAPY: 24-WEEK AND END OF TREATMENT DATA FROM THE DIRECT TRIAL"
Bruce Bacon1; Arie Regev2; Reem Ghalib3; Giuseppe Morelli4; Kenneth Rothstein5;Tarek Hassanein6; Mitchell Shiffman7; Yi Xu8; Brian Murphy8
1Saint Louis University Liver Center, St. Louis University School of Medicine, St. Louis, MO, USA; 2University of Miami, Miami, FL, USA; 3Liver Institute at Methodist Hospital, Dallas,TX, USA; 4University of Florida, Gainesville, FL, USA;
5Albert Einstein Center for Liver Disease, Philadelphia, PA, USA; 6University of California San Diego, San Diego, CA, USA; 7Virginia Commonwealth University, Richmond,VA, USA; 8Valeant Pharmaceuticals International, Costa Mesa, CA, USA
BACKGROUND
- Up to 40%-50% of patients undergoing initial treatment for chronic hepatitis C with PEG IFN and RBV fail to achieve SVR
- Fewer than 15% of previous PEG IFN and RBV treatment failures respond to retreatment with a second course of PEG IFN and RBV
- Interferon alfacon-1 (consensus interferon, CIFN) has been shown in vitro to be more effective than IFN alfa-2a and IFN alfa-2b and to apply constant pharmacologic pressure on HCV when combined with RBV
- Preliminary studies suggest that daily dosing with CIFN plus RBV can improve outcomes in patients who are nonresponsive to previous PEG IFN and RBV therapy
STUDY OBJECTIVE
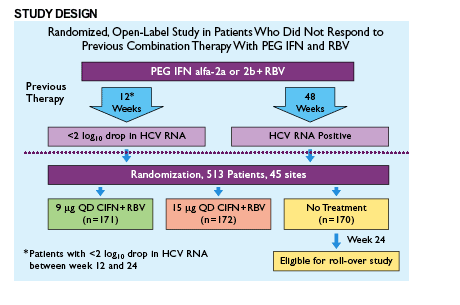
- DIRECT is a Phase 3, randomized (1:1:1), open-label research study to determine the sustained virologic response (SVR) rate of combination therapy with daily CIFN at either of two doses plus RBV compared with no treatment in HCV-infected patients who were nonresponsive and were compliant to previous PEG IFN plus RBV therapy
- This interim analysis reviews the proportion of patients who are serum HCV RNA negative by bDNA/TMA assay at week 24 and end of treatment (EOT) while being treated with 9 or 15 _g of CIFN plus RBV. EOT response data were also analyzed by fibrosis score, compliance, and median washout period
METHODS
Key Eligibility Criteria
- Patients must have been previously treated with the following starting doses:
- 1.5 mg/kg/week PEG IFN alfa-2b
- 180 mg/week PEG IFN alfa-2a
- Patients must have had documented viral nonresponse to past treatment with PEG IFN alfa-2a or -2b plus RBV
- Patients with all viral genotypes and cirrhotics were considered for enrollment
- Chronic HCV infection based on a history of positive serum HCV antibody and/or HCV RNA
- Liver biopsy performed within 3 years of screening must have demonstrated chronic liver disease consistent with chronic HCV infection
No adjunctive growth factors were used.
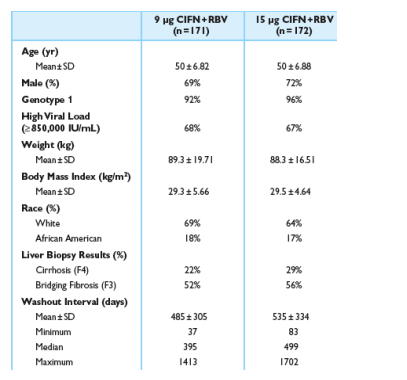
Demographics
Average age: 50; 70% men; 94% genotype 1; 68% high viral load )850,000 IU/mL; mean weight: 89 kg; mean BMI: 29.5 kg/m2; 66% white; 17% African-American; 22& & 29% cirrhosis (F4) in the 9 & 15 ug dosing regimens, respectively; 52% & 56% had bridging cirrhosis in the 9 & 15 ug dosing regimens, respectively; washout interval: mean 485 days in 9 ug & 535 days in 15 ug arms, respectively.
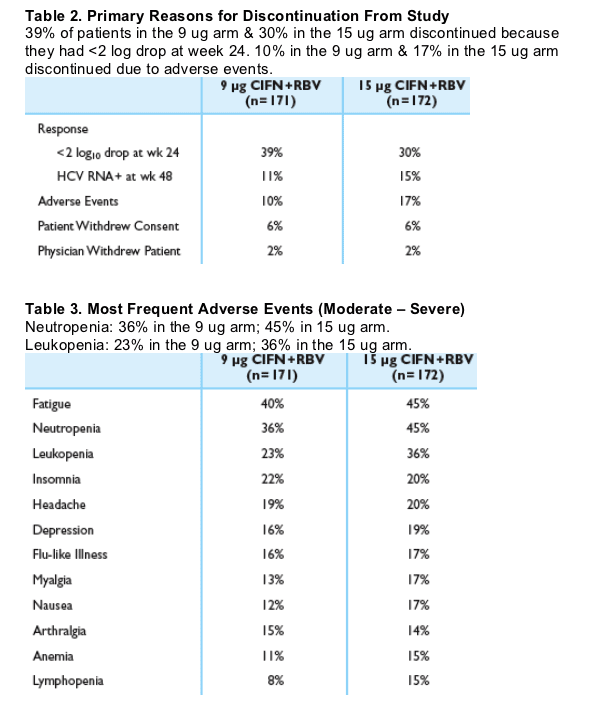
BRESULTS
Figure 2.Week 24 and EOT Viral Response (ITT)
At week 24, 14% in the 9ug dose group & 20% in the 15 ug group had <5 IU/mL (TMA). At week 48, 16% in the 9 ug & 19% in the 15 ug had <5 IU/mL (TMA). Using the bDNA assay, at week 24, 26% in the 9 ug arm & 32% in the 15 ug arm had <615 IU/mL. At week 48, 22% in the 9 ug & 25% in the 15 ug group had <615 IU/mL.
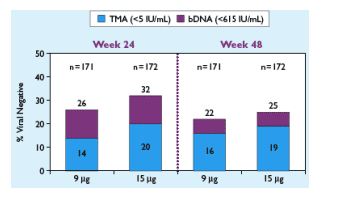
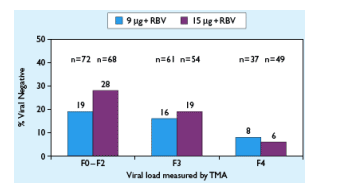
Figure 3. EOT Response by Fibrosis Score
For patients with F0-F2 fibrosis 19% with 9 ug & 28% with 15 ug were virus-negative (TMA). Patients with F3, 16% & 19% using 9 ug & 15 ug, respectively, were virus-negative (TMA). For patients with F4 cirrhosis, 8% using 9 ug & 6% using 15 ug were virus negative.
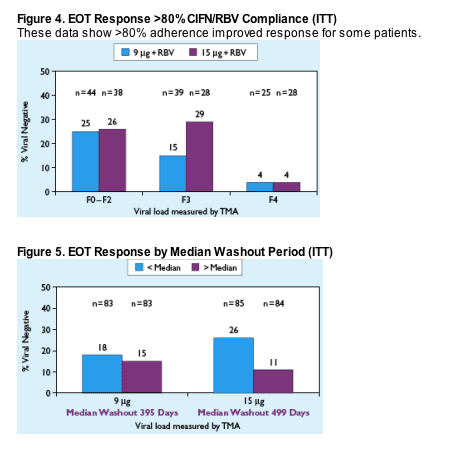
AUTHOR CONCLUSIONS
- Shorter "washout" duration after PEG IFN/RBV therapy optimized response
- Maximizing CIFN dose improved response
- Approximately 25% of non-cirrhotic patients achieved an EOT response
- Minimal viral breakthrough between weeks 24 and 48
- Daily CIFN in combination with RBV was well tolerated at doses up to 15 _g
Based on this interim data, viral clearance could be enhanced by maximizing
CIFN/RBV exposure through the use of adjunctive therapy with growth factors.
Further study of these interventions is warranted, including assessing the impact of washout period on viral clearance.
|
| |
|
 |
 |
|
|