 |
 |
 |
| |
The Impact of Growth Factors on Pegylated Interferon alfa-2b and
Ribavirin Dose Reduction in Patients Treated for Genotype 1 Chronic Hepatitis C
|
| |
| |
Reported by Jules Levin
DDW, May 2006, Los Angeles
M. Kugelmas,1 A. L. Sabel-Soteres,2 G. Spiegelman,3 M. Mah'Moud,4 and the KATT Study Group
1South Denver Gastroenterology, Englewood, Colorado; 2Denver Health, Denver, Colorado; 3Spiegelman Gastroenterology, Knoxville, Tennessee; 4Boice-Willis Clinic, Rocky Mount, North Carolina
RESULTS
Early Virologic Response Rates
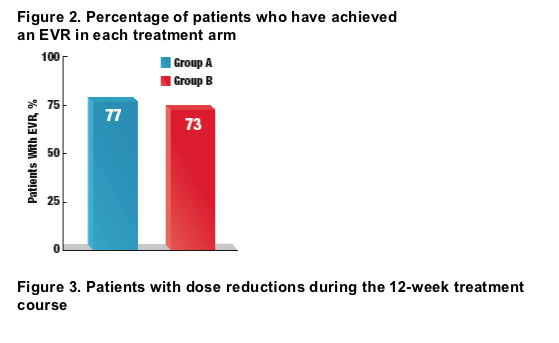
- In total, 23 (77%) of 30 patients in group A and 24 (73%) of 33 patients in group B achieved an EVR (Figure 2)
Dose Reduction and Discontinuations
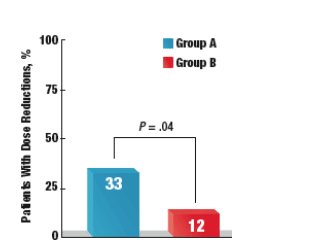
- Overall, doses of anti-HCV treatments were significantly more likely to be reduced in group A than in group B (33% vs 12%, respectively; P = .04) (Figure 3)
Group A
- Ten (33%) of 30 patients in group A had dose reductions
- Ribavirin dose was reduced in 5 patients (n = 2 [anemia] and n = 1 each [flulike symptoms, shortness of breath, and nondefined side effect])
- Peginterferon alfa-2b dose was reduced in 9 patients (n = 3 [low ANC]; n = 2 [thrombocytopenia]; n = 2 [flulike symptoms]; and n = 2 [weight loss])
Group B
- Dose reductions occurred in 4 (12%) of 33 of patients in group B
- Two patients required temporary dose reduction (ribavirin for hemoglobin <10 g/dL and peginterferon alfa-2b for ANC <750/mm3)
- Two patients had permanent dose reductions (ribavirin for nausea and peginterferon alfa-2b for weight loss)
- Twenty-one (64%) of 33 patients in group B received growth factors (darbepoetin [n = 16]; filgrastim [n = 2]; darbepoetin and filgrastim [n = 3])
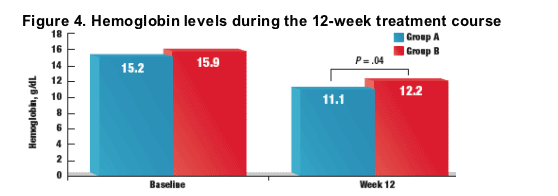
- In both treatment groups, hemoglobin levels decreased from baseline through week 12 (Figure 4)
- At week 12, hemoglobin levels were significantly lower in group A patients than in group B patients (11.1 g/dL vs 12.2 g/dL, respectively; P = .04)
Author Conclusions
- Use of growth factors prevents dose reductions of peginterferon alfa-2b and ribavirin in HCV genotype 1 patients receiving anti-HCV therapy
- Use of growth factors maintains more physiologic hemoglobin levels
- Ongoing results of this study will show whether use of growth factors allows for higher SVR rates
Abstract
Background: Hematologic toxicity is one of the most common reasons for dose reduction or discontinuation when treating chronic hepatitis C. In turn, these dose reductions negatively affect the chance of achieving early virologic response (EVR) during therapy of patients with genotype 1 chronic hepatitis C (HCV-1).
Aims and Methods: We are comparing standard of therapy (ST) with pegylated interferon alfa 2b (P-IFN) plus weight-based ribavirin (RBV) in patients with HCV-1 to an adjuvant therapy (AT) regimen where anemia is pre-emptively treated with darbepoetin alfa (3 _g/kg SQ Q2W starting with hemoglobin <12 g/dL or <75% of baseline), neutropenia is pre-emptively treated with filgrastim (300 _g SQ QW for ANC <900 mm3), and more lenient cut-off levels for thrombocytopenia are allowed in order to try to prevent dose reductions due to hematologic toxicity. The study is powered to show a 22% difference in sustained viral response (SVR) in favor of the adjuvant therapy arm.
Results: To date, 63 patients with HCV-1 have received at least 12 weeks of therapy. Forty-two are men, 60% have high viral load (HVL >800,000 IU), age is 46±8 years old, fibrosis score is 1.7±1.0, average weight is 80±15 kg, and 10% are African American.
Of 30 patients receiving ST, 77% have achieved EVR, and of 33 receiving
AT regimen, 73% have EVR. Twenty-one patients in the AT arm (64%) received growth factors (16 darbepoetin, 2 filgrastim and 3 received both), two required temporary dose reduction (RBV for hemoglobin <10 g/dL and P-IFN for low ANC) and 2 had permanent dose reductions (RBV for nausea, and P-IFN for weight loss). Ten patients in the ST arm had dose reductions. RBV was dose reduced for anemia twice and in one occasion each for flulike symptoms, shortness of breath and non-defined side effect. P-IFN dose reduction was done in 9 patients
for low ANC (n=3), thrombocytopenia (n=2), flu-like symptoms (n=2) and weight loss (n=2). Antivirals were more likely to be dose reduced in the standard of therapy arm (33% in the ST arm vs. 12% in the AT arm, P = 0.04).
Baseline hemoglobin fell from 15.2±1.1 g/dL to 11.3±1.2 g/dL in the ST arm and from 15.9±1.4 g/dL to 12.2±1.1 g/dL in the AT arm; the difference was significant and sustained since week 2 and P = 0.004 for the difference of the nadirs.
Conclusions: Use of growth factors prevents dose reductions of P-IFN and RBV in patients receiving anti- HCV-1 therapy and maintains more physiologic hemoglobin levels. Ongoing results of the study will show whether use of growth factors will allow for higher SVR. Study drugs and support were provided by Amgen Inc. (darbepoetin and filgrastim) and Schering Plough (PegIntron and Rebetol).
Background
- When treating patients with chronic hepatitis C with pegylated interferon and ribavirin, hematologic toxicity is one of the most common reasons for dose reduction or discontinuation. These dose reductions negatively affect the chance of achieving an early virologic response (EVR) during therapy in patients with genotype 1 chronic hepatitis C virus (HCV) infection
- EVR, which is defined as a >/= 2 log drop in HCV RNA from baseline at week 12 of treatment, is a recognized positive predictor of sustained virologic response (SVR)
Aim
- To determine whether the use of growth factors and more lenient platelet cutoff points during anti-HCV therapy results in better adherence to target doses of peginterferon alfa and ribavirin, higher EVR rates, and higher SVR rates
Methods
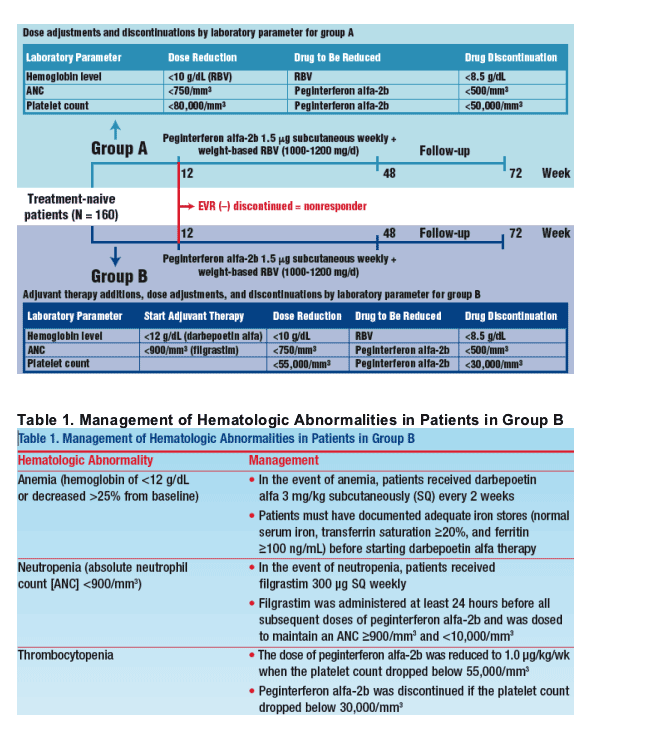
- Treatment-naive patients (N = 160) with compensated chronic liver disease secondary to chronic hepatitis C due to genotype 1 HCV infection were randomly assigned to 2 treatment groups (Figure 1)
- Patients who did not achieve a >/= 2 log drop in HCV RNA from baseline by week 12 of treatment were excluded from the study and considered nonresponders
Figure 1. Study design
In patients randomly assigned to group B, management of hematologic abnormalities was carried out with the additional use of growth factors (Table 1).
Study End Points
- Primary end point was assessment of end-of-treatment response at 48 weeks and SVR after 24 weeks of follow-up
- Secondary end points
- Biochemical response (alanine transferase [ALT] levels)
- Safety
- Interim data are presented for patients who completed at least 12 weeks of therapy
Statistical Analyses
- A sample size of 160 achieves 80% power to detect an effect size (W) of 0.232 using a 1-degree of freedom chi-square test with a significance level (_) of 0.05000. The W corresponds to SVRs of 54% (group A) and 76% (group B)
- This calculation allows for a dropout rate of 15% but assumes dropouts occur randomly among the 2 groups and 2 responses
RESULTS
- To date, 122 patients are enrolled in the study
- Sixty-three patients (n = 30 [group A]; n = 33 [group B]) have completed at least 12 weeks of therapy
- Patient demographics for the 63 interim-analysis patients are shown in Table 2
- There were no significant differences in baseline characteristics between the 2 groups

|
| |
|
 |
 |
|
|