 |
 |
 |
| |
Gilead HIV-1 Integrase Inhibitor GS-9137 (JTK-303) Exhibits Potent Antiviral Activity in Treatment-Naive and Experienced Patients
|
| |
| |
Reported by Jules Levin
13th Conference on Retroviruses and Opportunistic Infections
February 5-9, 2006, Denver, CO
Edwin DeJesus , Daniel Berger, Martin Markowitz, Calvin Cohen, Trevor Hawkins, Peter Ruane 6, Richard Elion7, Charles Farthing8, Andrew Cheng9 and Brian P. Kearney9 for the 183-0101 Study Team
1 Orlando Immunology Center, Orlando, FL, 2 NorthStar Medical Center, Chicago, IL, 3 The Rockefeller University Hospital and Aaron Diamond AIDS Res. Ctr, New York, NY, 4 Community Research Initiative of New England, Boston, MA, 5 Southwest C.A.R.E., Santa Fe, NM, 6 Light Source Medical, Los Angeles, CA, 7 C.A.R.E.-ID, Washington, DC, 8 AHF Research Center, Beverly Hills, CA, 9 Gilead Sciences, Foster City, CA
The most exciting news at the 13th CROI was the presentation on the last day of the latest study results on the two integrase inhibitors from Merck & Gilead. The Merck integrase is further along in development as it is entering large global phase III studies now. 10-day monotherapy data on the Gilead integrase inhibitor was presented. In theory, there is a chance that both integrase inhibitors can either be used together in combination or serially, as there may not be cross-resistance. Researchers will explore this possibility. The Merck integrase is administered with one pill twice daily. The Gilead integrase has been studied with a once daily and twice daily administration without ritonavir, and in a once daily small dose boosted by 100 mg of ritonavir. By reading the results from both the Merck & Gilead studies, the Merck integrase may appear more potent, but the Gilead study was only for 10 days so it is too soon to evaluate if one of the two integrase inhibitors is more potent than the other. Merck presented safety and side effects in their study and the drug so far appears very clean, so side effects or toxicities of note. As the gilead study was only 10 days monotherapy, it is too soon to evaluate safety and side effects or toxicities.
THE STUDY
Data from the first study in patients on the Gilead integrase inhibitor was presented for the first time publicly, along with the presentation on data in patients on the Merck integrase inhibitor. The Gilead integrase drug GS-9137 was administered in this early 10-day monotherapy study in various doses which included a ritonavir boosted regimen along with a low dose of GS-9137 (50 mg GS-9137/ 100 mg RTV. After 10 days of dosing the boosted dose reduced HIV RNA viral load by -2.03 log copies/ml. The 800 mg once daily dose of GS-9137 reduced HIV RNA by -1.78 log copies/ml.
Author conclusion:
GS-9137 monotherapy demonstrated substantial antiviral activity at all doses: about 2-log median reductions observed at 400 mg and 800 mg bid or 50 mg QD with ritonavir 100 mg GS-9137 was well tolerated, there were no study drug discontinuations. With similar adverse events and laboratory abnormalities to placebo. Once daily dosing of GS-9137 + RTV will be evaluated in a dose-ranging phase 2 study in treatment-experienced patients.
GS-9137 (JTK-303) is a potent (in vitro protein binding-adjusted inhibitor of HIV-1 integrase (strand transfer) with favorable human pharmacokinetics (PK), including QD dosing with ritonavir 100 mg (RTV).
GS-9137 Background
Preclinical pharmacokinetics
--Metabolism: oxidative (CYP3A) and glucuronidation pathways
--Moderate inducer of CYP3A, not an inhibitor
Pharmacokinetics of unboosted 9137 (poster 580, next article/posted on NATAP website)
--Single dose PK volunteer generally dose dependent
--Higher relative systemic exposure when dosed with food
--Half-life - 3 hours
Pharmacokinetics of boosted 9137 (ritonavir 100 mg)
--about 20-fold higher systemic exposure (AUC)
--No autoinduction of metabolism
--Long plasma half-life ( about 9 hr)
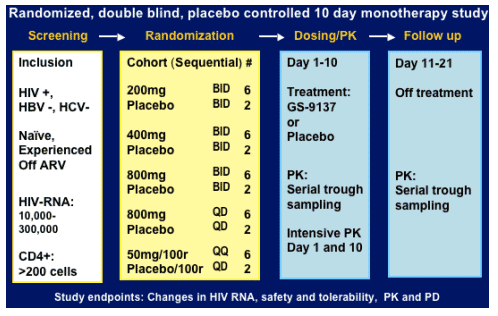
The antiviral activity, safety, pharmacokinetics (PK) and pharmacodynamics (PD) of GS-9137 were evaluated in a prospective, randomized, double-blind, placebo-controlled monotherapy study in 40 HIV-1 infected treatmentnaive and -experienced patients. Eligible patients were not currently receiving antiretroviral therapy with a screening HIV-1 RNA between 10,000 and 300,000 copies/mL and CD4 count ≥ 200 cells/_L. GS-9137 was administered with food for 10 days in dosage cohorts of 200, 400 or 800 mg BID, 800 mg QD or 50 mg boosted with 100 mg RTV QD (6 active + 2 placebo/cohort). Clinical chemistries, HIV-1 RNA, and CD4 cell counts were evaluated throughout the study with intensive PK sampling on days 1 and 10. The primary efficacy endpoint was the maximum reduction in log10 HIV-1 RNA from baseline.
Study design
DEMOGRAPHICS & BASELINE CHARACTERTISTICS
N=40
Mean age- 39
Male/female: 39/1
White/black: 36/4
Treatment-naïve: 15 (11/4)
Treatment-experienced: 25 (19/6)
--N (active drug/placebo)
Mean HIV RNA: 4.75 log copies/ml; min, max (3.69, 5,73 log)
HIV RNA >100,000: 30%
Mean CD4: 442
RESULTS
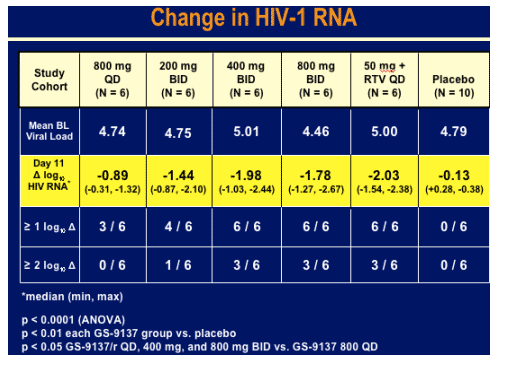
40 patients [30 active (15 experienced and 15 naive), 10 placebo] were enrolled and completed study. There were no study drug discontinuations. All GS-9137 dose cohorts exhibited substantial antiviral activity vs. placebo (p < 0.0001, ANOVA); median (min, max) results are shown below.
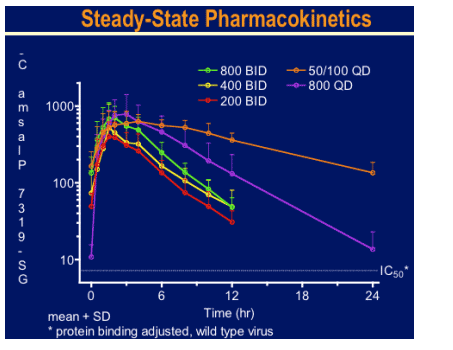
An Emax dose-response model demonstrated that antiviral activity was associated with GS-9137 steady state Cmin concentrations. All GS-9137 dosage regimens were well tolerated with no study drug discontinuations or serious adverse events (AEs). All AEs related to study drug were Grade 1/2 in severity, resolved on treatment, and were not associated with GS-9137 dose.
CHANGE IN HIV RNA
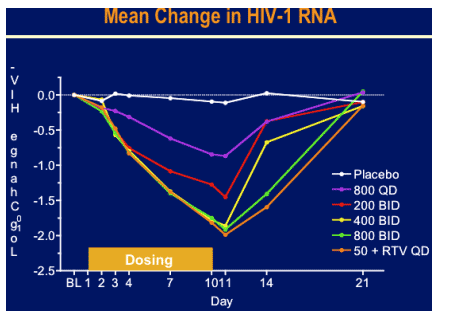
Day 11 log HIV RNA reduction:
50 mg + RTV once daily: -2.03 log; 6/6 had 1 log or more HIV RNA decline; 3/6 had 2 logs or more HIV RNA decline
800mg twice daily: -1.78 log; 6/6 had 1 log or more decline in HIV RNA
400 mg twice daily: -1.98 log; 6/6 had 1 log or more viral load decline; 3/6 had 2 logs or more viral load decline.
200 mg twice daily: -1.44 log; 4/6 had 1 log or more HIV RNA decline; 1/6 had 2 logs or more viral load decline.
800 mg once daily: -0.89 log; 3/6 had 1 log or more HUV RNA decline; 0/6 had 2 logs decline.
placebo: -0.13 log; 0/6 had 1 log or more decline in HIV RNA.
RESISTANCE TESTING
--HIV integrase genome was fully sequenced by RT-PCR at Baseline, Day 10 and Day 21
--Genotypic and phenotypic analysis of integrase sequences are ongoing
ADVERSE EVENTS
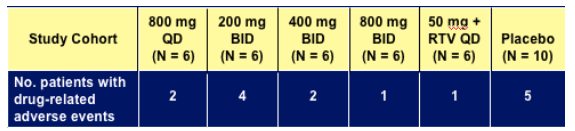
treatment-emergent AEs.
Drug-related adverse events experienced in > 1 patient on GS-9137
- Fatigue: only AE with a greater frequency with GS-9137 vs. placebo
2 in 200 mg BID vs. 1 in placebo
- Diarrhea
- Headache
- Nausea
- Adverse events generally mild and resolved on treatment
- No serious adverse events
- No evidence of QT / QTC changes
Laboratory Abnormalities
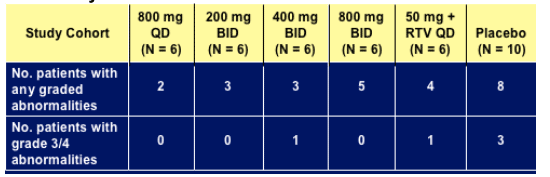
Treatment-emergent laboratory abnormalities
Grade 3 or 4 laboratory abnormalities
- 400 mg BID: non fasting triglycerides (G3)
- 50 mg + RTV: total amylase (G3)
- Placebo: AST (G3), total amylase (G3), creatine kinase (G4)
|
|
| |
| |
|
 |
 |
|
|