 |
 |
 |
| |
HBV Resistance Determination From
the Telbivudine GLOBE Registration Trial
|
| |
| |
Reported by Jules Levin
EASL April 2006, Vienna, Austria
Poster 514
DN Standring,1 M Seifer,1 A Patty,1 C Chapron,1 LJ Van Doorn,2 G Chao,1 N Brown,1 CL Lai3
1Idenix Pharmaceuticals Inc., Cambridge, Mass, USA; 2Delft Diagnostic Laboratories, Voorburg, The Netherlands; 3University of Hong Kong
The safety and efficacy of telbivudine (LdT) are being evaluated vs lamivudine (LAM) in a large, international randomized trial (the GLOBE trial) involving 1367 intent-to-treat (ITT) patients with chronic hepatitis B. Here we report a detailed analysis of hepatitis B virus (HBV) genotypes and treatment-emergent viral breakthrough and resistance after 48 weeks of therapy.
Methods
- HBV genomes were amplified from sera obtained at Screen for 1367 study patients by RT-PCR and sequenced
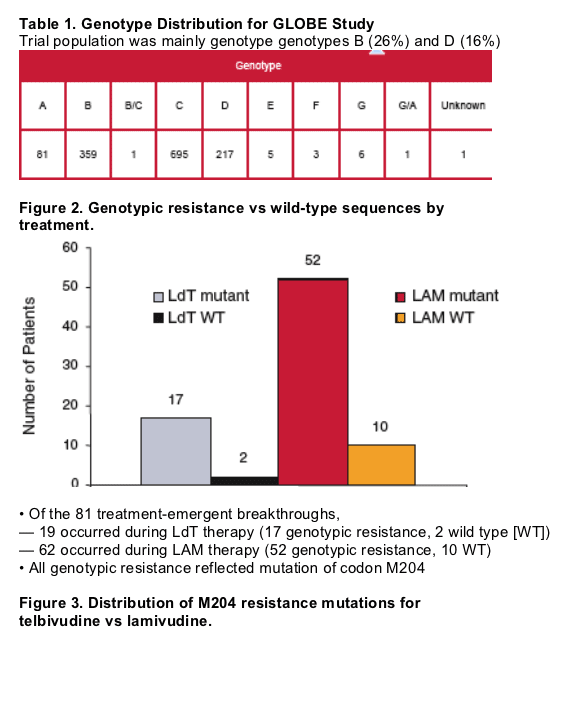
- The 344 amino acid polymerase RT domain was sequenced to determine HBV genotype by comparison with 5 reference standards
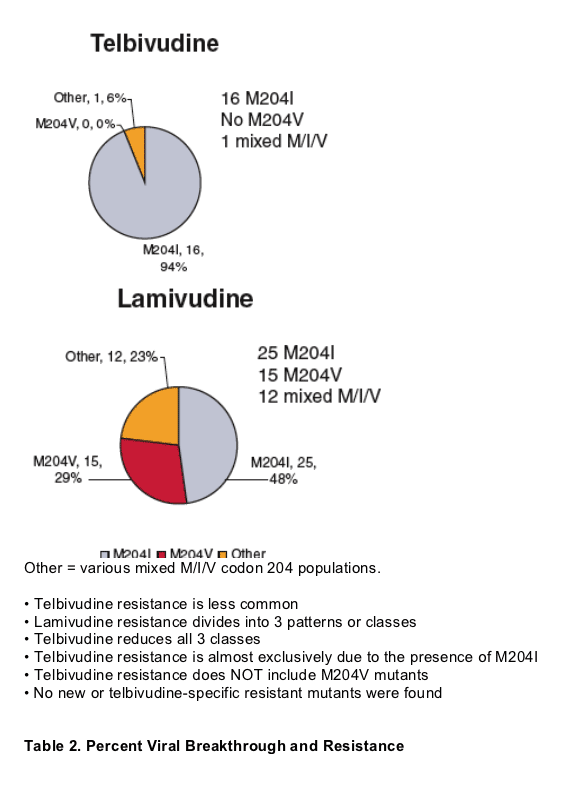
- The 81 patients (19 telbivudine, 62 lamivudine) experiencing protocol-defined viral breakthrough* (BK) were resequenced at Week 48 or post-BK to identify resistance mutations via pairwise comparisons
- In vitro studies in stably transfected HepG2 cell lines evaluated potential cross-resistance with telbivudine vs lamivudine and adefovir
*BK definition: HBV DNA >105 after being <105
or
For points never <105 but with 2-log drop, HBV DNA increasing to within 1 log of baseline
NIH BK definition of 1 log above nadir was also used
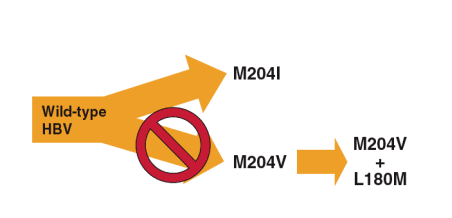
- Telbivudine is very active against the M204V single mutant
- Selection of the M204V single mutant is the first step in a major pathway to LAM resistance involving L180M/M204V1-2
- The activity of telbivudine against M204V leaves selection of M204I as the only pathway to emergence of YMDD mutants
Summary of Key Findings
- Viral BK with genotypic resistance rates for telbivudine were significantly lower than for lamivudine in all patients (HBeAg positive and negative) by both protocol and NIH (1 log above nadir) BK definitions
- All genotypic resistance seen to date for both drugs is due to M204 variants (in the YMDD motif)
- No novel telbivudine resistance determinant has been identified
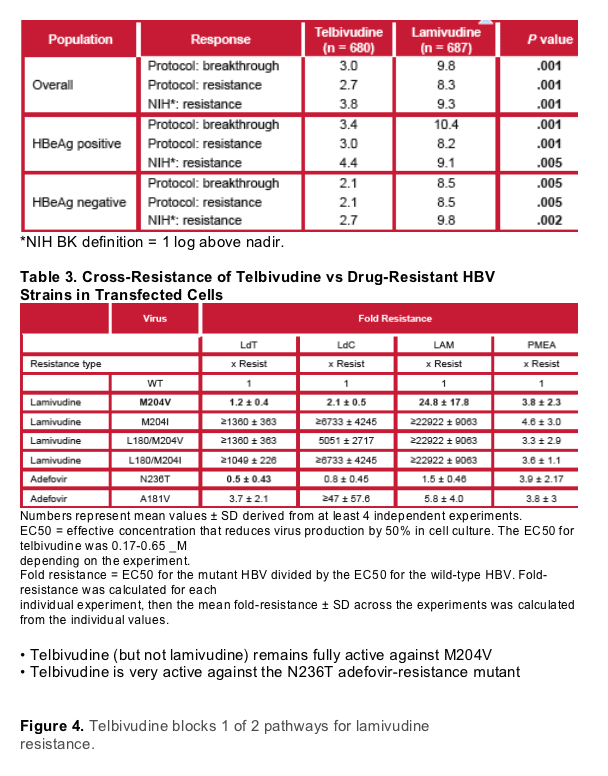
- Lamivudine-resistant patients fell into 3 distinct mutation patterns: 25 (48%) M204I, 15 (29%) M204V, 12 (23%) mixed M204M/I/V
- Telbivudine therapy decreased the incidence of all 3 resistance categories, removing M204V and almost eliminating the mixed M204M/I/V mutants, leaving only M204I (16/17)
- The absence of M204V-based resistance is consistent with in vitro studies showing that telbivudine remains fully active against M204V single mutant
- In vitro, telbivudine resistant mutants remain sensitive to adefovir; conversely, telbivudine is 2-fold more active against the N236T adefovir mutant compared with wild-type HBV
- The M204V lamivudine-resistant variant was ALWAYS associated with the L180M compensatory mutation
- M204I-based telbivudine resistance was NOT associated with L180M
Conclusions
- Telbivudine was associated with significantly lower genotypically confirmed resistance relative to lamivudine
- This difference likely reflects telbivudine's greater antiviral effect and its activity against the M204V mutant in vitro
- The absence of M204V-based resistance suggests that telbivudine may suppress the emergence of fully resistant HBV containing the M204V/L180M double mutant
|
| |
|
 |
 |
|
|