 |
 |
 |
| |
Abacavir vs Nevirapine, Safety 1st-Line ART in Uganda
|
| |
| |
"Safety of Nevirapine Compared to Abacavir on a Background of Zidovudine/Lamivudine as First-line Antiretroviral Therapy:
a Randomised Double-Blind Trial conducted in Uganda"
These study results were reported at the 13th CROI by P Munderi
on behalf of the DART Trial Team.
AUTHOR CONCLUSIONS
In Ugandan patients with low CD4 counts initiating ART with CBV/NVP or CBV/ABC
-- a trend towards a lower rate of SARs with ABC
-- a lower discontinuation rate with ABC
-- a lower rate of any grade 4 AE with ABC
Considerable overlap in clinical manifestations of NVP and ABC reactions
Rate of ABC HSR in this population is 2%
Ongoing assessment of
-- genetic polymorphisms
-- virological and immunological efficacy
BACKGROUND
Standard 1st line regimen in Africa is 2NRTI/NNRTI
-- Nevirapine (NVP) is the most frequently used NNRTI
Consideration of an Abacavir based regimen in Africa remains warranted
-- High rates of TB co-infection
-- Initiation of treatment in women
High rates of HSR may limit use of Abacavir (ABC)
-- In Africa HSR may be difficult to distinguish from intercurrent infections, for example malaria
-- No randomised trials have reported on toxicity of nevirapine and abacavir in Africa
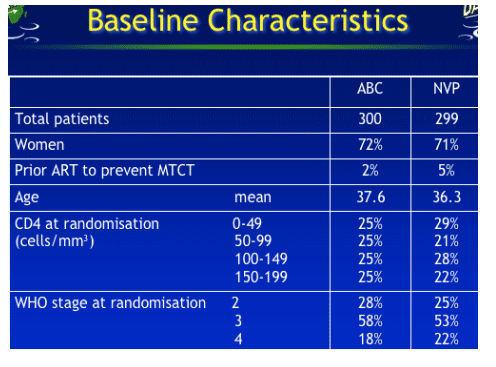
NORA Trial Design
-- a randomised, double-blind, 24 week, phase II trial at
2 centres in Uganda.
-- evaluating the safety of Nevirapine OR Abacavir [NORA]
-- a substudy of the DART Trial
-- 600 ARV naïve adults with symptomatic HIV infection
CD4<200 cells/mm3 and no contraindications to ART
were randomised in a 1:1 ratio, to receive :
zidovudine/lamivudine (Combivir) twice daily, plus
-- 300 mg ABC and nevirapine placebo twice daily
-- 200 mg NVP and abacavir placebo twice daily
ENDPOINTS
Primary: SAE as defined by ICH-GCP definitely/probably or uncertainly related to blinded nevirapine/abacavir (SAR),
-- independently adjudicated by clinicians blind to randomised allocation
-- Secondary: AE of any grade leading to permanent discontinuation of blinded nevirapine/abacavir
-- Secondary: Grade 4 events irrespective of whether or not they resulted in nevirapine/abacavir discontinuation
OUTCOME
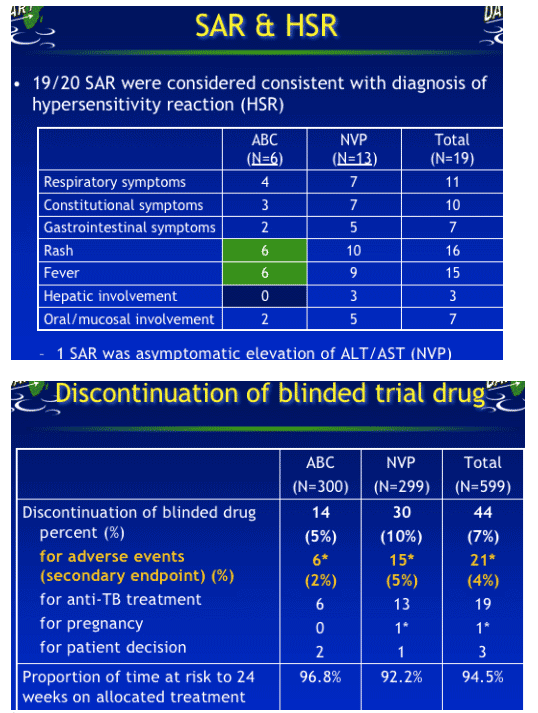
34 SAEs occurred on blinded drug in 33 patients
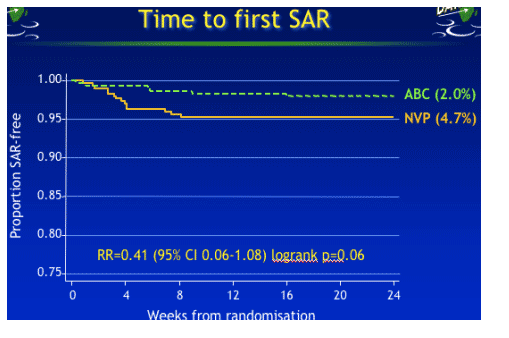
Primary endpoint: 20 SAEs in 20 patients were definitely/possibly related to blinded abacavir/nevirapine and classified as Serious Adverse Reactions (SAR)
-- 6 (2.0%) on ABC
-- 14 (4.7%) on NVP p=0.06
14 SAEs considered unlikely to be related to blinded drug were anaemia (n=7), pancytopenia, death from unknown cause, head trauma, DVT, duodenal ulcer/haematemesis, fever*, rash**
* rabies vaccination
** open label NVP following discontinuation of blinded ABC for HSR
* patients and clinicians were unblinded
exact p=0.05
exact p=0.05
Secondary endpoint: Grade 4 AE
- 187 (78 ABC, 109 NVP) Grade 4 AEs in 155 patients (64 ABC, 91 NVP) on blinded drug
-- 59 per 100 PY at risk in ABC
-- 88 per 100 PY at risk in NVP p=0.008
- Majority were haematological
-- greatest difference in neutropenia: 46 ABC, 71 NVP
-- anaemia: 17 ABC, 16 NVP
d4T substituted for ZDV in 45 patients, (22 ABC, 23 NVP)
- 30 (19%) adverse reactions to blinded trial drugs
- 8 patients (all NVP) had Grade 4 elevations in LFTs
|
|
| |
| |
|
 |
 |
|
|