 |
 |
 |
| |
Combination HBV Therapy (lamivudine+adefovir) Prevented HBV-DNA Viral Rebound and Adefovir Resistance Compared to Adefovir Monotherapy in Cirrhotics
|
| |
| |
Reported by Jules Levin
EASL, April 26-30, Vienna, Austria
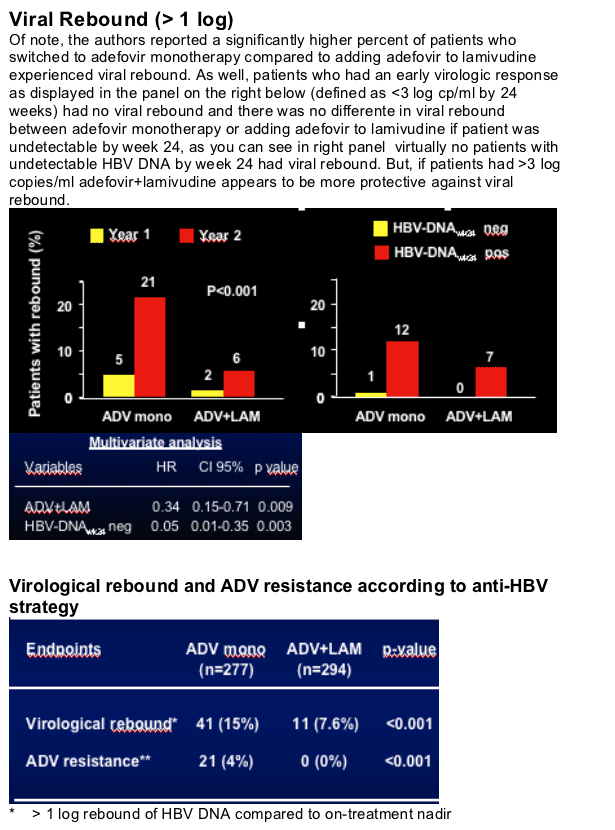
Although the study was conducted in cirrhotics the implication is that combination therapy can prevent viral rebound and adefovir resistance in lamivudine failures who switch to adefovir. The author Lampertico reported similar findings in a second study he reported at EASL in which 50% of the study patients had cirrhosis. Lampertico made a few important points, (1) HBV genotypic resistance testing should be utilized, (2) switching therapy at the time of genotypic resistance will result in better patient outcomes, earlier is better than waiting for clinical failure 9ALT elevation). If switching to adefovir following lamivudine resistance or clinical/virologic failure, combination therapy with lamivudine+adefovir is preferable to adefovir monotherapy.
"FIVE YEARS OF SEQUENTIAL LAM TO LAM+ADV THERAPY SUPPRESSES HBV REPLICATION IN MOST HBeAg-NEGATIVE CIRRHOTICS, PREVENTING DECOMPENSATION BUT NOT HEPATOCELLULAR CARCINOMA"
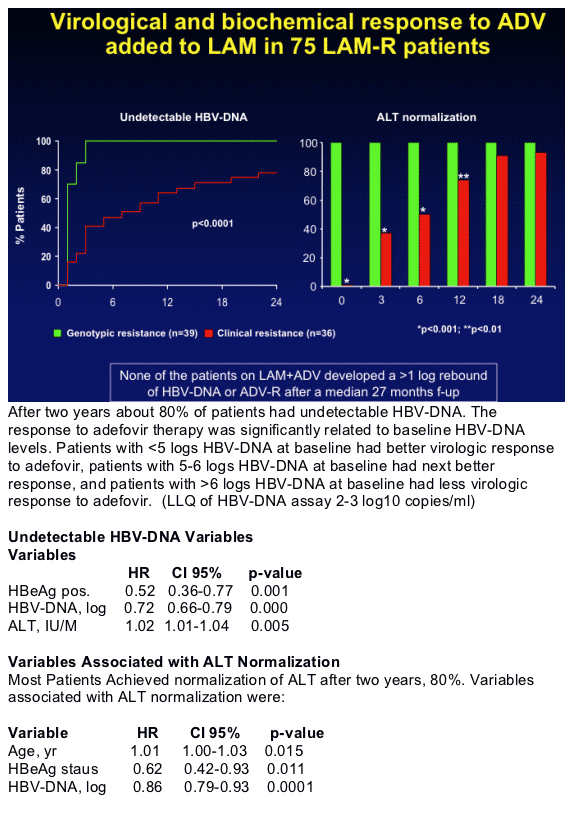
".....None of the patients on LAM+ADV developed a >1 log rebound of HBV-DNA or ADV-Resistance after a median 27 months follow-up..."
authors: P Lampertico, M Viganò, E Manenti, M Iavarone, G Lunghi, M Colombo
Department of Gastroenterology and Division of Hygiene, IRCCS Maggiore Hospital, Fondazione Policlinico, Mangiagalli and Regina Elena, University of Milan, Milan, Italy
Background and Aim
-- Long-term viral suppression is the most important goal of treatment in patients with chronic HBV infection
-- In cirrhotic patients, response to lamivudine reduces liver decompensation and improves survival but the emergence of resistant variants (70% at 5 years) significantly reduces the benefit of therapy
-- ADV is an effective treatment for LAM-R patients
-- Aim of the study was to evaluate the long-term efficacy of a sequential LAM to LAM+ADV therapy in patients with HBeAg-negative cirrhosis
AUTHOR SUMMARY
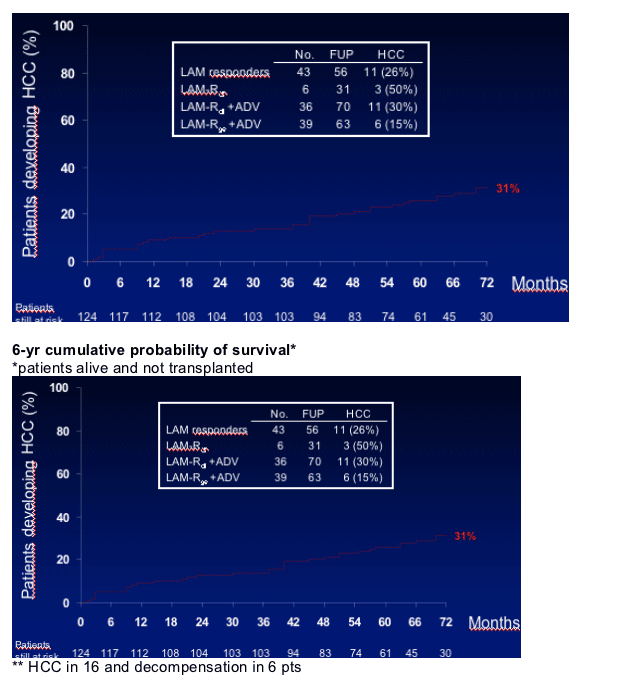
43 (35%) patients treated with LAM monotherapy for 5 years maintained undetectable HBV DNA, four (3%) lost HBsAg and 3 developing anti-HBs successfully withdrew from treatment.
-- In LAM-R patients, ADV added to LAM at the time of genotypic resistance rapidly suppressed HBV-DNA and prevented ALT increase.
-- None of the patients treated with LAM+ADV developed a > 1 log virological rebound or genotypic resistance to ADV.
-- None of the patients on long-term HBV suppression, i.e responding to LAM monotherapy or to LAM+ADV for LAM-Rge, developed clinical decompensation. However, these patients remained at significant risk for HCC development.
By year 2, 75% of the patients had undetectable HBV-DNA by sensitive assays. Independent predictors were baseline HBeAg status, HBV-DNA, and ALT levels.
80% of the patients achieved normal ALT. Age, HBeAg status, HBV-DNA were independently associated with ALT normalization.
Virological rebound occurred in 52 patients (9%), adefovir-resistance was confirmed in about 50% of the patients, all in the ADV mono group, ADV+LAM therapy and undetectable HBV-DNA at week 24 were independt predictors of a low risk of virological rebound.
3% of the patients reduced the dose of ADV because of increased serum creatinine and 2% had side effects, related or possible related to ADV.
Patients and Methods
Study: open-label, prospective cohort study (1997-2002
Patients: 124 consecutive HBeAg(-)/HBV-DNA(+) cirrhotics
Treatment: lamivudine 100 mg/day +/- adefovir dipivoxil 10 mg/day
Follow-up: 61 months (5-108)
Assays: HBV-DNA (LLQ: 3.3 log10cps/mL, Versant 3.0) Genotype (INNO-LiPA, direct sequencing). Drug-resistance (INNO-LiPA, direct sequencing)
Monitoring: every two months; abdominal US and AFP every 6 months
Endpoints of the Study
Virological:
- undetectable HBV DNA (<3.3 log10cps/mL)
- LAM or ADV-resistance: >1 log HBV DNA rebound vs nadir and confirmed by molecular analysis
a) genotypic (HBV-DNA 2-6 log10 and ALT < ULN)
b) clinical (HBV-DNA >6 log10 and ALT > ULN)
Biochemical: ALT < ULN
Clinical: Decompensation and HCC
Death or liver transplantation


|
| |
|
 |
 |
|
|