 |
 |
 |
| |
Efficacy and safety of Atazanavir With & Without Ritonavir in Antiretroviral-Naive Subjects
|
| |
| |
Reported by Jules Levin
13th CROI (Retrovirus Conference)
Denver, Feb 5-8, 2006
Author summary: ATV/r 300/100 (86%) was non-inferior to ATV 400 (85%), HIV RNA <400 copies/ml. The viral failure rate was lower in the ATV/r than the ATV group (3 vs 10 patients). No PI resistance was seen in subjects with viral failure in the boosted Reyataz group. One subject with viral failure on ATV developed phenotypic resistance to ATV with an I50L mutation. Subjects on ATV without RTV had a lower rate of hyperbilirubinemia. More subjects discontinued due to adverse events on the ATV/r (8 vs 1). Lipid changes were greater in the ATV/r group and were generally not associated with treatment intervention or discontinuations. See data below.
Niel Malan reported 48-week results from a 96-week study comparing boosted Reyataz with unboosted Reyataz. The primary endpoint of the study is the proportion of subjects with HIV RNA <400 copies/ml (non-inferiority). Secondary study endpoints include the proportion of subjects with <50 c/ml and laboratory abnormalities and lipid changes.
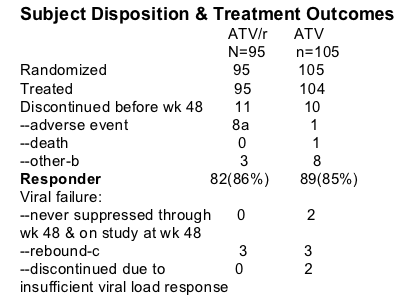
200 patients were randomized to Reyataz (300mg) boosted by 100mg ritonavir or Reyataz 400 mg. study patients were stratified by <100,000> c/ml. The primary study analysis was at week 48.
a-4 subjects (4%) discontinued due to hyperbilirubinemia (per protocol for
persistent grade 4 hyperbilirubinemia).
b-includes lack of efficacy, lost to followup, and no longer met study criteria
c-viral rebound defined as confirmed on-study HIV RNA >400 c/ml, or last
on-study HIV RNA .400 c/ml followed by discontinuation.
BASELINE CHARACTERISTICS
Baseline HIV RNA (median): 4.83 log (ATV/r); 5.02 log (ATV)
Cd4 count (median): 201 (ATV/r); 194 (ATV
CD4 count <200: 48% (ATV/r); 52% (ATV)
HBV or HCV coinfection: 18% (ATV/r); 19% (ATV)
70-73% men
age: 34-35 yrs
white: 53-57%
black: 26%
HIV RNA <400 c/ml and <50 c/ml through week 48 (ITT)
ATV/r: 86% <400 c/ml
ATV: 85% <400 c/ml
Difference estimate (95% CI): 1.5(-8.2, 11.1)
ATV/r: 75% <50 c/ml
ATV: 70% <50 c/ml
Difference estimate (95% CI): 5.0 (-7.0, 17.0)
CD4 increase was 224 for ATV group & 189 for ATV/r group.
Time-averaged difference estimate (95%): -21.1 (-48.9, 6.6)
LIPID CHANGES to Week 48
Total Cholesterol
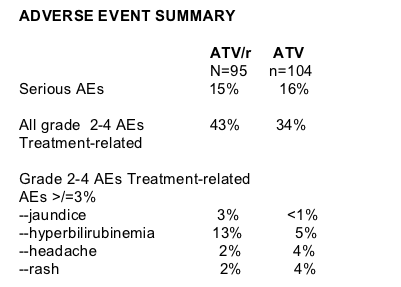
Total cholesterol increased in both groups from baseline to week 48 but increased more in the ATV/r group compared to the ATV group (15% vs 6%), but for both groups mean TC remained well below 200 mg/dL.
Mean TC was about 155 mg/dL in ATV/r group at baseline and increased 15%, while mean TC was about 160 mg/dL at baseline for ATV group and increased 6% (15% vs 6%, p=0.0034). So, TC remained well below 200 mg/dL in both groups at week 48.
Fasting LDL-C
Mean fasting LDL-C increased by 23% in the ATV/r group & by 16% in the ATV group, but the difference in change between ATV/r & ATV was not significant (p=0.21). Mean LDL was about 95 mg/dL at baseline and increased 23% in the ATV/r group, and LDL was about 95 mg/dL in the ATV & increased by 16%. Fasting LDL-C remained below 130 mg/dL in both groups.
HDL-C
Mean HDL-C was about 40 mg/dL at baseline in the ATV/r group & increased by about 30% in the ATV/r group, and HDL-C was about 40 mg/dL in the ATV group and increased by 29%, the difference in change between the groups was not significant (p=0.69).
Fasting TG increased by 26% in the ATV/r group after 48 weeks from about 140 mg/dL at baseline, and fasting TG decreased by 3% in the ATV group from about a50 mg/dL at baseline. The difference in change between ATV?r & ATV was significant, p=0.0004).
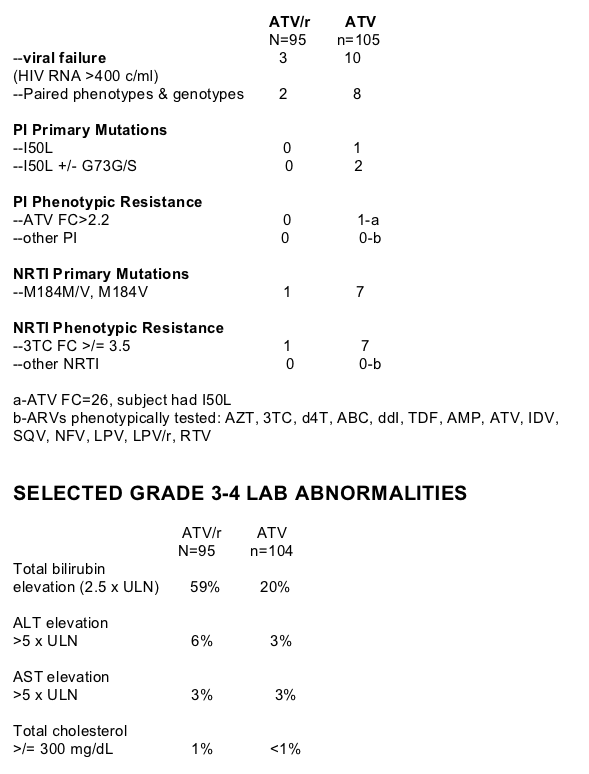
TREATMENT-EMERGENT RESISTANCE
Of note no primary PI resistance mutations developed among the 3 viral failures in the ATV/r group, and among the 10 viral failures in the ATV group 1 patient had I50L and 2 patients had I50L +/- G73G/S. Similarly regarding NRTI mutations, 1 viral failure study participant in the ATV/r group developed 3TC resistance while 7 viral failure study participants in the ATV group developed NRTI (3TC) resistance.
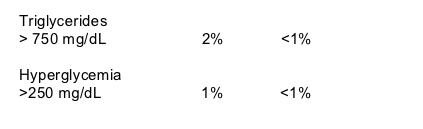
|
|
| |
| |
|
 |
 |
|
|