 |
 |
 |
| |
VX-950 HCV Protease Inhibitor + Pegasys/RBV: 28 days study results in treatment-naive
|
| |
| |
Reported by Jules Levin
DDW, May 23, 2006, Los Angeles
I'm reporting to you in "Real-Time" as many of my key conference reports are, immediately following the final oral session on hepatitis C at DDW. This abstract was reported late for presentation at this meeting, as Vertex just completed gathering this data, so the presentation was reported in the AGA Late Breaker session rather than an AASLD session. So I had to listen to 1 _ hrs of anti-constipation drugs and drugs for Crohns Disease until getting to this talk; and I'm glad to be heading home in the morning. This meeting was at the LA Convention Center, which is downtown and uninteresting. Jules Levin
"28 Days of the Hepatitis C protease Inhibitor VX-950, in Combination with Peginterferon-alfa-2a (Pegasys) and Ribavirin, is Well-tolerated and Demonstrates robust Antiviral Effects"
Lawitz E1, Rodriguez-Torres M2, Muir A3, Keane J4, Kieffer T4, McNair L4, McHutchison, J3
1Alamo Medical Research, san Antonio, TX
2Fundacion de Investigation de Diego, santuce, PR
3Duke Clinical research Institute, Durham, NC
4 Vertex Pharmaceuticals, Cambridge, MA
VX-950 Background
- One of a new class of Specifically Targeted Antiviral Therapies for HCV (STAT-C)
- Reversible tight binding to HCV protease active site with slow off-rate
- Demonstrated good tolerability and rapid and dramatic antiviral activity as a single agent in a 14-day study, and in a 14-day study with pegIFN.
VX05-950-102 Clinical study Design
Objectives:
1- to assess the safety of 28 days of dosing with VX-950, pegIFN, and RBV in combination
2- to examine antiviral effects and pharmacokinetics of VX-950, PegIFN, and RBV in combination
Study dosing period was 28 days.
Study population:
- 12 subjects
- genotype 1 (all subtypes)
- treatment-naive
VX-950 was dosed as a tablet (750 mg every 8 hrs, q8h), RBV at 1000 or 1200 mg daily, and Peg-IFn at 180 ug weekly.
At the completion of study dosing, off-study standard therapy (PegIFN and RBV) was provided for all subjects.
Data presented today is based on an end-of-dosing interim analysis but includes clinical followup data available at this time:
- includes clinical safety and antiviral activity data
- includes viral sequencing data during 28-day dosing period
- pharmacokinetics analyses still in progress
Baseline Status for Subjects
Male/female: 6/6
Race:
Caucasian/Hispanic/other: 1/10/1
Median age: 43.7 yrs
Median weight: 78.7 kg
Median baseline HCV RNA (log10): 6.5 iu/mL
Median duration of infection (n=7): 20.5 yrs
Stage of liver fibrosis (biopsy within 2 yrs)
- No/mild fibrosis: 6
- Portal fibrosis: 4
- Bridging fibrosis: 2
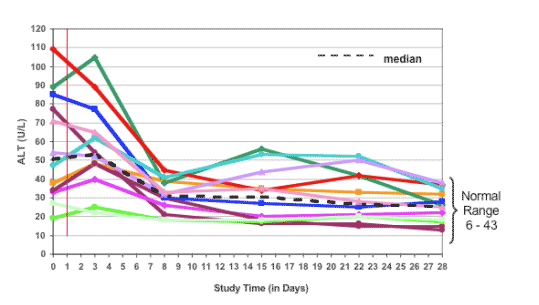
VX-950/PegIFN/RBV was Well-tolerated
no serious adverse events or discontinuations for adverse events
no PegIFN or RBV dose reductions during 28 day period
compliance with dosing regimen was excellent:
- 98-100% compliance with VX-950 dosing
- 94-100% compliance with RBV dosing
- 100% compliance with PegIFN dosing
Most Common Related Adverse Events
(includes all AEs recorded by investigators as possibly, probably or definitely related to the study drug combination)
Most of the events appeared to be related to Peg/RBV and the rates appeared to be in-line with standard effects seen in studies with Peg/RBV:
Flu-like illness: 50%
Fatigue: 41%
Headache: 41%
Nausea: 41%
Anemia: 25%
Pruritis: 25%
Depression: 17%
Insomnia: 17%
Pruritic rash: 17%
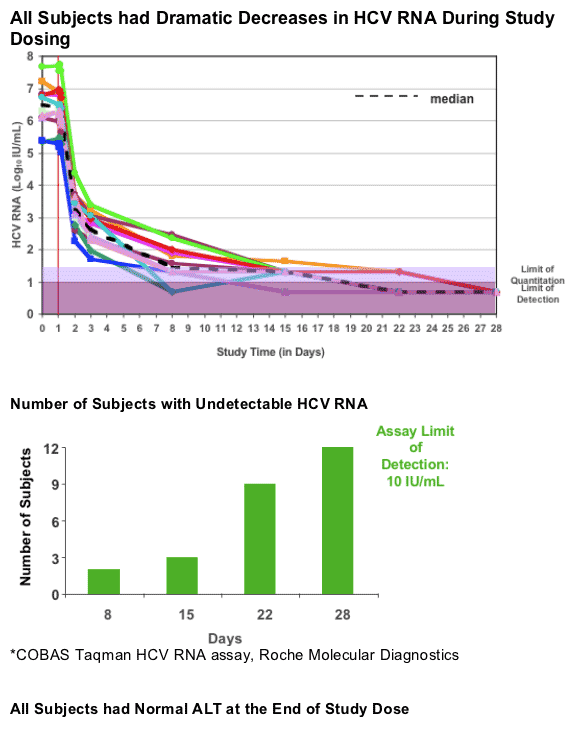
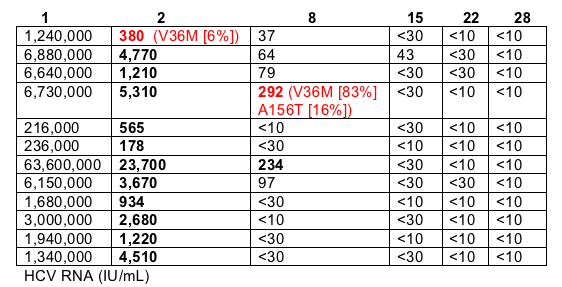
Viral Sequencing Results & Viral Load Reductions at Time Points
Sequencing was possible when HCV RNA was >100 Iu/ml so at only two time points could sequencing be performed.
There were two time points were vatiants were discovered and both are in red (380 on day 2 & 292 on day 8). The vast majority of sequencing showed Wild-Type virus, in fact all sequenced virus was reported as WT except for these two time points. You can see the mutations below and the speaker said "the important point is with continued therapy at subsequent time points these variants were suppressed".
Time Points (Days)
(numbers in bold were when sequencing could be performed; not enough virus to sequence for unbolded numbers)
Viral Sequencing Conclusions
Variants were only identified in two subjects:
- one subject had a small (6%) amount of V36M detectable at day 2, which was subsequently suppressed.
- Although V36M & A156T resistant variants were detected in one subject at day 8, the subject's HCV RNA level continued to be suppressed.
Both subjects had undetectable HCV RNA by day 22 and remain undetectable at week 12 followup.
Post Study Followup
All subjects started standard therapy with PegIFN/RBV at the end of 28 days of study dosing.
At 12 week followup, 11 subjects remain undetectable, and one subject had detectable HCV RNA:
- this subject had detectable HCV RNA (<30 IU/mL) at Week 2 followup, and undetectable HCV RNA <10 IU/mL) at weeks 4 and 8 followup.
- Viral sequencing at baseline and day 2 shows only wild-type virus.
- Viral sequencing at time of relapse is pending.
Conclusions & Future Directions
All subjects treated with VX-950 in combination with PegIFN & RBV had a rapid viral response (100% RVR).
There were no new adverse events with VX-950 & no apparent worsening of typical PegIFN & RBV adverse events.
Although viral variants were identified early in therapy, they were rapidly cleared by continued therapy.
A phase 2 program which will include evaluation of a 12-week VX-950-based treatment regimen is now being initiated.
|
| |
|
 |
 |
|
|