 |
 |
 |
| |
Ritonavir Boosted Saquinavir 1000/100 twice daily vs Kaletra: Preliminary Study Report After 24 Weeks
|
| |
| |
8th International Congress on Drug Therapy in HIV Infection
November 12-16, 2006
Glasgow, Scotland
Mark Mascolini
Interim analysis of a trial comparing lopinavir/ritonavir with the 500-mg saquinavir tablet plus 100 mg of ritonavir twice daily showed similar viral load and CD4 responses after 24 weeks [1]. Some who saw these results detailed at the "Glasgow meeting" questioned early unveiling of the findings in this 48-week trial.
Jihad Slim from St. Michael's Medical Center in Newark and colleagues in Bangkok and Toronto randomized 150 treatment-naive people to saquinavir/ritonavir or lopinavir/ritonavir, both plus fixed-dose once-daily tenofovir/emtricitabine. Median viral loads stood above 100,000 copies in both groups, and average CD4 counts were low--134 in the saquinavir arm and 121 in the lopinavir arm. About one third overall had a pretreatment CD4 count under 50, and 11% in both groups had hepatitis C virus coinfection.
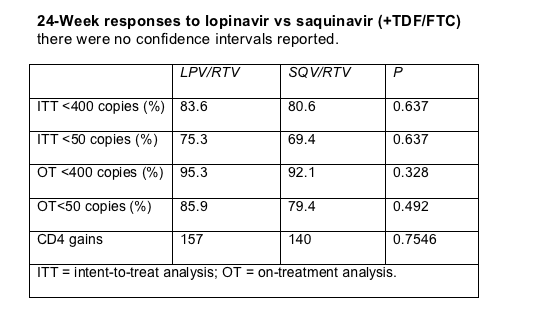
Intent-to-treat and on-treatment analyses showed statistically similar 24-week virologic responses with saquinavir and lopinavir (Table). CD4 gains through 24 weeks were also similar in the two treatment groups.
Five people taking saquinavir/ritonavir had a virologic failure by week 24 (including 2 with poor adherence), as did 2 taking lopinavir/ritonavir (1 with poor adherence). As in earlier studies of ritonavir-boosted protease inhibitors (PIs), new PI-related mutations did not emerge during any of these virologic failures.
Gastrointestinal side effects proved almost twice as common with lopinavir/ritonavir (23%) as with saquinavir/ritonavir (14%). Two serious treatment-related problems in the saquinavir group--hypokalemia and an acute psychotic episode--resolved without further developments. One Thai woman without known liver problems stopped lopinavir/ritonavir and tenofovir/emtricitabine because of liver failure and died several weeks later.
Lipid upswings appeared to favor the saquinavir group. Whereas 17% starting saquinavir had a total cholesterol above 200 mg/dL, 21% had a reading that high at week 24. Respective percentages in the lopinavir group were 13% and 38% (P = 0.036 for 38% vs 21%). While no one had triglycerides above 400 mg/dL when starting saquinavir, 1% did after 24 weeks. Respective percentages in the lopinavir arm were 4% and 13% (P = 0.009 for 13% vs 1%).
Brian Gazzard of London's Chelsea and Westminster Hospital criticized presentation of these interim results ahead of the primary endpoint analysis, a 48-week intent-to-treat reckoning of proportions with a viral load under 50 copies.
Reference
1. Slim J, Avihingsanon A, Ruxrungtham K, et al. Saquinavir BID vs lopinavir plus emtricitabine/tenofovir QD in ARV-naïve HIV-1 infected patients: GEMINI study. 8th International Congress on Drug Therapy in HIV Infection, November 12-16, 2006, Glasgow. Abstract 2.5.
|
| |
|
 |
 |
|
|