 |
 |
 |
| |
Integrase Inhibitor: JTK-303/GS-9137, a Novel Small Molecule Inhibitor of HIV-1 Integrase:
Anti-HIV Activity Profile and Pharmacokinetics in Animals
|
| |
| |
Reported by Jules Levin
Poster Number 508
13th Conference on Retroviruses and Opportunistic Infections
February 5-9, 2006, Denver, CO
Yuji Matsuzaki1, Wataru Watanabe1, Kazunobu Yamataka1, Motohide Sato1, Seiji Enya1, Mitsuki Kano1,
Eiichi Kodama2, Masao Matsuoka2, and Satoru Ikeda1
1Japan Tobacco Inc., Osaka, Japan and 2Institute for Virus Research, Kyoto University, Japan
Currently approved therapies for HIV-1 infection target the virus's reverse transcriptase or protease, or inhibit fusion of the virus with the cell membrane. Integrase inhibitors fill an unmet need to target the third viral enzyme; none is approved to date, although several inhibitors are under development.
JTK-303/GS-9137 (chemical name: 6-(3-chloro-2-fluorobenzyl)-1-[(2S)-1-hydroxy-3-methylbutan-2-yl]-7-methoxy-4-oxo-1, 4- dihydroquinoline-3-carboxylic acid) was discovered at the Central Pharmaceutical Research Institute of Japan Tobacco Inc. It was found to be a low molecular weight HIV-1 integrase inhibitor through screening of inhibitory activity against recombinant HIV-1 integrase
(based on the full-length sequence of HIV-1 NL4-3 gene).
Author conclusions:
- JTK-303/GS-9137 is a highly selective HIV-1 integrase inhibitor
With potent antiviral activity against both B and non-B subtypes of
HIV-1.
- JTK-303/GS-9137 retains antiviral activity against drugresistant
HIV-1 carrying resistance mutations to multiple drug classes.
- JTK-303/GS-9137 has additive to highly synergistic antiviral
activity with three currently approved anti-HIV-1 drug classes.
- In preclinical animal studies, JTK-303/GS-9137 has good oral
bioavailability.
- JTK-303/GS-9137 has potential as a novel, orally bioavailable
anti-HIV agent and has progressed to early clinical studies.
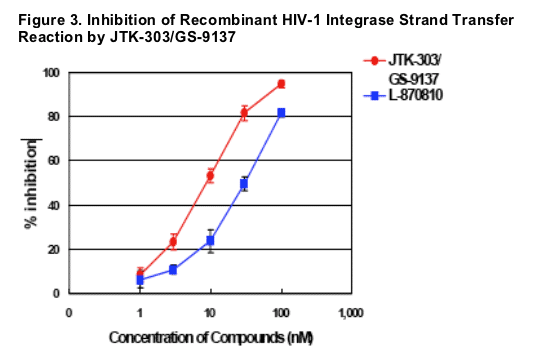
IC50s of JTK-303/GS-9137 and L-870810 on strand transfer reaction (microtiter plate assay) were 8.8 ± 0.9 nM and 30.2 ± 4.0 nM (mean ± SD of 3 experiments),
respectively.
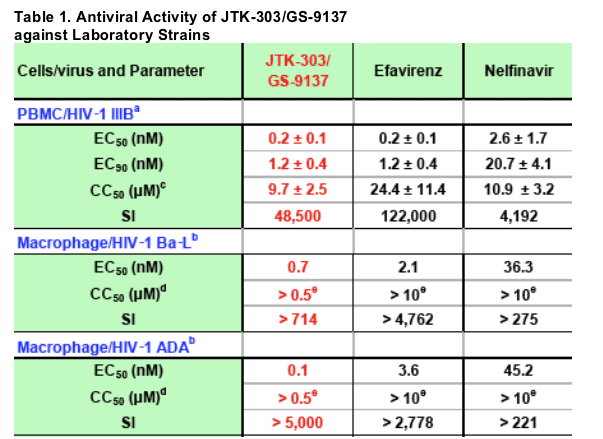
a: Mean ± SD of 3 experiments
b: 1 experiment (triplicate)
c: Cytotoxicity was assessed by uptake of [3H]-thymidine into cells.
d: Cytotoxicity was determined using MTT assay.
e: CC50s were higher than maximum concentrations of these compounds in each assay.
JTK-303/GS-9137 showed potent antiviral activity against the laboratory
strains tested, with EC50 ranging from 0.1 to 0.7 nM. The selectively
index (SI, CC50/EC50) also showed that the antiviral activity of JTK-303
was not the results of cytotoxicity. The potency of JTK-303 was greater
than that of NFV and similar to that of EFV. The antiviral activity of JTK-
303 was moderately reduced by the addition of 50% human serum.
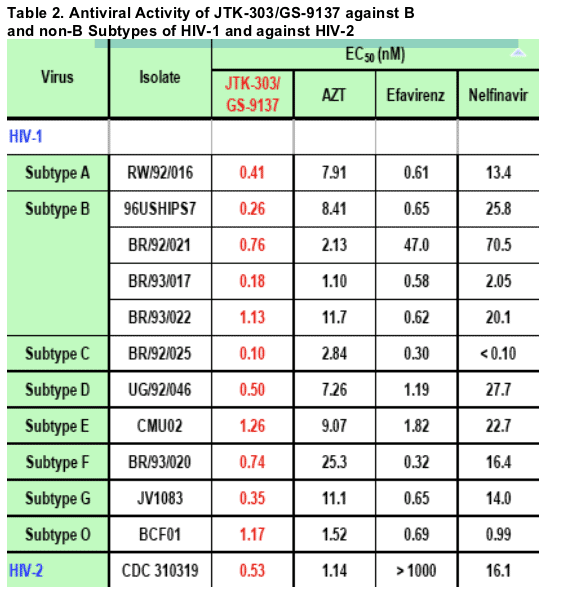
The experiments were conducted in human PBMCs (1 experiment, triplicate).
JTK-303/GS-9137 showed potent antiviral activity against all clinical isolates representing 4 subtype B and 7 non-B subtypes of HIV-1. The average EC50 of JTK-303 was 0.62 nM for the 8 subtypes of HIV-1 (11 viruses), and was 0.53 nM for the single HIV-2 clinical isolate. JTK-303 showed comparable or greater potency than AZT, EFV and NFV against all the isolates tested.
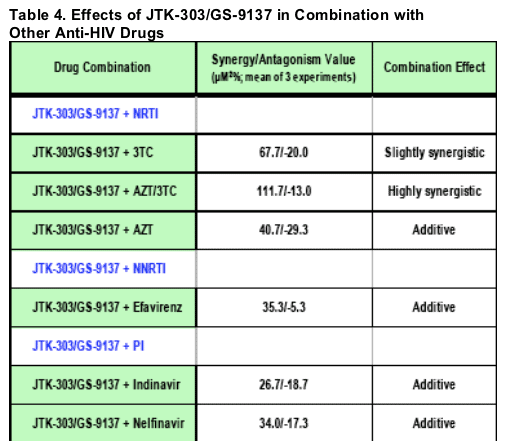
The combination effects of JTK-303/GS-9137 were analyzed by the Prichard and
Shipman method using MacSynergy II software. Results of the combination
assays were expressed as the mean synergy/antagonism volume values (_M2%)
that were calculated at the 95% confidence interval from 3 separate experiments.
Combination effects were defined as:
Highly synergistic: > 100 _M2%
Slightly synergistic: > 50 and ≦ 100 _M2%
Additive: ≦ 50 and > -50 _M2%
Slightly antagonistic: ≦ -50 and > -100 _M2%
Antagonistic: ≦ -100 _M2%
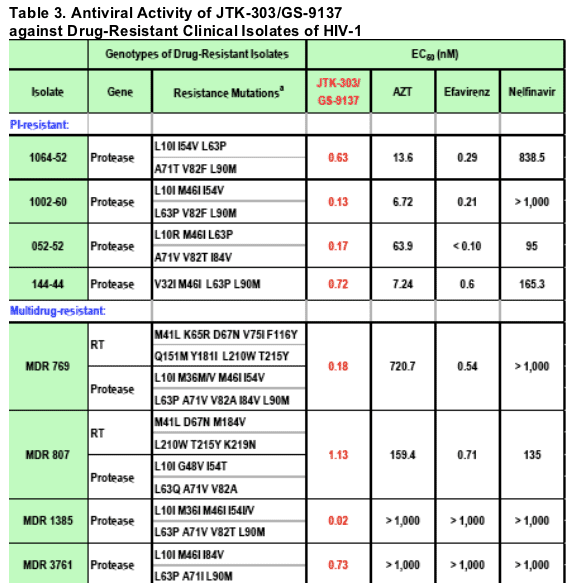
The experiments were conducted in human PBMCs (1 experiment, triplicate).
a: Mutation sites are represented as the position number in the RT- or protease-coding region, and the amino acid residue in the wild-type and drug-resistant viruses.
The average EC50 of JTK-303/GS-9137 was 0.46 nM. Although two of the isolates (MDR 1385 and MDR 3761) were completely resistant to AZT, EFV, and NFV (EC50s > 1,000 nM), they were highly sensitive to JTK-303 (EC50s < 1 nM).
METHODS
HIV-1 Integrase Assay
Gel Electrophoresis: HIV-1 NL4-3 integrase (1.5 _M) and compounds were incubated with 50 nM of a 32P-labeled 21 bp viral DNA substrate (derived from HIV-1 U5 LTR) for 60min. After electrophoresis of reaction products, 19-mer oligonucleotides (3'-processed products), >21-mer oligonucleotides (strand transfer products, STP) and total radioactivity present in the lane were
quantified by measuring radioactivity of corresponding bands using an
image analyzer.
Microtiter Plate Assay: Biotin-labeled donor DNA representing HIV-1 U5 LTR was immobilized onto streptavidin-coated microtiter plates. HIV-1 NL4-3 integrase (300 nM) was incubated with 3'-end processed donor DNA, followed by removal of unassociated enzyme, and the DNA strand transfer reaction was initiated by the addition of target DNA (5 nM) and compounds. The amount of target DNA ligated to the donor DNA was measured by detecting digoxigenin-labeled target DNA.
Antiviral Assay in Human PBMCs and Macrophages: Human PBMCs (PHA-stimulated) and macrophages were infected with each isolate, then treated with serially diluted compounds and cultured for seven days. Antiviral activity was determined using p24 antigen ELISA.
In Vitro Combination Study: CEM-SS cells were infected wtih HIV-1IIIB strain at an MOI of approximately 0.01, then treated with serially diluted JTK-303/GS-9137 and approved anti-HIV-1 drugs. After cultivation for six days, antiviral activity was determined using MTT assay.
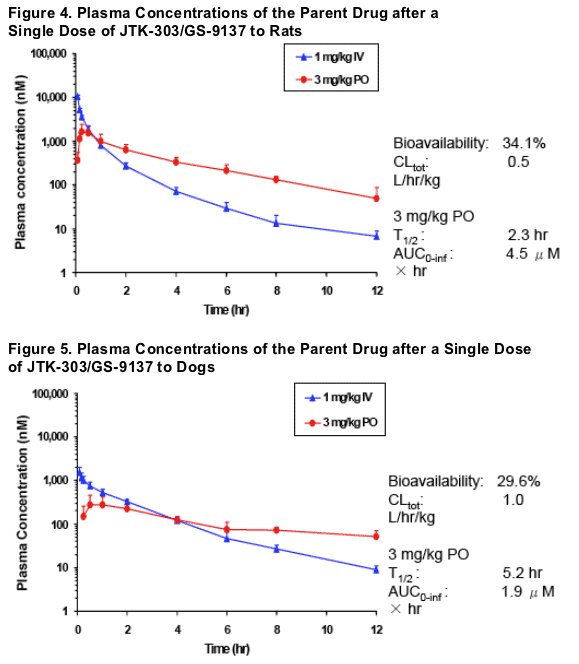
Pharmacokinetic Study in Animals: In rats and dogs, plasma concentrations of JTK-303/GS-9137 were quantified after a single oral dose of 3 mg/kg or intravenous dose of 1 mg/kg, and relevant pharmacokinetic parameters were calculated.
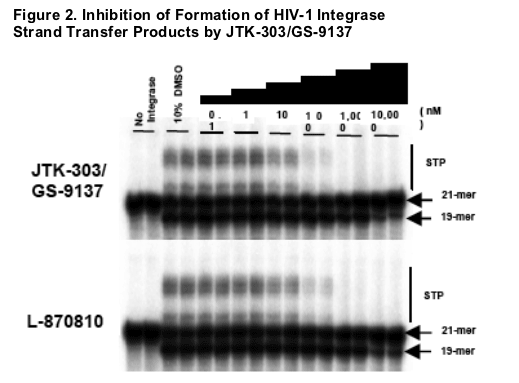
Gel electrophoresis showing 21-mer, 19-mer and STP, corresponding to DNA substrate, 3'-processed products and strand transfer products, respectively.
|
|
| |
| |
|
 |
 |
|
|