 |
 |
 |
| |
Is 24-week HCV therapy useful in coinfection?
|
| |
| |
Reported by Jules Levin
13th CROI
Feb 5-8, 2006
Denver
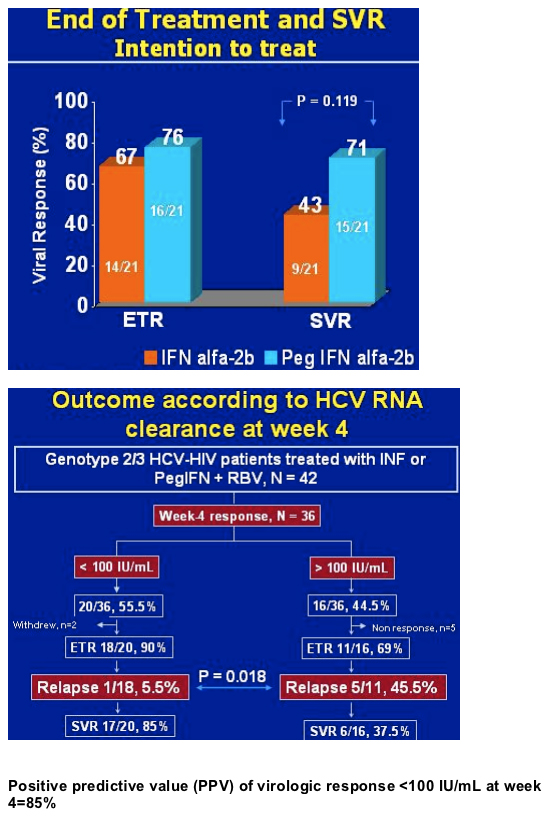
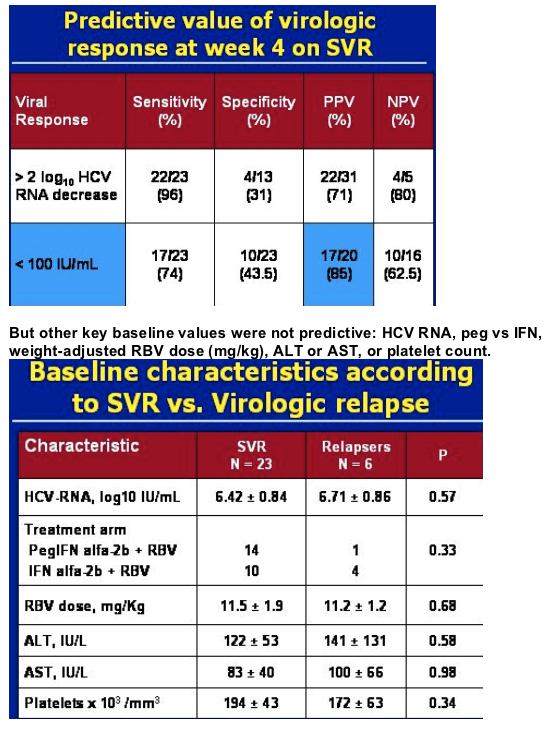
.....42 genotype 3 patients were randomized to in this study at one center in Spain. 36 patients were evaluable at week 4. 20/36 (55.5%) had <100 IU/mL at week 4. 90% (18/20) had ETR, early treatment response. 85% (17/20) of these patients treated with IFN or peginterferon plus 800 mg of ribavirin for 24 weeks had <100 IU/mL at week 4 in this study achieved SVR. Only 5.5% (1/18) had relapse. Of patients with >100 IU/ml at week 4 [44% (16/36); 5 non-responders], 69% (11/16) had ETR, 5/11 (45%) relapsed, and the SVR for this group was 37% (6/16). Crespo said the week 4 virologic response was a strong predictor: 85% PPV, positive predictive value. Other important baseline characteristics were not predictive, see table below.....
The study author Manual Crespo said limitations of this study were it was a retrospective analysis with a small number of patients. Since liver biopsies were not available we can't evaluate the impact of cirrhosis on the predictive accuracy of the early virologic response. Crespo concluded: very early responders, around 55% in our study, are at low risk for virologic relapse after a 24-week schedule of combination therapy. The reduced exposure to treatment will probably translate into a better benefit-risk ratio among coinfected patients. The utility of the 4-week virologic response as a guide to taolir the duration of treatment for genotype 2 or 3 chronic HCV in HIV-coinfected patients can only be established through randomized trials.
"Utility of Early Viral Response To Individually Adjust the Duration of treatment for Genotype 2/3 HCV in HCV/HIV Coinfected Patients"

In an oral talk at the 13th CROI Manuel Crespo presented study results finding that 24-week peginterferon + ribavirin therapy was adequate in genotype 3 patients in HCV RNA was <100 IU/mL at week 4. Of course these data, if confirmed, are of limited value because in the USA 70-80% of coinfected patients are genotype 1. However, the study underscores the values of testing HCV RNA at week 4 and that a good response at week 4 can be useful in predicting outcome to therapy. In a similar study reported by Doug Dieterich at CROI, for genotype 1 patients taking Pegasys + RBV who had <50 IU/mL at week 4, 82% achieved SVR with 48 weeks therapy. Thus, testing HCV RNA at week 4 can be useful in providing incentive to patients and care providers.
Crespo maintained some patients are overtreated & the duration of therapy can be shortened for some patients, if we can identify a way to do this. In this table just below he outlines studies where the relapse rate is higher when coinfected patients were treated for 24 weeks rather than 48 weeks. So the objective of Crespo's study was to assess the utility of week 4 virologic response as a guide to individiually adjust the duration of treatment for genotype 2/3 chronic HCV in HIV-coinfected patients.

ELIGIBILITY CRITERIA
Age: 18-60 yrs
ALT >ULN
CD4 count >200
HIV RNA <50 c/ml
Patients were excluded for cirrhosis decompensation & contraindication to IFN or RBV.
TRIAL DESIGN
42 patients, genotype 2/3 were randomized to IFN 2-2b 3 MIU 3x/wk + RBV 800 mg/d (n=21) or PegIFN a-2b 1.5 ug/kg/w + RBV 800 mg/d (n=21). Patients were treated for 24 weeks & there was a 24-week follow-up period.
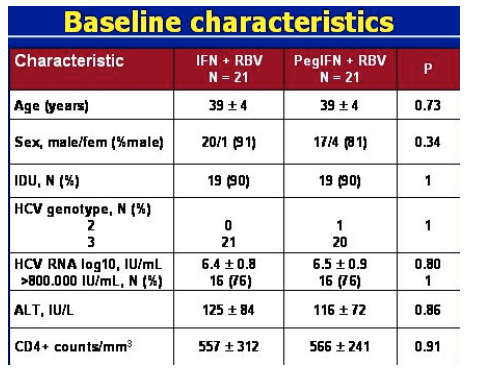
BASELINE CHARACTERISTICS
There was no difference between the IFN & PegIFN groups in these characteristics outlined in the table below. Of note, 90% of patients were IDUs, all patients but 1 were genotype 3, 75% of patients had high HCV RNA (>800,000 IU/mL), average CD4 count was 550.
RESULTS
This slide was presented at AASLD in Nov 2005. I think this slide refers to all patients regardless of genotype showing that genotype 1 patients responded better to pegIFN than standard IFN and that for genotype 1 patients 24 week therapy regardless of 4-week response may not be useful.
|
|
| |
| |
|
 |
 |
|
|