 |
 |
 |
| |
SAQUINAVIR/R BID VS. LOPINAVIR/R BID PLUS FTC/TENOFOVIR QD IN ARV-NAIVE HIV-1 INFECTED PATIENTS: GEMINI STUDY
|
| |
| |
Reported by Jules Levin
8th Intl Congress on Drug Therapy In HIV Infection
Glasgow, Scotland
1Slim J, 2Avihingsanon A, 2Ruxrungtham K, 3Schutz M and 4Walmsley S
1St Michael's Medical Center, Newark, USA; 2HIV-NAT, Thai Red Cross AIDS Research Centre, Bangkok, Thailand;
3Roche, Nutley, USA; 4University of Toronto, Toronto, Canada.
Jihad Slim presented today in an oral presentation at the conference an interim analysis at week 24 of the planned 48-week GEMINI Study which compares 1000/100 Saquinavir/r mg twice daily to Lopinavir/r (Kaletra) 400/100 mg twice daily, and both regimens included FYC/tenofovir. . Of note, lipids data and antiviral efficacy were reported. This study examines the Invirase 500 mg tablet formulation of saquinavir. In addition, two posters reported lipids data on this saquinavir dose formulation from the Ipamema Cohort. The lipids look good in all 3 studies.
Author Summary
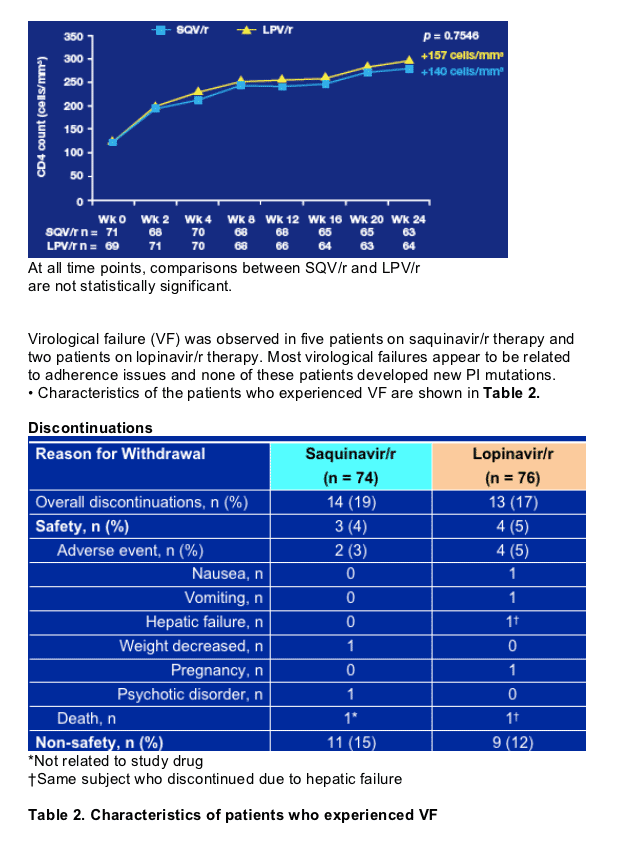
These first data indicate that saquinavir/r is associated with similar rates of viral load suppression, and similar CD4 count increases, to lopinavir/r in ARV-naive patients with advanced HIV disease.
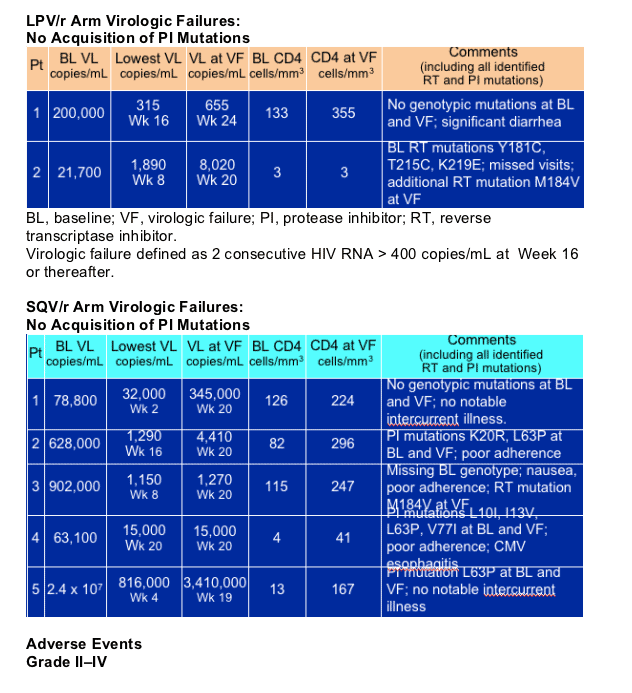
Five patients in the saquinavir/r arm vs. two patients in the lopinavir/r arm experienced virological failure. Poor adherence may have contributed to these failures. None of these patients developed any new PI mutations.
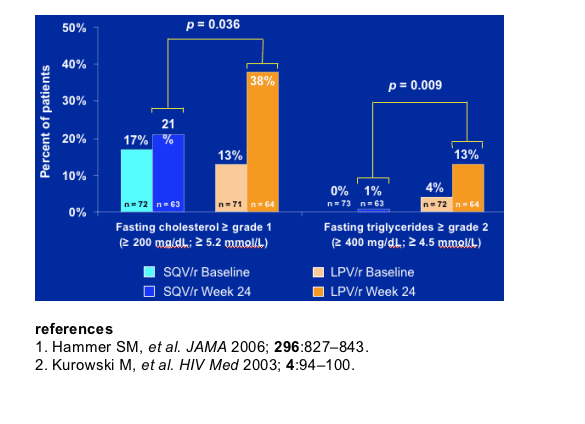
Adverse events (grade 2-4), especially GI events, were more common with lopinavir/r therapy, and significantly more patients in the lopinavir/r arm developed elevated lipids on treatment.
Significantly more patients developed hyperlipidemia on lopinavir/r than on saquinavir/r.
Introduction
Boosted saquinavir (SQV) is a well tolerated and potent protease inhibitor (PI) which is recommended as a treatment for ARV-naive patients in current international HIV treatment guidelines.1
The Invirase formulation of saquinavir has better GI tolerability than the Fortovase (soft capsule) formulation when boosted with ritonavir2 and is now available in a 500 mg film-coated tablet, meaning that the approved saquinavir/r dosage (1000/100 mg bid) can be delivered as two Invirase tablets plus one ritonavir capsule twice daily.
The Gemini study is the first prospective, randomized, controlled study of the new Invirase 500 mg tablet formulation administered at the approved Invirase/r
1000/100 mg bid dosage. The study will compare the efficacy of saquinavir/r bid vs. lopinavir/r bid, each taken in combination with two NRTIs (emtricitabine/tenofovir qd), in ARV-naive patients over 48 weeks.
Two planned interim analyses of safety parameters, CD4 cell count and the proportions of patients with HIV-1 RNA < 50 and < 400 copies/ml are defined in the study protocol: one when the first 150 randomized patients have completed 24 weeks, and one when all patients complete 24 weeks.
Study Objectives
Here they present present the results from the first planned interim analysis. The Gemini study enrolled 337 patients and this interim analysis reports on 150 patients at week 24.
To evaluate the efficacy of saquinavir/r vs. lopinavir/r, each taken twice daily and in combination with once-daily emtricitabine/tenofovir (administered as the co-formulation Truvada) in ARV-naive HIV-1-infected adults
Primary endpoint: the number and percentage of patients with a viral load
< 50 copies/ml at week 48 (intent-to-treat analysis).
Secondary objectives include the evaluation of:
-- Efficacy (including viral load reduction from baseline, change in CD4 count and time to virological failure)
Safety and tolerability (including fasting lipids - total cholesterol, HDL, direct LDL and triglycerides).
Study Design and Patients
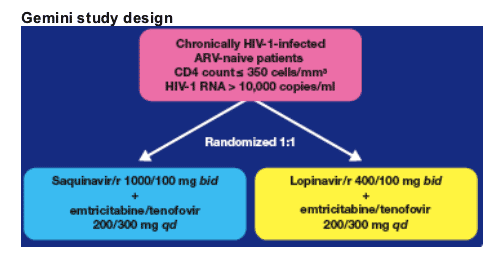
The Gemini study is a prospective, Phase IIIb, randomized, multi-centre, open-label, 48-week study (Figure 1).
Chronically HIV-1-infected ARV-naive patients are randomized to one of the following treatment arms:
-- Saquinavir/r 1000/100 mg bid + FTC/TDF
-- Lopinavir/r 400/100 mg bid + FTC/TDF
Patients in the saquinavir/r arm receive the new Invirase 500 mg film-coated tablet formulation administered with ritonavir.
Key inclusion criteria were:
- HIV-1-infected adults ≥ 18 years of age
- HIV RNA viral load > 10,000 copies/ml
- CD4 count ≦ 350 cells/mm3
- No previous treatment with ARVs (< 2 weeks' total exposure).
Key exclusion criteria were:
- Significant renal and/or hepatic dysfunction
- Current grade 4 clinical/laboratory parameters (patients with asymptomatic grade 4 abnormalities are permitted to participate at the discretion of the investigator).
Key assessments
Protocol-defined study assessments take place at clinic visits at the end of Weeks 2, 4, 8, 12, 16, 20, 24, 36 and 48 (± 7 days of the scheduled visit), or at early termination. Patients return for a follow-up visit 4 weeks after the end of treatment.
Plasma samples are taken for assessment of HIV-1 RNA viral load and CD4 count at all visits.
A fasting lipid profile (cholesterol, HDL, direct LDL and triglycerides) is obtained and fasting insulin is assessed at baseline (BL), Week 24, Week 48 and the 4-week follow-up visit.
Genotyping samples are obtained at screening and stored. HIV genotype resistance testing is performed only in cases of virological failure, defined as any two consecutive HIV-1 RNA > 400 copies/ml at Week 16 or thereafter (censored at Week 48).
Patients who experience virological failure or ARV-related toxicity requiring a change in PI medication are withdrawn from the study and considered a treatment failure.
Statistical analysis
The proportion of patients with a viral load < 50 copies/ml at 48 weeks will be assessed for noninferiority of saquinavir/r vs. lopinavir/r. The calculation of sample size for the Gemini study assumed a response rate of 85% for the primary efficacy endpoint, set the noninferiority margin at a 12% difference in response rate with a significance level of 2.5% and used a 1-sided test of the hypothesis of noninferiority.
Using these criteria, an overall sample size of at least 310 patients (allocated in a
1:1 ratio between the two study arms) provides approximately 90% power for declaring noninferiority of saquinavir/r to lopinavir/r.
All efficacy data will be analysed based on the intent-to-treat population (ITT), which includes patients who are randomized and receive at least one dose of trial medication. For the data analysis of patients experiencing treatment failure, the last on-treatment value/observation will be carried forward (LOCF) to the planned analysis timepoint.
RESULTS
Patient demographics
The Gemini study enrolled a total of 337 patients at 38 study sites between May 2005 and August 2006: 191 patients in the USA and Puerto Rico, 48 in Canada, 38 in France and 60 in Thailand.
Baseline characteristics of the 150 patients who are included in the first pre-planned analysis were similar between the two treatment groups (Table 1).
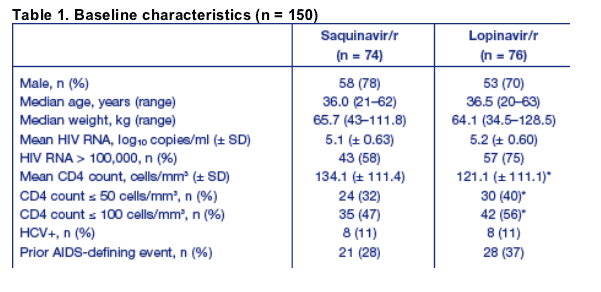
*unavailable for one patient.
The patients in this analysis represent patients with advanced HIV disease for initial ARV therapy:
- 66.7% (100/150) of patients had a BL viral load > 100,000 copies/ml
- 36.2% (54/149) of patients had a BL CD4 count ≦ 50 cells/mm3.
- By Week 24, 14 patients in the saquinavir/r arm and 13 patients in the lopinavir/r arm had discontinued study treatment, mostly for non-safety reasons.
Efficacy
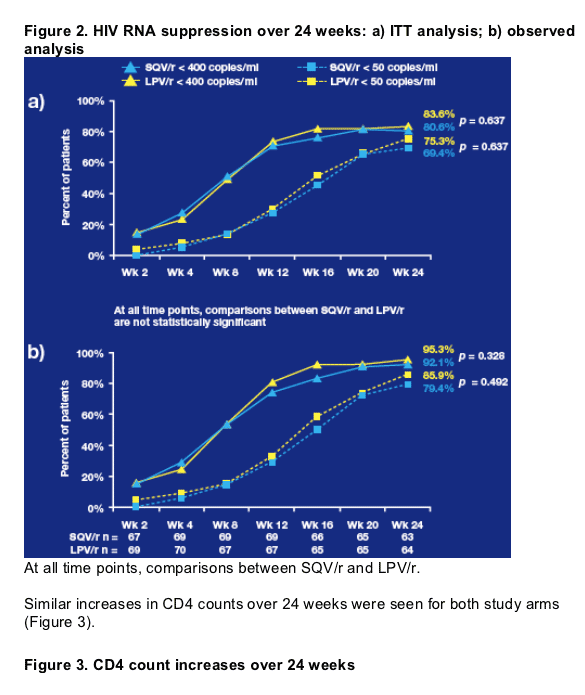
Saquinavir/r therapy resulted in a comparable level of viral load reduction to lopinavir/r, with similar rates of virological suppression seen at 24 weeks in both treatment groups:
-3.2 and -3.5 log10 copies/ml in the saquinavir/r and lopinavir/r arms, respectively (ITT analysis; p = 0.2957).
The observed analysis showed consistent results.
- Virological suppression was similar between the two treatment groups at all interim study timepoints, with no significant differences (Figure 2).
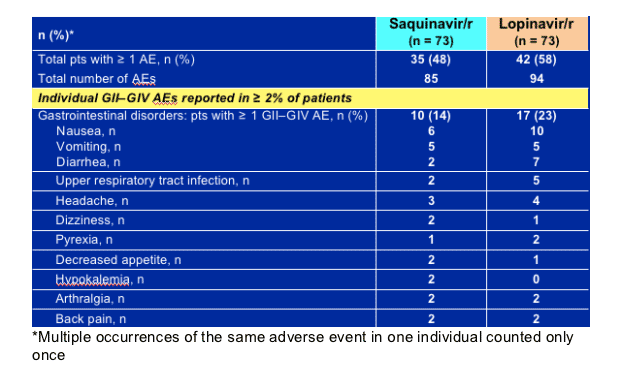
Gastrointestinal adverse events were less common in the saquinavir/r arm than in the lopinavir/r arm (14% vs. 23%, respectively).
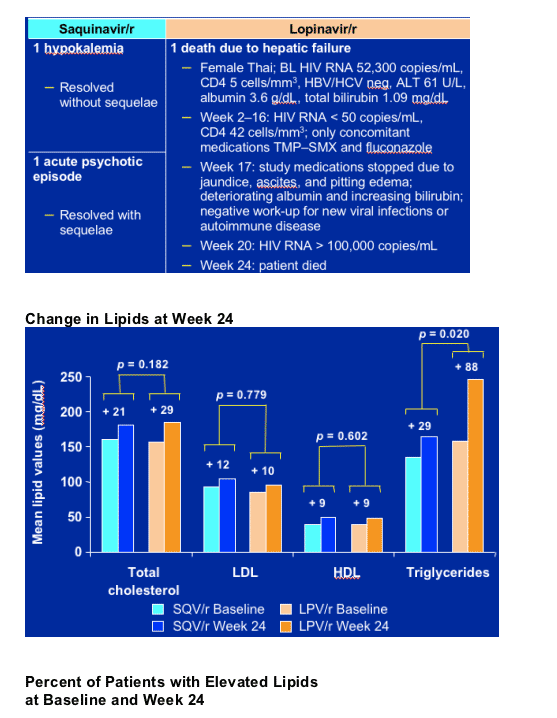
Two serious adverse events, judged by the investigator as study drug related, were reported in the saquinavir/r arm (one hypokalemia, which resolved without sequelae, and one acute psychotic episode, which resolved with sequelae).
There was one death in the lopinavir/r arm that was judged by the investigator to be drug related (a patient without known prior liver disease discontinued due to hepatic failure and subsequently died).
Study Drug-Related* Serious
Adverse Events and Deaths
(*As indicated by the investigator)
|
| |
|
 |
 |
|
|