 |
 |
 |
| |
Week 48 Resistance Surveillance of HIV/HBV Co-Infected Patients
Treated with Entecavir in Study AI463038
|
| |
| |
Reported by Jules Levin
13th Conference on Retroviruses
and Opportunistic Infections
Denver, CO Feb. 5-9, 2006
Poster 832
R.J. Colonno, R.E. Rose, C.J. Baldick, K. Pokornowski, B. Eggers, M. Plym, J. Fang, J. Yang, E. Ledesma and D.J. Tenney
Bristol-Myers Squibb Pharmaceutical Research Institute, Wallingford, CT, USA
ABSTRACT
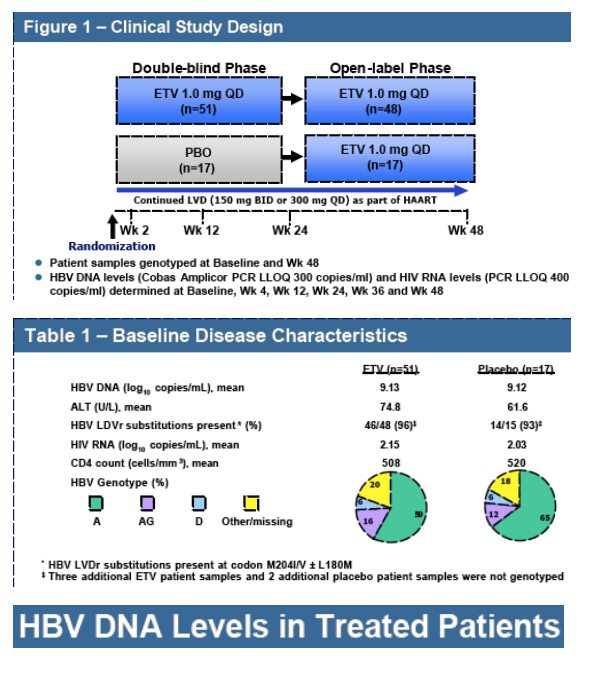
Background: Entecavir (ETV) is a potent and selective HBV inhibitor with no known intracellular interactions with other nucleoside/nucleotides. Resistance emergence has not been observed in nucleoside naive HBV patients treated with ETV for 2 years. Virologic rebounds due to ETV resistance (ETVr), observed in 1% of lamivudine (LVD) refractory patients treated with ETV at 1 year, required the pre-existence of LVD resistance (LVDr) substitutions (M204I/V +/ - L180M) and an additional substitution at HBV RT residues T184, S202 or M250. These additional substitutions are selected by LVD and were present at low frequencies at study entry. Since LVD is frequently used for prolonged periods in HIV/HBV co-infected patients, a double-blind comparative trial (AI463038) was conducted to determine the efficacy of 1.0 mg daily ETV in HIV/HBV co-infected patients
experiencing HBV viremia during therapy on LVD-containing HAART regimens.
Methods: The HBV RT gene was PCR amplified from serum DNA and sequenced at study entry and Week 48. Susceptibility assays on all emerging substitutions were conducted in HepG2 cells transfected with recombinant viruses.
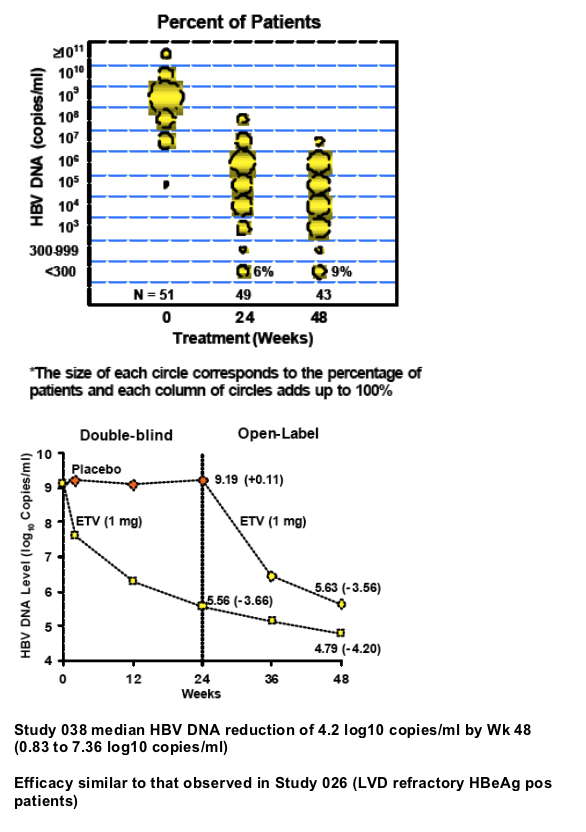
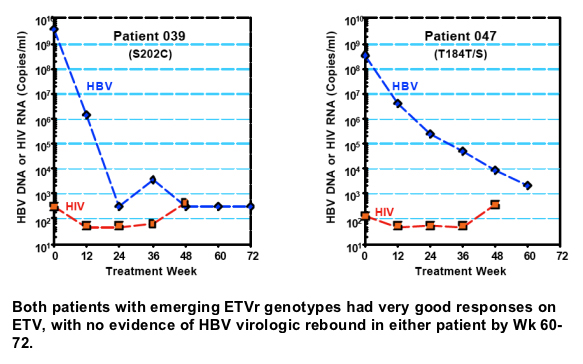
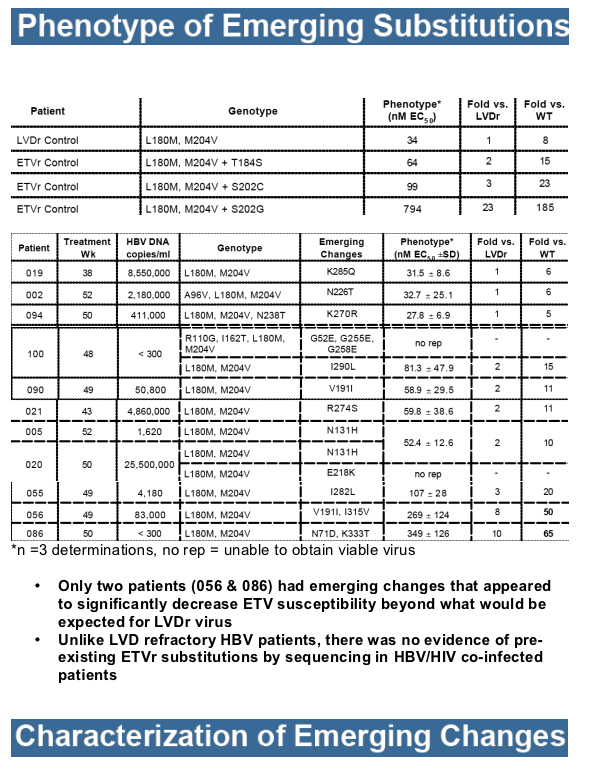
Results: At study entry, 46/48 (96%) of the samples sequenced had LVDr substitutions in the HBV RT, while none had ETVr substitutions (T184, S202 or M250) detectable by standard sequencing. The median ETV EC50 of these 48 baseline samples was 22.7 nM (range = 3.3 to 64.8 nM), within the EC50 range normally observed for LVDr HBV. The mean HBV DNA level at baseline was 9.13 log10 copies/ml, which was reduced 4.20 log10 copies/ml for the 43 patients that reached Week 48 of treatment. Nine percent of treated patients achieved HBV DNA levels <300 copies/ml and no patients exhibited a virologic rebound. Genotypic detection of known ETVr substitutions at residues T184S and S202C was evident in 2 patients at Week 48. Nineteen novel substitutions also emerged on ETV treatment, but none were correlated with reduced susceptibility to ETV in phenotypic assays. These evaluations are continuing.
Summary: HIV/HBV co-infected patients with LVD refractory HBV treated with ETV for 48 weeks experienced significant HBV DNA reductions and no virologic rebounds. Two patients experienced an enrichment of virus containing ETVr substitutions, although both showed a significant treatment response. ETV is a valuable treatment option for chronic HBV in patients co-infected with HIV.
Author Summary and Conclusions
ETV added to a HAART regimen is highly effective in reducing viral DNA levels in HBV/HIV co-infected patients harboring LVDr HBV.
Median viral DNA reduction of 4.2 log10 copies/ml achieved by Wk 48, with 84% of patients experiencing at least a 2-log drop by Week 24 and 9% showing reductions in HBV DNA levels to <300 copies by Wk 48.
There were no virologic rebounds evident during the first year of ETV treatment.
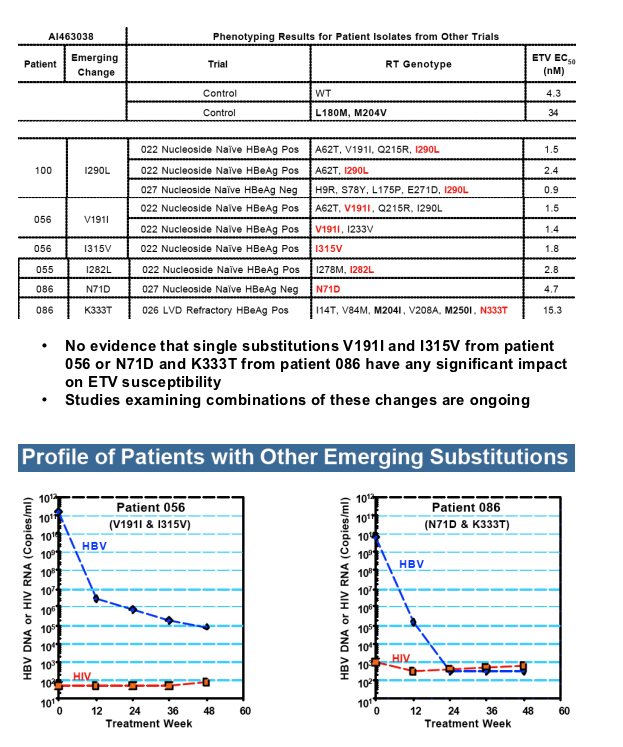
Only 2 patients showed genotypic evidence of emerging ETVr subst itutions at either residue T184 or S202, without evidence of virologic rebound or suboptimal response.
Emerging changes at other HBV RT residues remain under investigation, but do not appear to represent substitutions capable of yielding resistance to ETV in recombinant clones.
Unlike LVDr HBV patients, sequencing did not reveal pre-existing ETVr substitutions at residues T184, S202 or M250 in the baseline samples of co -infected patients.
Resistance Overview
Nucleoside Naive HBV Patients
No virologic rebounds due to resistance were observed following 2 years of ETV
therapy, likely due to the high genetic barrier to resistance created by:
- Intrinsic inhibitory potency against all 3 functional activities of viral polymerase
- High intracellular concentrations of ETV -TP
- Rapid and sustained suppression of viral replication
- Requirement of multiple substitutions to achieve meaningful resistance levels
No evidence that LVDr substitutions are selected on ETV therapy
LVD Refractory HBV Patients
LVDr substitutions result in an 8-fold decrease in ETV susceptibility and are a
prerequisite for virologic rebound due to ETV resistance.
Primary ETVr substitutions at RT residues T184, S202 and M250 only appear in
viruses containing LVDr substitutions, with these additional changes observed in 6% of LVD refractory patients at Wk 48.
Virologic rebounds due to resistance observed in 1% and 9% of LVD refractory
patients during the first and second years of ETV therapy, respectively, with ETVr
substitutions evident at baseline in many of these patients.


|
|
| |
| |
|
 |
 |
|
|