 |
 |
 |
| |
CPG10101 HCV Toll-Receptor 9 Antagonist Phase II Study results
|
| |
| |
Reported by Jules Levin
AASLD, Oct 2006, Boston, MA
Early Viral Response & On Treatment Response to CPG 10101 (ACTILON), in Combination with Pegylated Interferon and/or Ribavirin, in Chronic HCV Genotype 1 Infected Patients with Prior Relapse Response
I.M. Jacobson1, R. Ghalib1, E. Lawitz1, B. Freilich1, S.C. Gordon1,
P. Kwo1, T.R. Riley1, D. Bright2, M.L. Morris2, M. Al-Adhami2,
J.L. Himes2, F.E. Massari2, J.G. McHutchison1
1CPG 10101-003 Study Group
2Coley Pharmaceutical Group, Inc.
AUTHOR SUMMARY & CONCLUSIONS
CPG 10101 improves antiviral activity of PEG-IFN a-2b + RBV in Relapsed Responders
- Greater 2 log10 (at Week 4, 8, 12) & HCV RNA Undetectable responses in CPG 10101 + PEG-IFN a-2b + RBV group than PEG-IFN a-2b + RBV group
Early treatment responses seen with CPG 10101 + PEG-IFN a-2b + RBV are sustained while on treatment (Undetectable at Week 24)
CPG 10101 appears synergistic with PEG-IFN a-2b + RBV
CPG 10101 generally well tolerated in combinations with PEG-IFN a-2b and/or RBV
- AEs were predominantly mild and consisted of flu-like systemic reactions and local injection site reactions
EVR patients continuing treatment for 48 weeks
- SVR data forthcoming in 1H '07
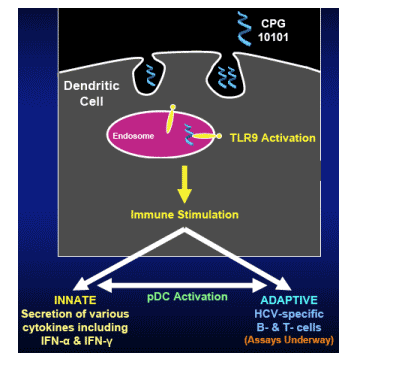
CPG 10101 (ACTILON ):
Is an Investigational TLR9 Agonist for Treatment of HCV
It is a Synthetic Oligodeoxynucleotide
(ODN)
Its Targeted Mechanism is
- TLR9 agonist
- Dendritic Cell-mediated immune stimulation
- B cell stimulation
Its Synergistic with IFN-_ (Vollmer, et al. Antiviral Therapy 2002; 7:L115)
Its Synergistic with RBV (AASLD 2006 Poster #1149, Tuesday Oct 31)
McHutchison, et al., AASLD 2005 Abstract #873; Krieg, Nature Med, 2003
CPG 10101 Clinical Development
Program to Date
Phase 1a Healthy
Volunteer Monotherapy
Dose Escalation
(N = 48)
0
Phase 1b Mixed HCV
Pop. Monotherapy
Dose Escalation
N=60
COMPLETED
0
48 week Combinations + PEG-IFN + RBV
in Relapsed Responders (n=74) (enrolled)
and
48 week 2 dose level combination + PegIFN + RBV Nonresponders (n=90) Enrolling
CPG 10101 Combination Therapy
in Treatment-Refractory (Relapsed) HCV Patients Study
Objectives
- Safety & tolerability
- Plasma viral RNA levels
- (RVR, EVR, OTR, ETR, SVR)
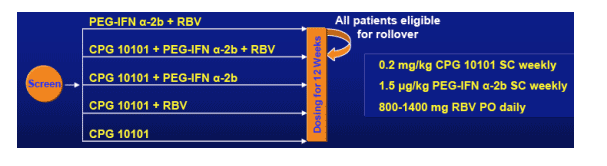
Study Design
- Multicenter, randomized Phase Ib
- Genotype 1, PEG-IFN + RBV
Treatment-Refractory (RRs)
68 patients receive 1 of 4 arms:
PegIFN a-2b +RBV
CPG 10101 + PegIFN a-2b +RBV
CPG 10101 + PegIFN a-2b
CPG 10101 +RBV
CPG 10101
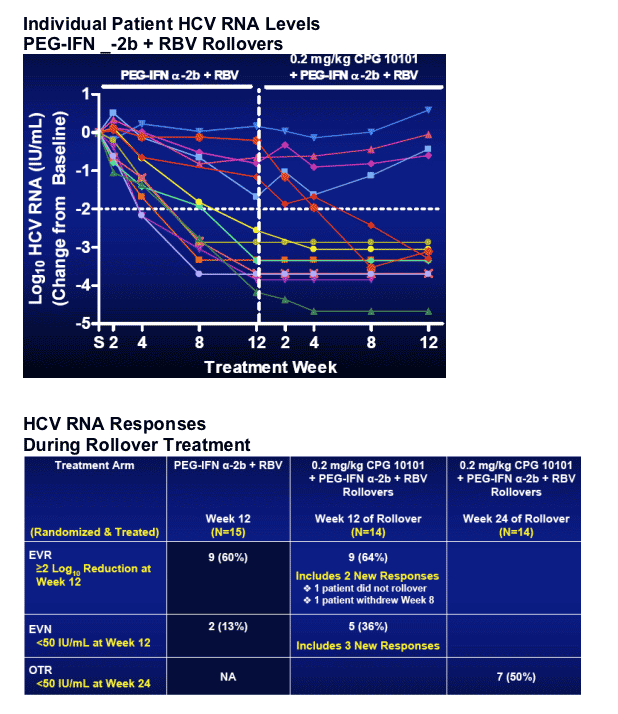
All patients eligible for rollover.
Dose of 0.2 mg/kg subcutaneous injection weekly used in this study is a low dose and higher dose will be studied. PegIFN dose is 1.5 ug/kg weekly, RBV 800-1400 daily.
-- 12 week treatment, then 48 week if ≥2 log10 reduction at Week 12
-- If HCV RNA Undetectable at Week 48, then follow-up through Week 72
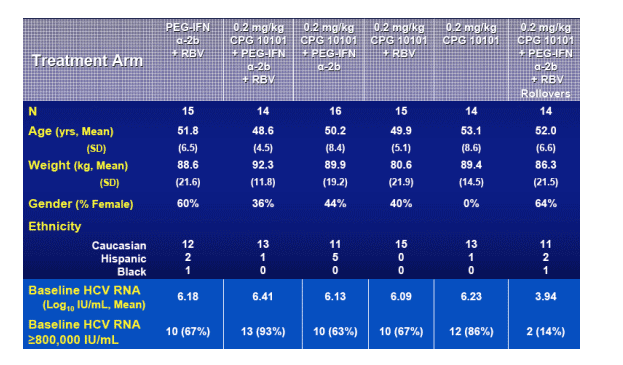
Study Population & Demographics
About 15 patients in each arm including rollover. Average age 48-50. Mean weight 86 to 92 kg, 80 in 0.2 mg/kg CPG10101 + RBV. Mostly Caucasians. Baseline HCV RNA mean 6.13 to 6.41, except its 3.94 in CPG 10101 0.2 mg/kg + Peg+RBV rollover arm. 67-93% had baseline HCV RNA >800,000 Iu/mL. HCV RNA for rollovers: 14% >800,000 IU/ml.
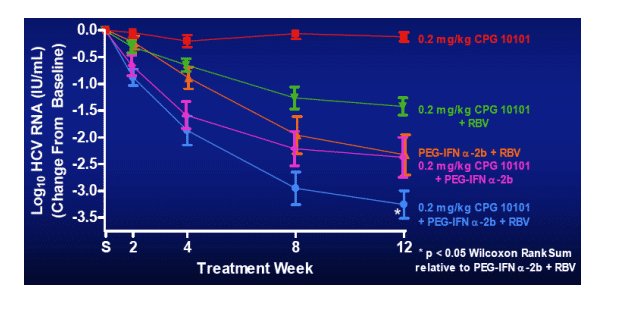
Group Mean HCV RNA Levels ± S.E.M.
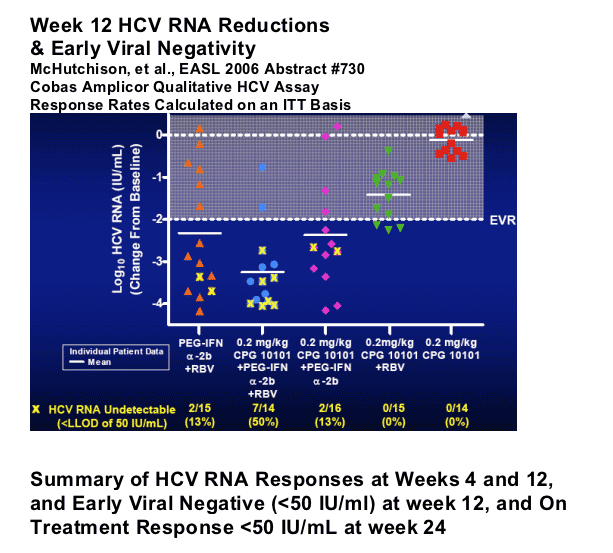
McHutchison, et al., EASL Spring 2006 Abstract #730
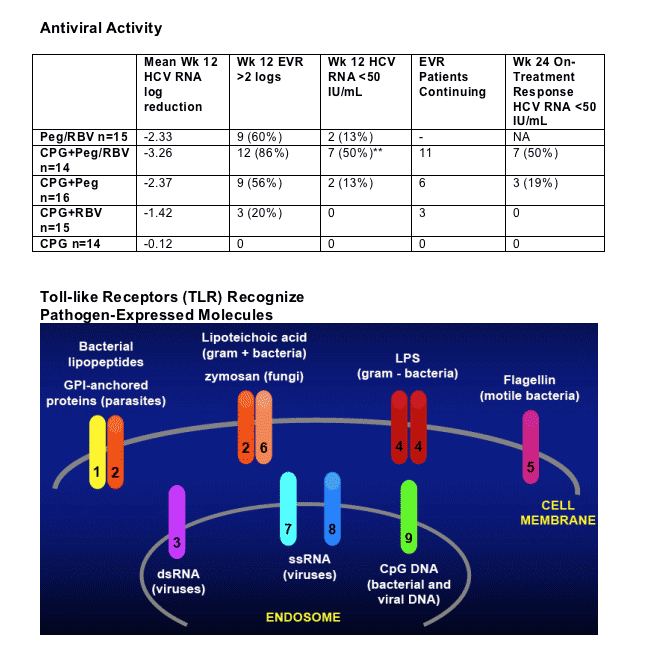
Little or no HCV RNA reduction (mean -0.12 log) in CPG10101 monotherapy arm (0.2 mg/kg; in speaking with company officials, they said this is a low dose & expect to examine highrr dose).
ITT
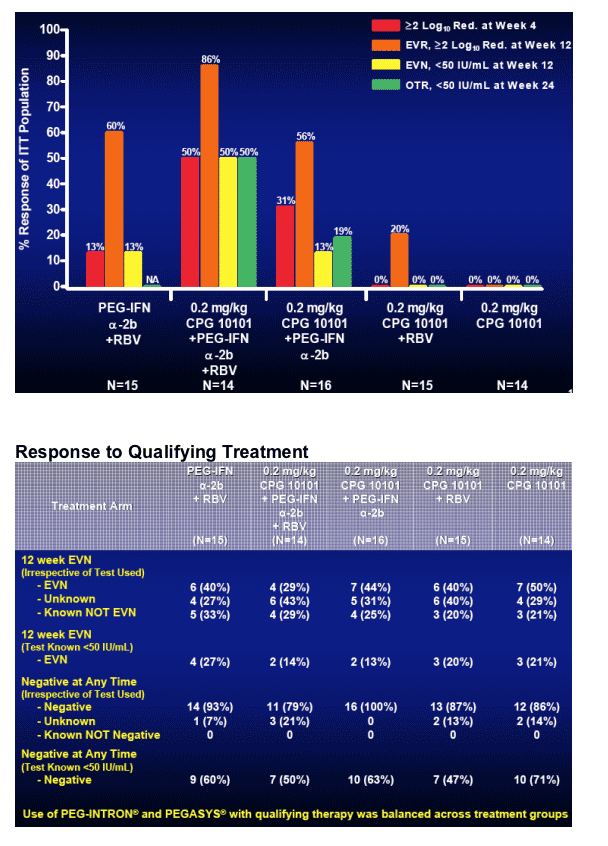
-- CPG 10101 0.2 kg/mg alone: mean -0.12 log; 0/14 <50 c/ml
-- CPG10101 0.2 mg/k +RBV: mean -1.42 logs; 0/15 <50 c/ml
-- Peg+RBV: mean -2.3 log; 2/15, 13% <50 c/ml
-- CPG10101 0.2 mg/kg + Peg: mean -2.37 log; 2/16, 13% <50 c/ml
-- CPG10101 0.2 mg/kg + Peg + RBV: mean -3.26 logs; 50%, 7/14 <50 c/ml
(p<0.05 Wilcoxon Rank Sum, relative to Peg+RBV, re: log reduction; viral negativity p=0.050, Fisher's Exact Test).
In other words Adding CPG10101 at 0.2 mg/kg dose to Peg/RBV resulted in increased HCV RNA reduction from -2.2 log with Peg/RBV alone to -3.3 log when CPG was added to Peg/RBV.
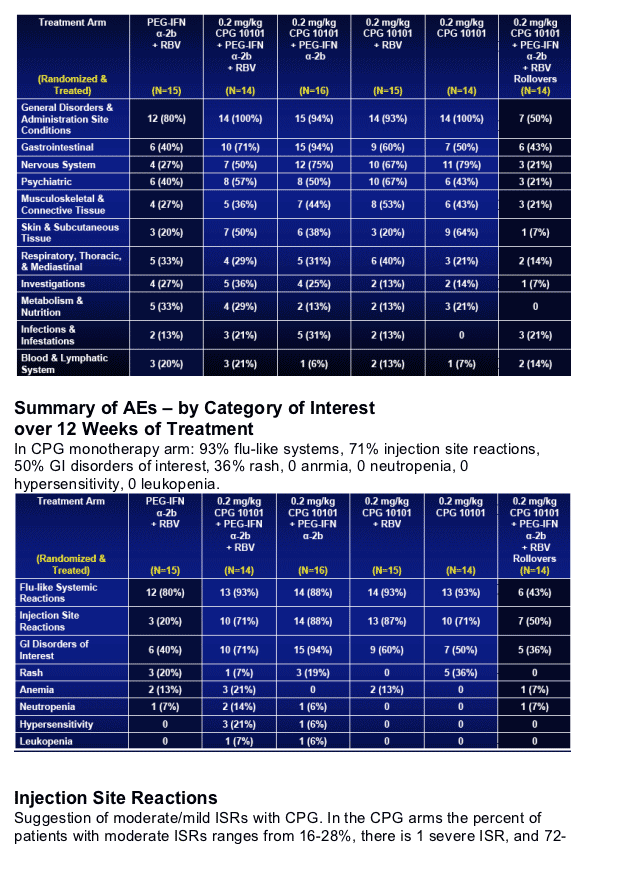
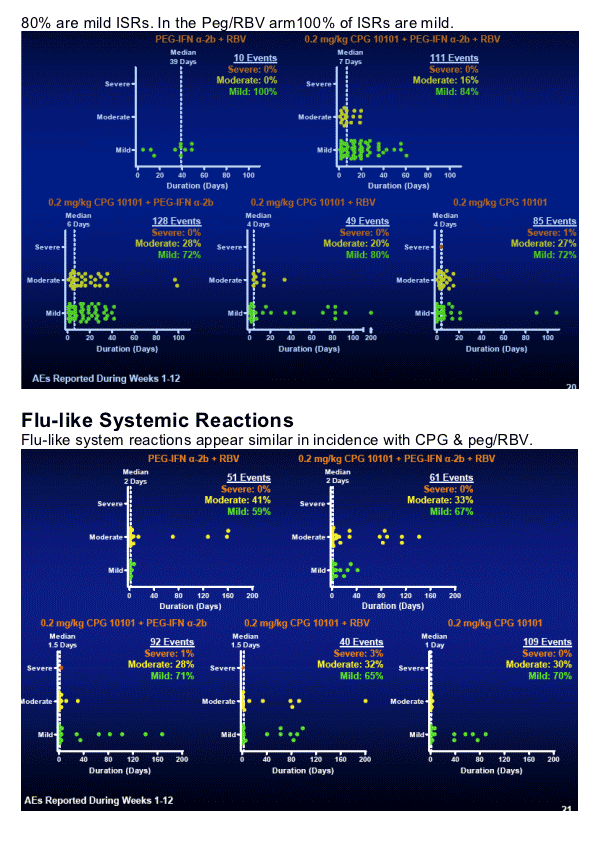
Safety & Tolerability
CPG 10101 combinations generally well tolerated
-- AEs were transient & consistent with mechanism
Predominantly mild/moderate in intensity in all groups
-- Most frequent: Pyrexia, fatigue, headache, nausea
-- CPG 10101 only added mild AEs
Predominantly injection site reactions
6 withdrawals up to Week 12
- 2 Drug-related AE
Injection site cellulitis & necrosis (CPG 10101 + PEG-IFN _-2b) (SAE)
Rash (CPG 10101)
- 2 Consent
- 1 Non-compliance & 1 lost to follow up
6 withdrawals after Week 12
- 2 Drug-related AE
Depression (CPG 10101 + PEG-IFN a-2b + RBV Rollover)
Chest Discomfort (CPG 10101 + PEG-IFN a-2b + RBV)
-- 2 Consent
-- 1 Withdrawn by Investigator & 1 lost to follow up
1 Additional CPG 10101-related SAE at Week 12
Immediate hypersensitivity (CPG 10101 + PEG-IFN a-2b)
|
| |
|
 |
 |
|
|