 |
 |
 |
| |
NM283 + Peg: differences in response between treatment naives & nonresponders; dose reductions for safety concerns
|
| |
| |
"Valopicitabine (NM283), Alone or with Peg-Interferon, Compared to Peg_Interferon/Ribavirin (pegIFN/RBV) Retreatment in Hepatitis C Patients with Prior Non-Response to PegIFN/RBV:
Week 24 Results" Safety & Efficacy
Reported by Jules Levin
DDW 2006
May 25-28, Los Angeles, CA
Reported at DDW by Paul Pockros for investigators.
Authors- P. Pockros, C. O'Brien, E. Godofsky, M. Rodriguez-Torres, N. Afdhal, S. Pappas, E. Lawitz, N. Bzowej, V. Rustgi, M. Sulkowski, K. Sherman, I. Jacobson, G. Chao, S. Knox, K. Pietropaolo, N. Brown and the Valopicitabine 04 Study Group
This presentation highlights the difference in response to NM283 + Peg between naives & nonresponders in studies (see slides below). And reports safety concerns prompting Protocol amendments reducing dosing in studies from 800 mg daily to 200 and 400 mg daily.
Protocol Amendments
(as has been reported study of NM283 continues with dosing limited to 200 and 400 mg daily)
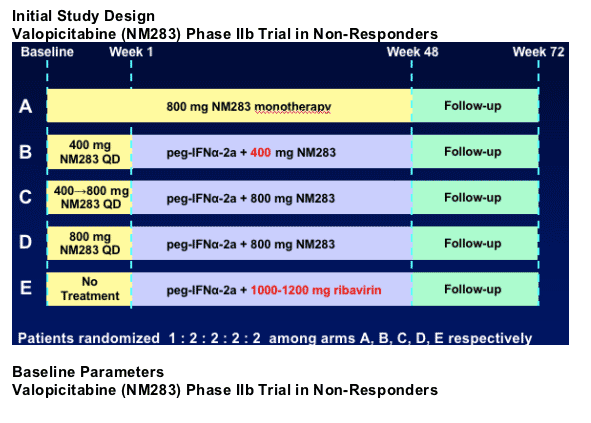
Valopicitabine (NM283) Phase IIb Trial in Non-Responders
First amendment:
- Allow Rx to max 72 weeks or until PCR-nondetectable x 36 weeks
- Goal to allow consolidation of treatment responses
Second amendment:
Severe GI side effects in some patients at NM283 800 mg/day
- More in treatment-naive study than non-responder study 1 (1 Dieterich et al, EASL 2006)
- Key concern was 3 patients (2 naive, 1 non-responder) with persistent nausea/vomiting requiring in-patient rehydration
Dose range reduced in both studies to a maximum 400 mg/day.
All 13 patients with HCV RNA >1000 IU/mL were discontinued.
Valopicitabine Development
Next Steps in Non-Responder Program
- Ribavirin / NM283 interaction study - starts 2Q 2006
- Triple regimens (NM283 + pegIFN + RBV) will be investigated clinically if no interaction with RBV
- Studies of high dosing in animal model underway, to evaluate mechanism and mitigation of GI toxicity
Valopicitabine (NM283)
First Nucleoside-type HCV Polymerase Inhibitor
- NS5b polymerase inhibitor
- Ribonucleoside; cytidine analogue
- Inhibits viral polymerase, causes viral RNA chain termination
- Oral agent
- Valyl ester pro-drug provides high oral bioavailability
- Plasma half life (4-6 hrs) & intracellular half life (15 hrs) support once-daily dosing
- 200 - 800 mg demonstrate anti-viral activity
Objectives
Valopicitabine (NM283) Phase IIb Trial in Non-Responders
Compare antiviral efficacy and safety/tolerability of:
- 3 different dosing regimens of valopicitabine + peg-IFN_-2a
- Versus re-treatment with peg-IFN_-2a + ribavirin
- Also included: valopicitabine monoRx arm
Patient population:
- HCV genotype 1 chronic hepatitis C
- Non-responders to pegIFN_ + ribavirin
- Relapsers to pegIFN + ribavirin excluded
Study Design
Valopicitabine (NM283) Phase IIb Trial in Non-Responders
Multicenter (22), randomized, active control design
HCV RNA response criteria for failure and discontinuation - required reductions from baseline:
- Week 4 >/= 0.5 log
- Week 12 >/= 1.0 log
- Week 24 > 2.0 log
Primary efficacy endpoint: Sustained Virologic Response
- HCV RNA assayed by COBAS TaqMan PCR (20 IU/mL)
Key Eligibility Criteria
Valopicitabine (NM283) Phase IIb Trial in Non-Responders
18-65 years of age, male or female
HCV genotype 1
Non-responders to previous adequate treatment course:
- At least 12 weeks of pegIFN + RBV
- At least 75% of the prescribed doses of pegIFN and RBV
- Failed to clear HCV RNA to non-detectable levels
- Patients must have previously failed for efficacy, not safety
Screen/Baseline
- HCV RNA >/= 105 IU/mL
- ALT < 5 x ULN
Compensated liver disease
Current Status and Patient Disposition
Valopicitabine (NM283) Phase IIb Trial in Non-Responders
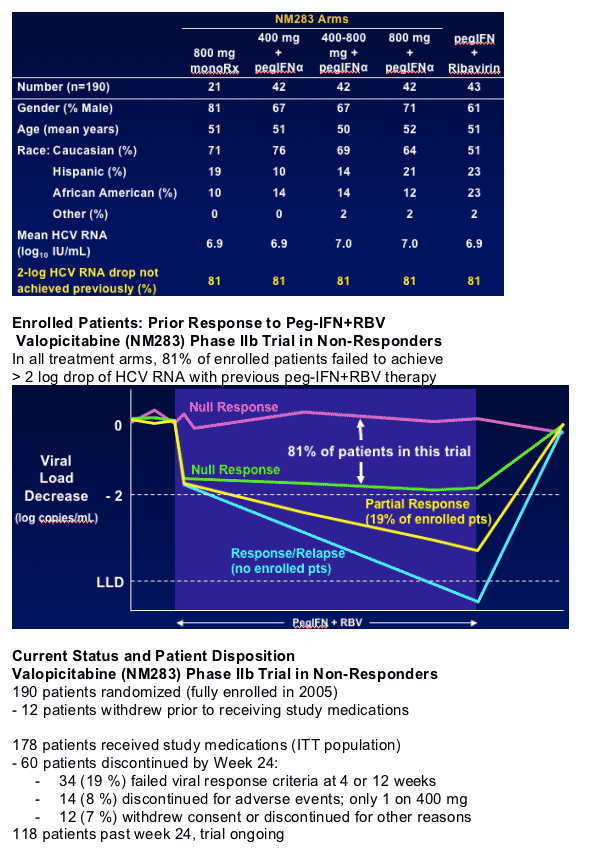
190 patients randomized (fully enrolled in 2005)
- 12 patients withdrew prior to receiving study medications
178 patients received study medications (ITT population)
- 60 patients discontinued by Week 24:
- 34 (19 %) failed viral response criteria at 4 or 12 weeks
- 14 (8 %) discontinued for adverse events; only 1 on 400 mg
- 12 (7 %) withdrew consent or discontinued for other reasons
118 patients past week 24, trial ongoing
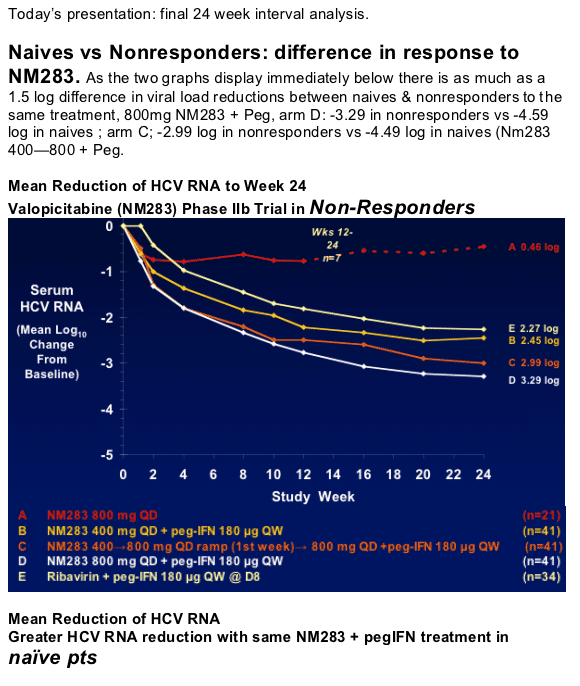
Today's presentation: final 24 week interval analysis.
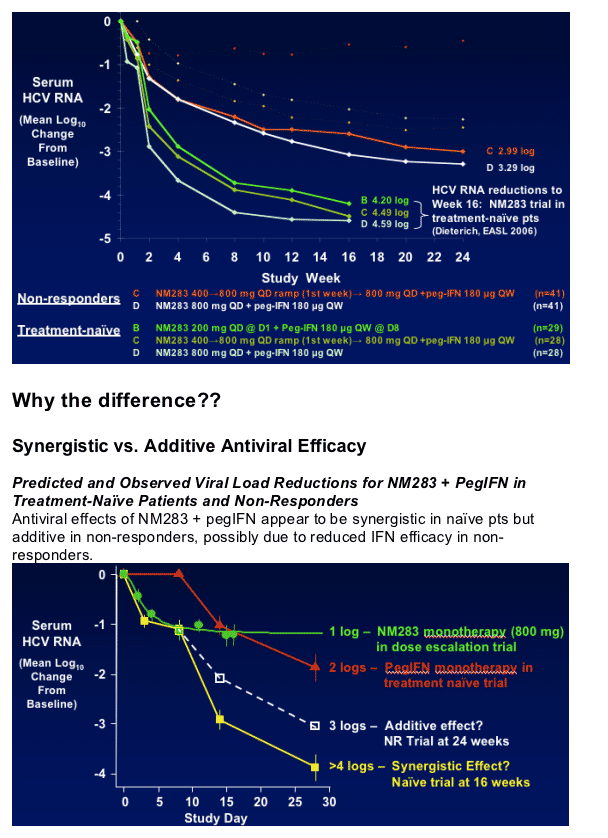
Naives vs Nonresponders: difference in response to NM283. As the two graphs display immediately below there is as much as a 1.5 log difference in viral load reductions between naives & nonresponders to the same treatment, 800mg NM283 + Peg, arm D: -3.29 in nonresponders vs -4.59 log in naives ; arm C; -2.99 log in nonresponders vs -4.49 log in naives (Nm283 400-800 + Peg.
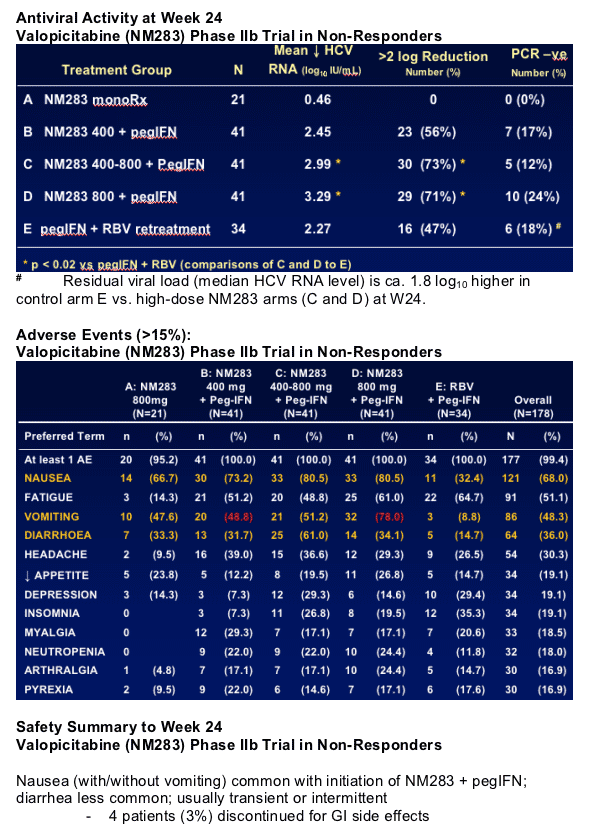
Safety Summary to Week 24
Valopicitabine (NM283) Phase IIb Trial in Non-Responders
Nausea (with/without vomiting) common with initiation of NM283 + pegIFN; diarrhea less common; usually transient or intermittent
- 4 patients (3%) discontinued for GI side effects
24 serious adverse events (SAEs) through Week 24, 6 reported possibly attributed to NM283
- Anemia, colitis, dehydration, fatigue, pancreatitis, gram-negative bacteremia 2 to UTI
Sporadic elevations of amylase, lipase, AST, ALT
- Rarely treatment-limiting
No hematologic toxicities (anemia, neutropenia) on NM283 monotherapy.
Author's Conclusions
Valopicitabine (NM283) Phase IIb Trial in Non-Responders
Statistically superior antiviral activity at Week 24 for NM283 800mg + pegIFN dose groups
- Log reduction in serum HCV RNA
- EVR rate
HCV RNA levels still declining beyond Week 24.
Antiviral synergy for NM283 + pegIFN in treatment-naive patients is less apparent in non-responders
- Appears related to differential effect of pegIFN
GI side effects common but tend to be transient and/or manageable. NM283 + pegIFN is satisfactorily tolerated by most non-responders.
o Only 3% discontinued for GI side effects
Valopicitabine Development
Next Steps in Non-Responder Program
Ribavirin / NM283 interaction study - starts 2Q 2006.
Triple regimens (NM283 + pegIFN + RBV) will be investigated clinically if no interaction with RBV.
Studies of high dosing in animal model underway, to evaluate mechanism and mitigation of GI toxicity.
|
| |
|
 |
 |
|
|