 |
 |
 |
| |
Differences In Treatment Outcome To Antiviral Therapy Based On Genotype And Viral Load In Hepatitis C Genotypes 2 And 3 In The WIN-R Trial
|
| |
| |
Reported by Jules Levin
DDW, May 2006, Los Angeles
R.S. Brown, Jr., I.M. Jacobson, N. Afdhal, B. Freilich, M.P. Pauley, F. Regenstein, S. Flamm, P. Kwo,
L. Griffel, C.A. Brass, & the WIN-R Study Group
WIN-R Study: final results of Weight-Based dosing study of ...Here is link to report on WIN-R Study presentation at AASLD Nov 2005 written by Jules Levin
www.natap.org/2005/AASLD/aasld_59.htm
WIN-R Trial: Background
Weight-Based Interferon and Ribavirin Trial
- Peginterferon (PEG-IFN) alfa-2b 1.5 mg/kg/week + ribavirin (RBV) 800 mg/day was superior to two other regimens using RBV 1000-1200 mg/day in a pivotal trial
- Manns et al, Lancet 2001
- Regression analysis showed a linear relationship between RBV dose in mg/kg and sustained virological response (SVR) supporting the use of weight-based dosing of RBV
- A large prospective trial - WIN-R - comparing RBV doses was designed to address fixed dose RBV 800 mg vs weight-based RBV dosing
WIN-R: G 2/3 Analysis
- Patients with G 2/3 chronic hepatitis C have high SVR rates with PEG-IFN alfa and RBV
- Require shorter duration of treatment
- May not need high dose of RBV
- Optimal dosing of RBV and differences between SVR in G 2/3 patients is undefined
Objective of analysis
To analyze the efficacy of PEG-IFN alfa-2b plus RBV in patients with HCV G 2/3 in the WIN-R trial
WIN-R Trial
- US multicenter, randomized, open-label, investigator-initiated trial between 12/2000 and 6/2005
- Randomized treatment-naive adults stratified by genotype (1 and non-1) and
fibrosis
- 25 regional investigators, 225 sites
- Nearly 5000 patients; 1829 patients with G 2/3
- Central lab (SPRI) for quantitative PCR for HCV RNA
- Lower limit of detection was 29 IU/ml
- 0, 24, 48, 72 weeks
Study Populations and Analyses
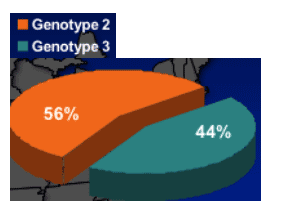
- Intent-to-treat (ITT) population
- All subjects who received at least one dose of study drug
- Used for safety analysis
- Primary efficacy population
- All subjects who received at least one dose of study drug and weighed ≥65 kg
- Used for primary efficacy end point - SVR
- Completers analysis population
- All subjects who received at least one dose of study drug, completed the study, and had end-of-treatment data available
Study Enrollment and Randomization:
HCV G 2/3 Subjects
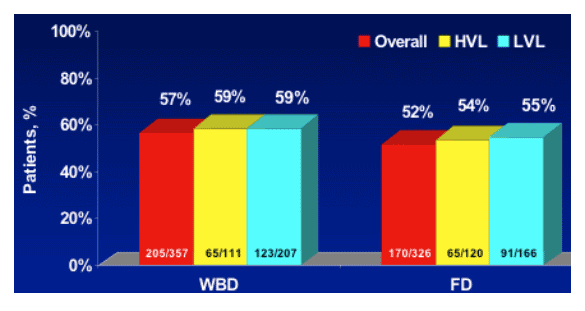
WIN-R: Genotype 2/3 Conclusions
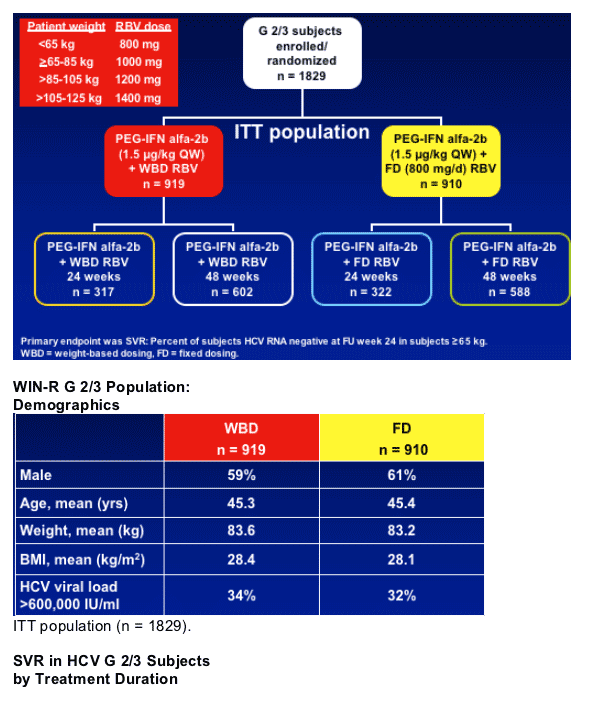
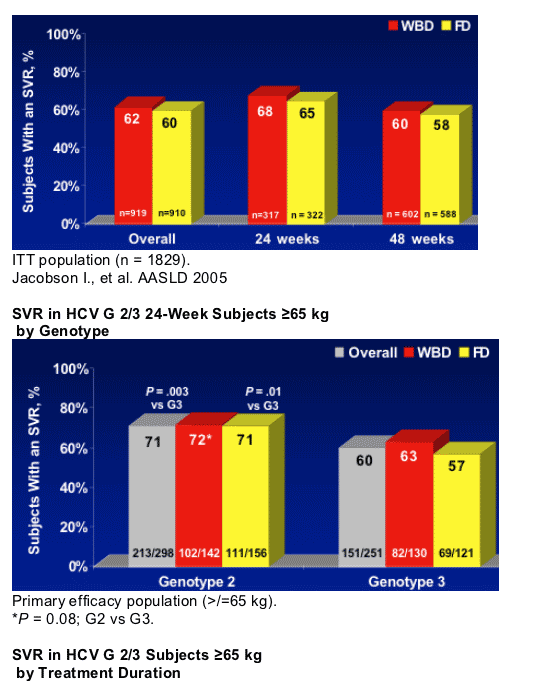
- 24 weeks of PEG-IFN alfa-2b + RBV was sufficient for HCV G 2/3 patients
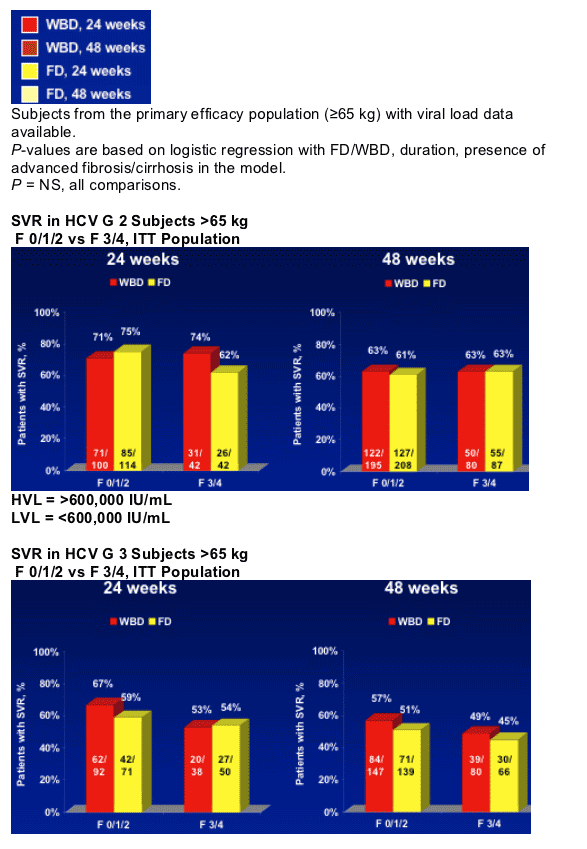
- PEG-IFN alfa-2b + fixed-dose (800 mg) RBV for 24 weeks was sufficient for Genotype 2 patients
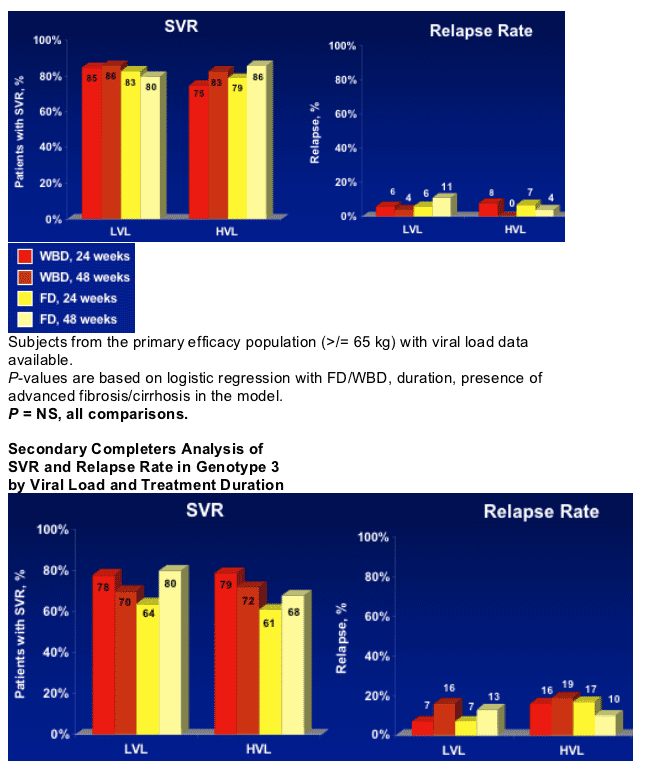
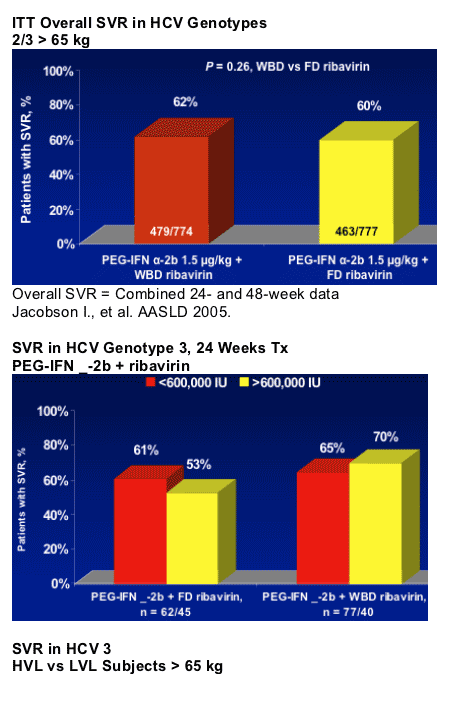
- Overall, G 2 patients had higher SVR rates from treatment with PEG-IFN alfa-2b + RBV than G 3 patients
- Weight-adjusted doses of RBV may benefit patients with G 3 and high viral load
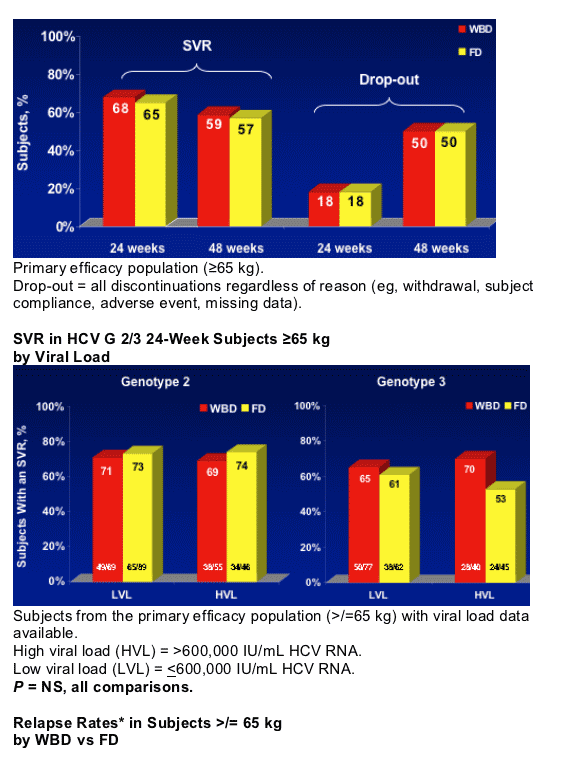
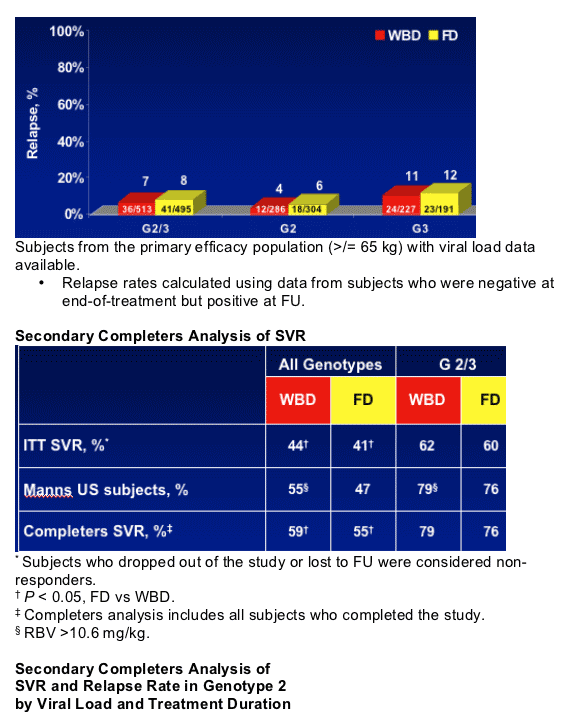
- Treatment with PEG-IFN alfa-2b + RBV resulted in consistently low relapse rates in G 2/3 patients treated for either 24 weeks or 48 weeks
- No clear differences were seen in SVR and relapse rates for G3 HVL with longer duration of treatment
- Further research is needed to define optimal treatment for G3 HVL
|
| |
|
 |
 |
|
|