 |
 |
 |
| |
HCV-796, new HCV non-nucleoside- 14 day study
|
| |
| |
"Antiviral Activity of the Non-Nucleoside Polymerase Inhibitor, HCV-796, in patients with chronic HCV: preliminary results from a randomized, double-blind, placebo-controlled, ascending multiple dose study"
Reported by Jules Levin
DDW, May 21-24, 2006, Los Angeles (Digestive Disease Week)
Stephaen Villano from ViroPharma reported these study results at the first oral presentation this morning at 8:30am at the opening session at DDW.
PRECLINICAL
- chemical class: Benzofuran
- target: HCV NS5B RNA dependent RNA plymerase
- mechanism: non-compettitive allosteric inhibitor (non-nucleoside)
In vitro Antiviral Activity:
RdRp Enzyme activity:
- genotypes 1a & 1b: IC50= 0.01 - 0.16 uM
- genotypes 2, 3, 4: IC50 + 0.22 - 1.7 uM
Replicon Activity
- 1a replicon; EC50 = 4.5 +/- 2.0 nM
- 1b replicon: EC50 = 8.6 +/- 4.0 nM
In vivo Antiviral Activity:
- 2 log HCV RNA reduction in chimeric mice infected with HCV
STUDY DESIGN
Randomized, double-blind, placebo-controlled dose-ranging study.
Dosing: 6 ascending dose groups
- 50, 100, 250, 500, 1000, 1500 mg
- twice daily dosing
- 3:1 allocation (12 active, 4 placebo) per group
- 14 days of dosing
- 4 week post-treatment followup
- objectives; safety, PK, antiviral activity
Patients were treatment-naïve with chronic HCV; no other causes of liver disease; no advanced or decompensated liver disease; plasma HCV RNA >10,000 at screening; HIV-neg.; ALT <5x ULN.
Baseline HCV genotyping with TRUEGENE HCV'NC Genotyping Kit (Bayer Healthcare); Roche Amplicor HCV Monitor. PK profiles on day I & 14.
DEMOGRAPHICS
There were about 12 in each dose group. Average age about 50 years. About 25% female. Weight: 80-90 kg. In the 500-1500 mg dose groups about 35% were Black, others White. Baseline HCV RNA: median 6.4 to 7.2.
725 were genotype 1.
PK
HCV-796 tmax is about 2-3 hours. Peak Cmax, AUC0-12, AUC (day14/day1), and t1/2 occurred at the 1000 mg dose and declined with the 1500 mg dose. With the 1000 mg dose:
Cmax: 2186 ng/mL;
AUC0-12 20048 ng hr/mL
T1/2: 54 hr
AUC day 14/day1: 4.2
Trough plasma concentrations (n=12) performed best with the 1000 mg Q12 dose. They were well above target concentration based on preclinical antiviral activity.
VIRAL LOAD REDUCTIONS
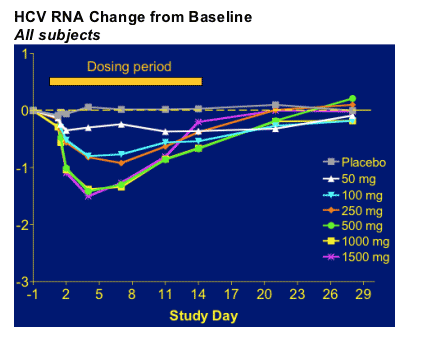
HCV-796 treatment resulted in reduced plasma HCV RNA levels. Maximal antiviral effects were achieved at study day 4, with peak mean reductions in HCV RNA across all doses ranging from 0.3 to 1.4 log10 (50% to 97%). In the 1000 mg cohort, the mean reduction in HCV RNA was 1.4 log10 (96%) on day 4, 1.3 log10 (95%) on day 7, and 0.7 log10 (80%) on day 14. In this group, 83% of patients had reductions from baseline > 1.0 log10 on day 4; 42% of these patients had reductions > 1.5 log10 and 33% had reductions > 2.0 log10. On day 14, 17% of patients in the 1000 mg cohort had reductions from baseline > 2.0 log10. Antiviral activity appeared similar among patients infected with HCV genotype 1 compared with those infected with other genotypes of HCV. Trough Pk appeared to be related to viral load reduction in the 1000mg dose group.
The speaker addressed the fact that peak viral load reduction of -1.4 log occurred on day 4 but viral load decline started to increase. The issue of drug resistance was discussed by the speaker as a possible reason and said that the resistance testing results analysis is ongoing and will be presented later this year at another conference.
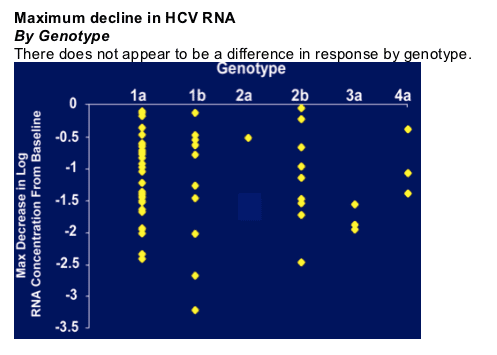
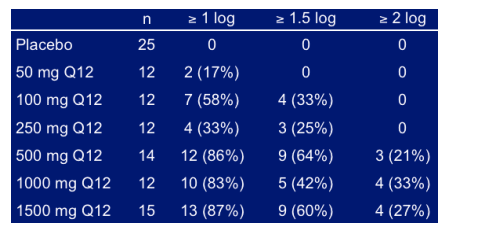
HCV-RNA Reductions
Maximum Change Over 14 Days
% Subjects With Maximum decline From Baseline in log10 HCV RNA
SAFETY
The drug was reported as generally well-tolerated. No dose-limiting toxicities identified across the range of study doses. No serious adverse events. Discontinuations due to AEs: placebo- 910 poorly controlled hypertension (preexisting); 500 mg: (10 elevated TSH level. 1500 mg: (1) elevated unconjugated bilirubin (3.7 mg/dL).
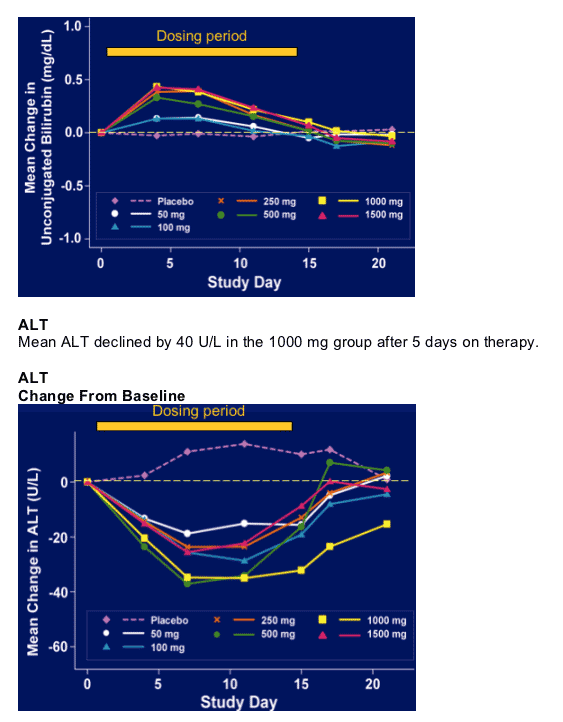
Most common AEs: headache 42%, constipation 17%; diarrhea: 25% in 1000 mg group; pruritis 17%; rash 25% in 100mg group. Across dose groups there did not appear to be a trend in AEs except in 1000 mg group 25% diarrhea, but 20% in placebo; and rash 25% in 1000 mg group & 0% in placebo, and 4, 8, & 7% in 3 other dose groups, so there appeared to perhaps be a rash issue.
Bilirubin (unconjugated)
Mean change in unconjugated bilirubin was +0.5 mg/dl during the dosing period, occurred after a few days 7 then appeared to decline.
Bilirubin (Unconjugated)
Change From Baseline
CONCLUSIONS by Authors
HCv-796 displays dose-related antiviral activity, across multiple HCV genotypes; peak response achieved at the 500-1000 mg Q12 dose. Exposure (PK) is less than dose-proportional with increasing dose, with plateau at the 1000 mg Q12 dose. Genetic sequencing of HCV NS5B is ongoing, with focus on subjects with 'breakthrough' virologic response pattern. HCV-796 was well-tolerated: no dose-limiting toxicities across the range of study doses; mild-moderate headache was the most common AE; ALT decrease temporally associated with antiviral activity. Study of combination therapy with peginterferon ongoing.
|
| |
|
 |
 |
|
|