 |
 |
 |
| |
Pharmacokinetic/pharmacodynamic (PK/PD) analyses of TMC114 in the POWER 1 and POWER 2 trials in treatment-experienced HIV-infected patients
|
| |
| |
Reported by Jules Levin
13th CROI, Denver, Feb 5-8, 2006
Sekar V,1 De Meyer S,2 Vangeneugden T,2 Lefebvre E,1 De Pauw M,2 Van Baelen B,2 De Paepe E,2 de Bethune M-P,2 Hoetelmans R,2 Parys W1
1Tibotec Inc., Yardley, PA, USA; 2Tibotec BVBA, Mechelen, Belgium; poster 639b
Here we report the analysis that was conducted after 24 weeks of treatment to evaluate the relationship between:
1) TMC114 PK parameters and efficacy, PK and safety; and
2) TMC114 inhibitory quotient (IQ) and efficacy.
".....TMC114 PK was statistically significantly associated with changes in VL from baseline and virologic response at Week 24, however, TMC114 FC at baseline, baseline VL, and use of sensitive ARVs in the OBR were stronger predictors of response (Table 2)..."
AUTHOR CONCLUSIONS
PK/PD relationships showed that there was a strong relationship between TMC114 IQ and virologic response. See table 4 below.
The relationship between IQ and virologic response was driven primarily by the baseline TMC114 FC and less by TMC114 PK, as TMC114 PK showed a less significant association with response compared with the TMC114 FC.
PK/PD relationships support the selection of the TMC114/r 600/100mg bid dose for the treatment of HIV-1 infected patients with prior experience of treatment.
Relationships between PK and safety
Descriptive analyses showed no apparent relationship between TMC114 PK and rash, lipid AEs, cardiac and hepatic AEs or disorders of glucose metabolism. TMC114 PK was also not related to any psychiatric, nervous system or
gastrointestinal disorders.6
Results: The relationship between TMC114 PK parameters and efficacy (virologic and response parameters), though statistically significant was strongly influenced by baseline TMC114 fold change (FC), baseline VL and use of sensitive drugs in the OBR.
The IQ (uncorrected for protein binding) was the strongest predictor of virologic response, with the relationship primarily driven by baseline TMC114 FC. The relationship between TMC114 PK parameters and efficacy parameters was observed mainly in patients whose baseline TMC114 FC was between 4 and 240. There was no clear relationship between TMC114 PK parameters and efficacy parameters in patients with TMC114 FC values >40, nor in patients with TMC114 FC values 24: (note from Jules: presumably, because most patients are expected to have good response when FC<4 and poor response when FC>40).
IQ values of TMC114 were generally high (mean values >200; uncorrected for protein binding) and increased with dose, with the highest IQ values observed in the TMC114/r 600/100mg bid group. There was no apparent relationship between TMC114 PK parameters and safety measures.
Background: TMC114 is a HIV protease inhibitor (PI) that is potent against wild-type and PI-resistant HIV. TMC114 is administered with low-dose ritonavir (TMC114/r). POWER 1 (TMC114-C213) and POWER 2 (TMC114-C202) are ongoing randomized, controlled, Phase IIb studies that evaluated dose-response after 24 weeks in 3-class-experienced patients with 31 primary PI mutation. Four TMC114/r doses (400/100mg qd, 800/100mg qd, 400/100mg bid and 600/100mg bid) were compared with control PI regimens. An optimized background regimen (OBR) consisting of nucleoside reverse transcriptase inhibitors ± enfuvirtide was used in each treatment arm.
Methods: Blood sampling was performed in 468 patients randomized to the TMC114/r groups to determine TMC114 pharmacokinetic (PK) parameters area under the curve (AUC24h) and trough concentration (C0h). At Week 24, PK/PD relationships were assessed for TMC114 using analysis of covariance models. Efficacy PD measures tested in the model included change in log10 viral load (VL) from baseline and proportion of patients with 31.0 log10 decrease in VL. Inhibitory quotients (IQs), the ratio between steady-state TMC114 trough concentration and baseline TMC114 EC50 were also related to the efficacy PD measures. Relationships between TMC114 PK and safety parameters were investigated using descriptive methods.
Conclusions: PK/PD relationships for TMC114 provide evidence to support the recommendation of the 600/100mg bid dosing regimen in treatment-experienced HIV-1 infected patients.
INTRODUCTION
TMC114 is an investigational PI that exhibits potent in-vitro antiviral activity against wild-type and resistant HIV isolates, including multidrug-resistant strains.1
TMC114 has been developed in combination with low-dose ritonavir (TMC114/r).
Results of the 24-week primary analysis of the ongoing POWER 1 and 2, randomized, controlled trials evaluating the efficacy and safety of TMC114/r in treatment-experienced HIV patients, demonstrated that TMC114/r significantly
improved efficacy outcomes compared with investigator-selected control PIs (in combination with an OBR) and was generally safe and well tolerated.2-5
In POWER 1 and 2, the dose-response relationship of efficacy between different TMC114/r treatment groups (400/100mg qd, 800/100mg qd, 400/100mg bid and 600/100mg bid) after 24 weeks of treatment was also evaluated.
All patients receiving TMC114/r were subsequently switched to the recommended dose of 600/100mg bid.
RESULTS
Patient characteristics
The baseline characteristics of the integrated population (POWER 1 and 2) for the primary 24-week analysis were well balanced between treatment groups.2-5 The majority of patients were Caucasian (73%) and male (88%).
For POWER 1 and 2, the mean baseline viral load was 4.66 log10 and 4.48 log10 copies/mL, and median CD4 cell count was 106 and 179 cells/mm3, respectively.
There was a higher median baseline TMC114 FC in POWER 2 (4.9 vs 3.5) compared with POWER 1.
PK
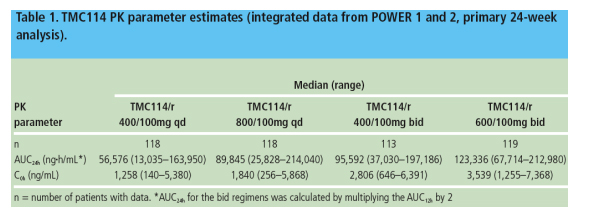
TMC114 PK parameters were available for 468 patients and are summarized in Table 1
- TMC114 exposure (AUC24h) increased less than dose proportionally; the 100% and 50% increase in TMC114 dose between the two qd and the two bid groups resulted in an approximate 60% and 29% increase in median exposure,
respectively.
Relationship between PK and efficacy
There was a weak relationship between TMC114 PK and efficacy (change in log10 VL from baseline at Week 24)
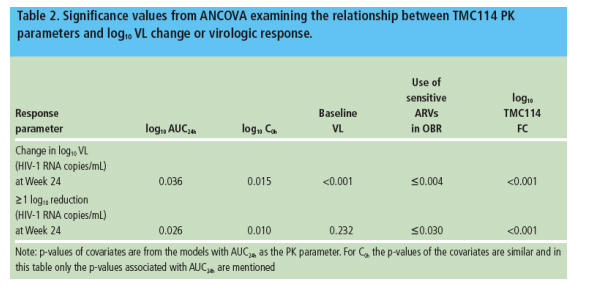
- TMC114 PK was statistically significantly associated with changes in VL from baseline and virologic response at Week 24, however, TMC114 FC at baseline, baseline VL, and use of sensitive ARVs in the OBR were stronger predictors of response (Table 2)
The observed relationship between exposure and response was primarily due to patients whose baseline TMC114 FC was 4 to 240 (Figure 1)
--there was no clear relationship between exposure and response in patients with TMC114 FC values >40 or 24
--even in this advanced population, only a small subset of patients had FC >40 (n=41).
IQ
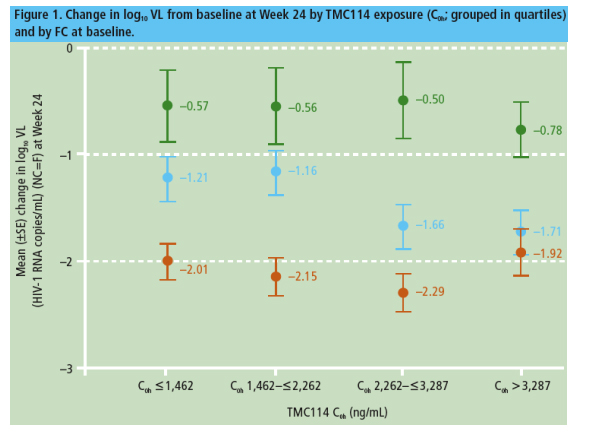
A substantial variability in IQ values was observed. This was mainly driven by the variability in baseline TMC114 EC50 values and less so by the variability in the PK parameters.
Values for IQ were high (mean >200 [uncorrected for protein binding] and >10 [corrected for 95% protein binding] with TMC114) and increased with increasing doses of TMC114/r (Table 3). Based on these IQ values, all dosing regimens of TMC114/r would be expected to be virologically active, with the most pronounced effects for the highest dose (600/100mg bid). This concurs with the integrated analysis of efficacy for these two studies.6
Relationships between IQ and efficacy
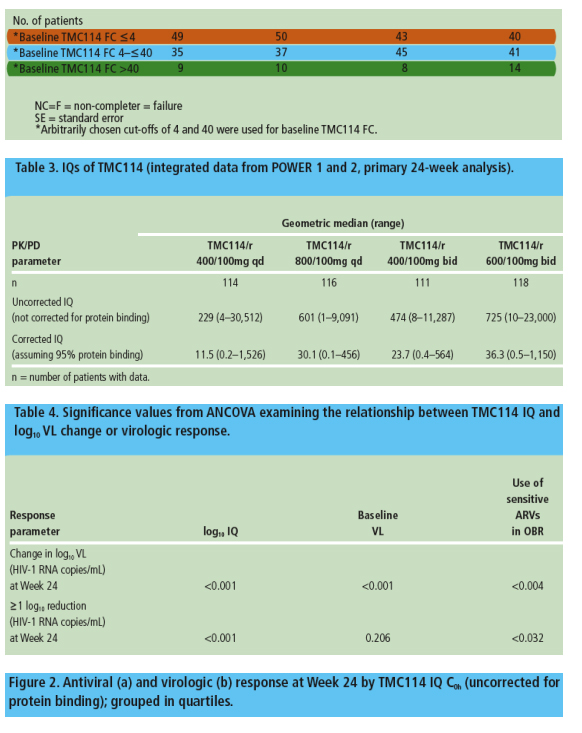
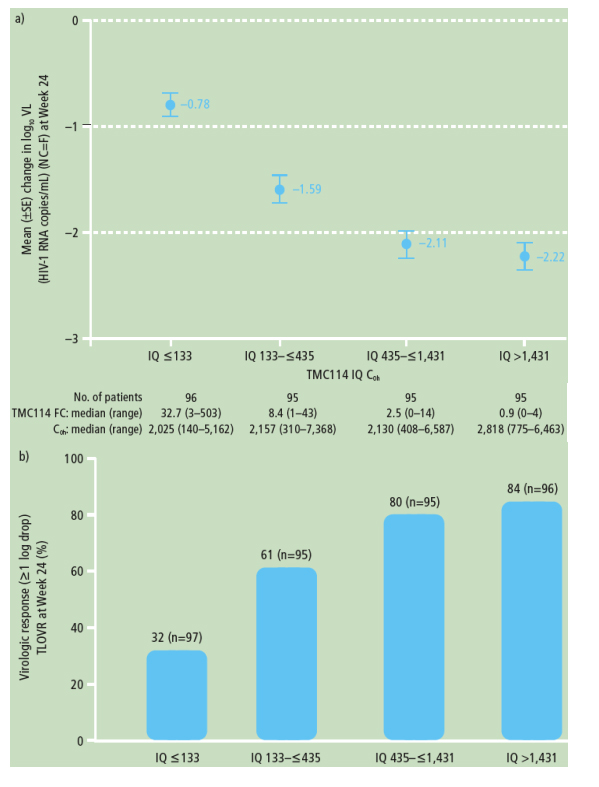
The IQ was a strong predictor of virologic response, with the relationship being primarily driven by the baseline TMC114 FC and less so by TMC114 exposure
- ANCOVA models showed that IQ was significantly and positively associated with change in log10 VL and virologic response at Week 24 (Table 4)
- higher values for IQ were significantly and positively associated with a higher probability of response, with the
relationship being driven by the baseline TMC114 FC (Figures 2a and 2b)
- the highest IQ values were observed in the TMC114/r 600/100mg bid group
- baseline VL and use of sensitive ARVs in the OBR were also significantly associated with the probability of response.
References
1. De Meyer S, et al. Antimicrob Agents Chemother 2005;49:2314-21.
2. Katlama C, et al. 3rd International AIDS Society (IAS) Conference on HIV Pathogenesis and Treatment, 24-27 July, 2005; Rio de Janeiro, Brazil. Abstract WeOaLB0102.
3. Wilkin T, et al. 45th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC), 16-19 December 2005;Washington, DC, USA. Abstract 2860.
4. Grinsztejn B, et al. 3rd International AIDS Society (IAS) Conference on HIV Pathogenesis and Treatment, 24-27 July, 2005; Rio de Janeiro, Brazil. Abstract WePeLB6.201.
5. Berger D, et al. 45th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC), 16-19 December 2005;Washington, DC, USA. Abstract H-1094.
6. Tibotec BVBA, data on file.
|
|
| |
| |
|
 |
 |
|
|