 |
 |
 |
| |
Pegasys Rapid Response at Week 4 in APRICOT Yields 75-82% SVR in Genotype 1 Patients
|
| |
| |
Reported by Jules Levin
13th CROI
Denver, Feb 5-8, 2006
Note from Jules: there wasn't much noteworthy at CROI on HCV. Apparently, HIV researchers often feel they have to repeat studies already conducted in HCV monotherapy, reinvent the wheel. I will however provide a report on HCV studies at CROI. Here are some brief notes on studies and information reported at the 13th CROI on HCV coinfection. The results of one study is worth repeating here, that insulin resistance reduces HCV therapy response rates. We knew this from HCV monoinfection studies, many of these have been conducted over the years. A second study by Pablo Tebas and Bill Powderly found that fatty liver was not associated with HAART. Fatty liver is an important condition because it can cause fibrosis & cirrhosis, and it can reduce response to HCV therapy just as insulin resistance. In fact insulin resistance & diabetes are associated as a risk factor for fatty liver, and so are elevated lipids. Therefore, if you conduct the study in the correct population of HIV+ individuals I think there is little doubt you will find fatty liver more prevalent: examine the prevalence of fatty liver in patients on ART with abnormalities in glucose and lipids and other risk factors. A care & treatment policy for HCV/HIV coinfection should include evaluation of depression and fatty liver and if found treatment intervention considered prior to peginterferon/ribavirin therapy. Anti-depressant therapy is utilized either prophylactically before peg/RBV or if depression emerges after therapy is initiated.
Pablo Barreiro reported for a group from Spain in a study that suggests fibrosis was reversed in HCV/HIV coinfected patients with SVR (sustained viral response). This study was very poorly done & not much at all can be taken from the poster. The author did not report pre-HCV therapy & post HCV therapy fibrosis tests; he only reported post-therapy FibroScan results. First, without a baseline fibrosis evaluation to compare to a post-therapy evaluation you cannot evaluate changes in fibrosis. Second, non-invasive fibrosis evaluation tests including FibroScan are not reliable & not yet proven to provide accurate fibrosis evaluations. It is felt that perhaps some of the the non-invasive test might provide accuracy in evaluating stage 1 or cirrhosis, stage 4, but that in-between stages are not reliably evaluated. Still, even these opinions need further confirmation. At this time their use is of limited benefit, but might provide some utility. Still, additional research is needed to examine their reliability & how they might be used. Nonetheless, this study merely provides a suggestion that the patients with an SVR had an omproved fibrosis. There have been, however, other well done studies in coinfected and monoinfected patients that do find that patients who achieve an SVR can improve fibrosis. At CROI two studies were reported finding increased sexual transmission of HCV among men who have sex with men in London & in Amsterdam (abstracts 86 & 87). Reports such as these have been coming out of western Europe for several years. No reports of this nature, however, have come from the USA. Why, I don't know, are sexual practices different or are US researchers & care providers not showing any attention to this? As well, at CROi there was a poster from France finding heterosexual transmission of HCV to women. They inquired about all possible routes of transmission and the authors concluded that unsafe sex was the only identified route for transmission (abstract 843). The report noted an increase in sexual transmission since 2002, as noted among MSM from other reports from Europe have also foi=und increased sexual transmission in recent years.
"Sustained virological response in HIV-HCV co-infected patients with HCV genotype 1 infection who have a rapid virological response at week 4 of treatment with peginterferon alfa-2a (40KD) (PEGASYS ) plus ribavirin (COPEGUS ): AIDS PEGASYS Ribavirin International Co-infection Trial (APRICOT)"
D.T. Dieterich,1 F. Duff,2 M. Sulkowski,3 F.J. Torriani,4 E. Lissen,5 N. Brau,6 M. Rodriguez-Torres7
1Mt Sinai School of Medicine, New York, NY, USA; 2Roche, Nutley, NJ, USA; 3Johns Hopkins University, Baltimore, MD, USA; 4University of California, San Diego, CA, USA; 5Virgen del Rocio University Hospital, Seville, Spain;
6Bronx VA Medical Center, Bronx, NY, USA; 7Fundacion de Investigacion de Diego, Santurce, and Ponce School of Medicine, Ponce, Puerto Rico
Qualitative HCV RNA testing at week 4 can identify rapid responders with a high probability of achieving a sustained viral response (SVR) & provide incentive to patients. The message from this analysis is that patients should test HCV viral load at week 4. If there is a rapid viral load response (<50 IU/mL) there is a high probability (75-83%) of achieving SVR. Knowledge of a good response at week 4 can help patients feel confident and provide incentive to continue therapy, particularly in hard-to-treat patients.
The combination of a pegylated interferon plus ribavirin is the treatment of choice for chronic hepatitis C in patients with human immunodeficiency virus/hepatitis C virus (HIV-HCV) co-infection.[1,2]
In APRICOT, a large, randomized, international, phase III study, overall sustained virological response (SVR) rates were significantly higher
with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) than peginterferon alfa-2a (40KD) plus placebo or conventional interferon plus ribavirin (Table 1). Among genotype 1 patients with a low baseline
HCV RNA level, defined as L800 000 IU/mL, peginterferon alfa-2a (40KD) plus ribavirin produced an SVR rate of 61%, which was similar to that achieved with this combination in genotype 2 or 3 patients with either a low (61%, 17/28) or high baseline HCV RNA level (63%, 42/67).[3]
In genotype 1 patients with HCV mono-infection, achievement of a rapid virological response (RVR; undetectable HCV RNA [<50 IU/mL] at week 4 of treatment) with peginterferon alfa-2a (40KD) plus ribavirin, resulted in an overall
SVR rate of 85%, regardless of baseline HCV RNA level.[4] We studied the effect of an RVR on the probability of an SVR in HCV genotype 1 patients co-infected with HIV who received the combination of peginterferon alfa-2a (40KD) plus ribavirin in APRICOT.
AUTHOR CONCLUSIONS
In APRICOT, achievement of an RVR at week 4 of treatment with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS ) was associated with an 82% SVR rate in HCV genotype 1 patients.[5] This response rate was similar to the response rate (14/16 patients, 88%) in patients with HCV genotype non-1 infection in APRICOT who achieved a week 4 RVR following treatment with peginterferon alfa-2a (40KD) plus ribavirin.
A higher proportion of patients with a low baseline HCV RNA level (L800 000 IU/mL) had an RVR than those with a high baseline HCV RNA level (>800 000 IU/mL); however, among those with an RVR, relapse was uncommon. These results are consistent with recently reported findings in patients with HCV
mono-infection treated with peginterferon alfa-2a (40KD) plus ribavirin.[4]
Qualitative measurement of serum HCV RNA at week 4 is useful because it can identify patients with a very high probability of achieving an SVR. Such information may be useful in motivating patients to adhere to therapy.

RESULTS
A total of 289 patients, including 176 individuals with HCV genotype 1 infection,
were randomized to peginterferon alfa-2a (40KD) plus ribavirin and received at least one dose of study medication.
Among the 176 patients with HCV genotype 1 infection, 46 individuals (26%) had a low baseline HCV RNA level (<800 000 IU/mL), and 130 individuals (74%) had a high baseline HCV RNA level (>800 000 IU/mL).
Week 4 response data
Four patients did not have a 4-week measurement recorded, resulting in a total of 172 HCV genotype 1 patients with week 4 HCV RNA data.
Of these 172 patients, 22 had an RVR (13%). Eighteen of the 22 patients who achieved an RVR had a low baseline HCV RNA level.
The SVR rate in HCV genotype 1 patients who had an RVR was 82% (Figure 1a).
Although HCV genotype 1 patients with a low baseline HCV RNA level (18 of 46, 39%) were more likely to achieve an RVR than patients with a high baseline HCV RNA level (4 of 126, 3%), the probability of achieving an SVR was similar once an RVR was attained (83% vs 75%; Figure 1a).
Only one patient with an RVR relapsed during follow-up.
In HCV genotype 1 patients without an RVR, the subset of patients with a high baseline HCV RNA level had a lower probability of achieving an SVR compared with those patients who had a low baseline HCV RNA level (Figure 1b).
Figure 1. End-of-treatment and SVR rates in patients
a) with, and b) without an RVR at week 4 of treatment with
peginterferon alfa-2a (40KD) plus ribavirin in APRICOT.
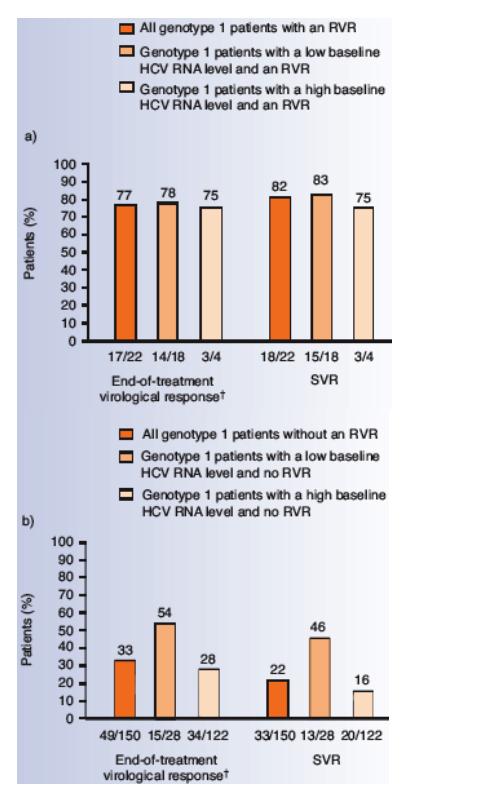
Four patients with high baseline HCV RNA levels did not have a
4-week measurement recorded.
†As a result of the short time window used for end-of-treatment
response, in APRICOT, several patients who had no end-oftreatment
response had an SVR response.
METHODS
Patients
HIV-HCV co-infected adults eligible for APRICOT had quantifiable serum HCV RNA (>600 IU/mL), elevated serum alanine aminotransferase (ALT) levels, compensated liver disease and stable HIV disease. Stable HIV disease was defined as a CD4+ cell count 3200 cells/_L (100–199 cells/_L if the HIV-1 RNA level was <5000 copies/mL) and the absence of opportunistic infections or malignancies.
Patients were required either to have been on stable antiretroviral therapy (ART) for at least 6 weeks before enrollment, with no changes to the regimen expected during the first 8 weeks of treatment, or not to have received ART for at least 8 weeks prior to randomization and to be able to delay ART for at least 6 weeks after initiation of treatment. Patients were excluded if they had previously received interferon or ribavirin.
Study design
The complete study design and primary results of APRICOT have been published.[3] Briefly, patients were randomized to 48 weeks of treatment with peginterferon alfa-2a (40KD) 180 _g/week plus either ribavirin 800 mg/day
or placebo, or to conventional interferon alfa-2a 3 MIU three times per week plus ribavirin 800 mg/day. SVR, the primary efficacy outcome in the trial, was defined as undetectable HCV RNA by qualitative polymerase chain reaction (PCR)
assay (COBAS AMPLICOR HCV Test v2.0, limit of detection 50 IU/mL) at the end of a 24-week untreated follow-up period (week 72).
For the purposes of this analysis, RVR was defined as undetectable HCV RNA
(<50 IU/mL) by qualitative PCR at week 4.
This analysis is based on data from patients treated with peginterferon alfa-2a (40KD) plus ribavirin.
|
|
| |
| |
|
 |
 |
|
|