 |
 |
 |
| |
A Prospective, Open-label, Pilot Trial of Regimen Simplification to Atazanavir/Ritonavir Alone as Maintenance Antiretroviral Therapy after Sustained Virologic Suppression (ACTG 5201)
|
| |
| |
Susan Swindells (Nebraska Med Ctr, Omaha) for the ACTG reported these study results in an oral session at the 13th CROI.
Ritonavir-boosted atazanavir (ATV/RTV) monotherapy alone is attractive as maintenance or simplification therapy because of its reduced pill burden, once daily dosing, safety/reduced toxicity/improved tolerability, and unique resistance profile. Encouraging recent data from lopinavir/ritonavir alone when compared to lopinavir/r plus NRTIs (4/21 viral failures vs 1/21 viral failures at 48 weeks) provided the impetus for studying atazanavir/r alone. Atazanavir/r is once daily dosing, has lipid advantages, and a unique resistance profile.
Subjects with virologic suppression for at least 48 weeks on their first protease inhibitor (PI)-based regimen containing 2 nucleoside reverse transcriptase inhibitors (NRTI) were enrolled into this single-arm, open-label, multicenter, pilot study. Participants switched from current PI to ATV/RTV at entry (week -6), 3 weeks later it was confirmed they had <50 copies/ml (week -3), and then at week 0 (baseline) discontinued NRTIs.
Study patients were on PI(s) plus NRTIs for at least 48 weeks, had viral load <50 c/ml for at least 48 weeks, had no prior viral failure, CD4 count >250, and were HBsAg negative.
The primary study endpoint was virologic failure (confirmed plasma HIV-1 RNA ≥200 copies/mL) 24 weeks after stopping NRTI (simplification). Followup is continued in this study for 48 weeks.
The study objectives were to evaluate
--risk of viral failure
--safety & tolerability
--change in CD4 count
--relationship among adherence, plasma ATV levels, and viral failure
--estimate prevalence of HIV RNA in genital compartment 24 weeks after simplification
A 90% one-sided confidence interval (CI) for the probability of virologic success (i.e., no virologic failure) 24 weeks after simplification was calculated using Greenwood’s variance. Plasma samples at virologic failure were tested for drug resistance (ViroSeq). Plasma ATV concentrations were measured (HPLC) serially throughout the study. Power calculation assumed success of continued triple therapy 90% vs 75% for ATV/r alone. 85% power to declare inferior to triple therapy with n=30 subjects.
Results
Median age=40 yrs. Gender: 92% men. Race: 61% white, 25% Black, 11% Hispanic, 3% Asian. Cd4 count median: 616 cells.
STEP 1 (n=36)
36 subjects were enrolled of whom 2 discontinued before simplification to ATV/RTV alone (1 developed icterus; 1 HIV RNA of 50 copies/mL at week 3).
STEP 2 (n=34)
--No treatment limiting adverse events.
--1 withdrew consent.
--91% without viral failure by week 24.
--Lower 90% CI limit=85%.
3 Virologic Failures
1,285 cps/ml at week 12
--persistently detectable ATV/r in plasma
4,730 cps/ml at week 14
--undetectable ATV at ≥1 clinic visit
--resuppressed on FTC/TDF plus LPV/r
28,397 cps/ml at week 20
--undetectable ATV ≥1 clinic visit
--resumed NRTIs; F/U viral load pending
Failure Genotypes (ViroSeq)
--no PI mutations
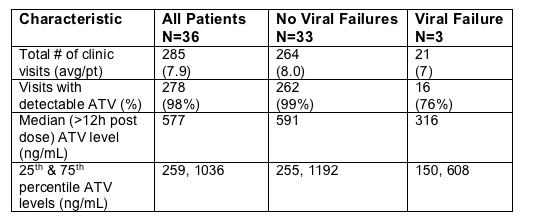
Atazanavir PK Data
Longitudinal CD4 Counts: week -6, 616; baseline, 588; week 24, 604.
Semen HIV-1 RNA
8 participants contributed semen samples at wk 24 after simplification. No detectable HIV RNA was found (<150 cps/ml (below LOD).
There were no treatment discontinuations for adverse events after simplification.
The observed virologic success 24 weeks after simplification was 91% (lower 90% CI limit = 85%).
Author Conclusions:
ATV/r alone appears to be safe & effective for 24 weeks after stopping NRTIs.
Viral failure was not associated with the development of PI mutations.
Viral failure was associated with low or undetectable plasma ATV levels in 2 of 3. (I think this is to suggest non-adherence)
HIV RNA quickly resuppressed in 2 of 3 subjects: HIV RNA on 3rd subject is pending.
Larger, randomized trials are warranted.
|
|
| |
| |
|
 |
 |
|
|