 |
 |
 |
| |
Safety, Tolerability and Antiviral Activity of Pradefovir Mesylate in Patients with Chronic Hepatitis B Virus Infection: 48-Week Analysis of a Phase 2 Study
|
| |
| |
Reported by Jules Levin
EASL, April 2006, Vienna, Austria
Lee KS et al. Yonsei University College of Medicine,
Seoul, Korea
Pradefovir (PDV) is a liver-targeted prodrug of PMEA with potentially improved
efficacy and less nephrotoxicity than adefovir dipivoxil (ADV).
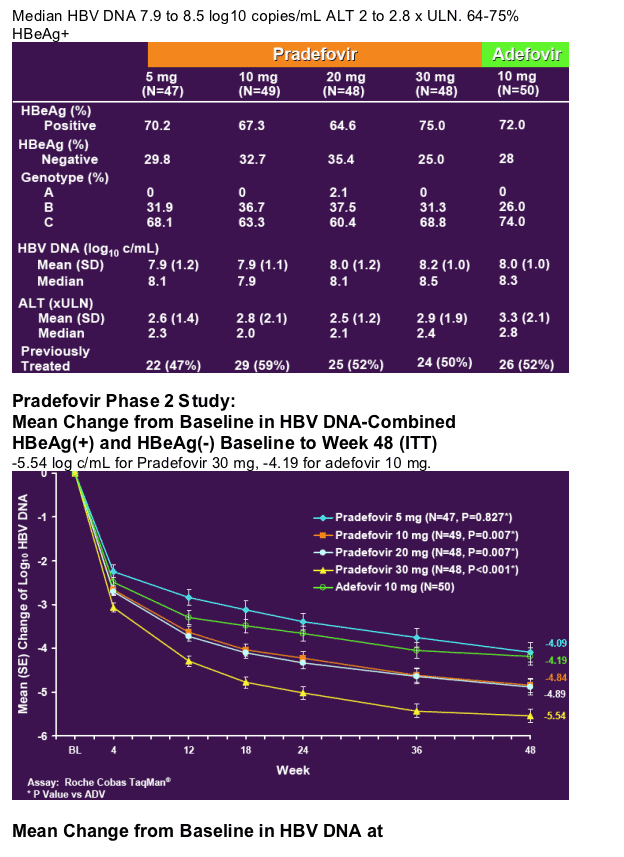
Pradefovir Phase 2 Study:
Study Objectives
Compare the safety profile of pradefovir 5 mg, 10 mg, 20 mg and 30 mg QD to
adefovir 10 mg QD.
Select the dose of pradefovir for the Phase 3 studies.
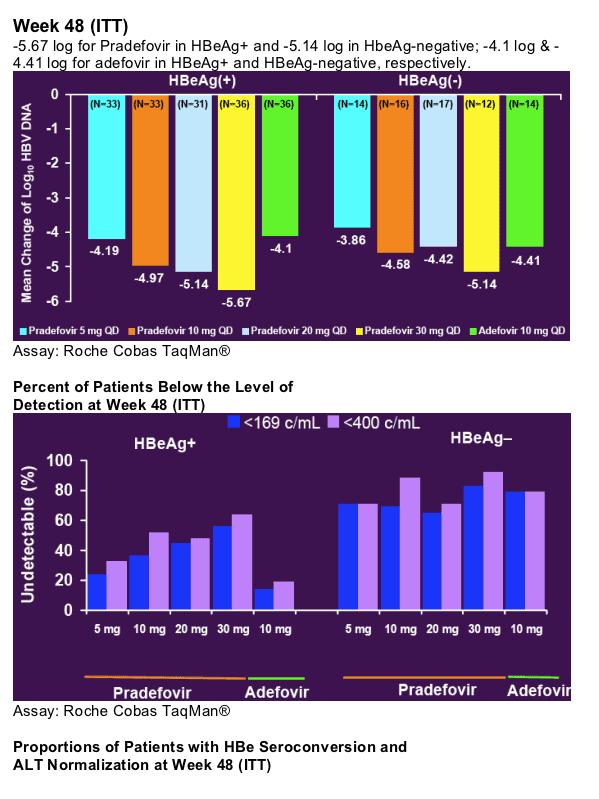
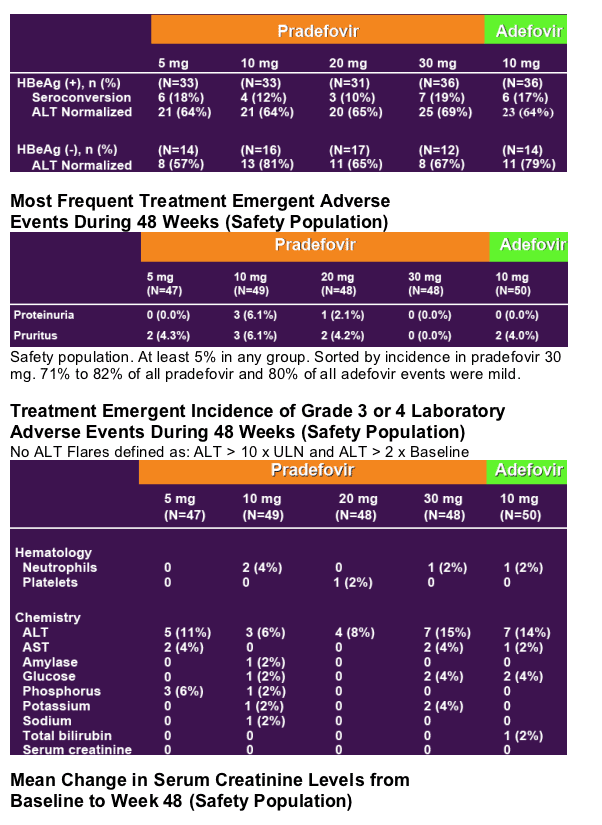
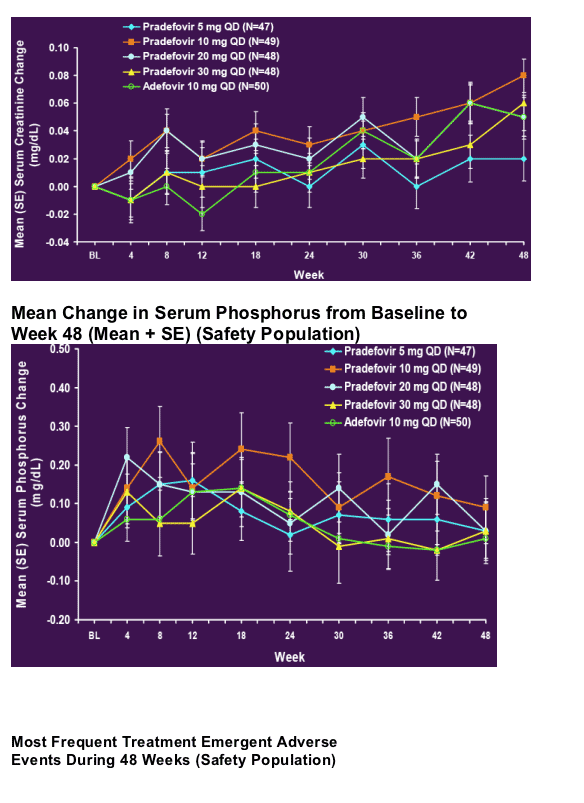
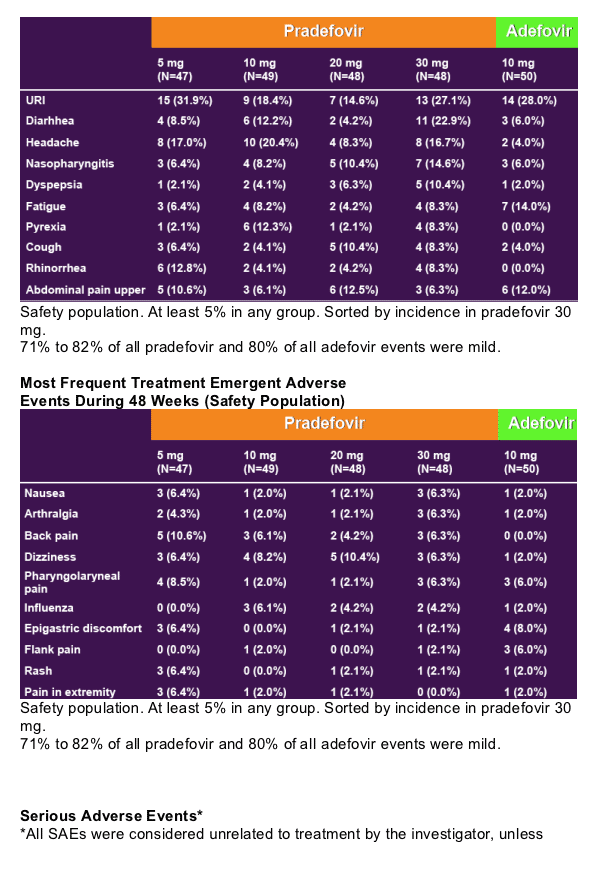
48 Week Author's Conclusions
- Pradefovir is a highly active, liver targeting prodrug of PMEA
- Significantly more active than adefovir at the registered dose
- Pradefovir, at daily oral doses of 5, 10, 20 and 30 mg per day, is well tolerated (see tables below)
- The safety of pradefovir 30 mg was comparable to adefovir at the registered dose
- The 30 mg dose selected for phase 3 studies
Pradefovir Phase 2 Study:
Design


|
| |
|
 |
 |
|
|