 |
 |
 |
| |
More Ribavirin, Longer Course Control HCV Better in HIV Patients
|
| |
| |
8th International Congress on Drug Therapy in HIV Infection
November 12-16, 2006
Glasgow, Scotland
Mark Mascolini
Higher weight-adjusted doses of ribavirin (plus pegylated interferon) and longer therapeutic courses improved sustained virologic response (SVR) in people coinfected with hepatitis C virus (HCV) and HIV [1]. But a high dropout rate among people randomized to longer therapy--especially among those with hard-to-treat HCV genotype 1 or 4--somewhat limited interpretation of the PRESCO trial results.
Because people with HIV respond more poorly to pegylated interferon plus ribavirin than do people without HIV, Vincent Soriano from Madrid's Carlos III Hospital and colleagues across Spain offered coinfected people two strategies they hypothesized may improve HCV control. All patients received 180 micrograms of pegylated interferon alfa-2a per week, while those weighing less than 75 kg got 1000 mg of ribavirin daily and those weighing more got 1200 mg daily. Earlier trials often used an unadjusted ribavirin dose of 800 mg. People with difficult-to-treat HCV genotype 1 or 4 were offered treatment for either 48 or 72 weeks, while those with HCV-2 or -3 could get treated for 24 or 48 weeks. Trial participants were not randomized to one therapeutic course or the other. Anyone without at least a 2-log (100-fold) drop in HCV RNA by week 12 had to leave the study.
PRESCO excluded people with active drug addiction, alcohol abuse, decompensated liver cirrhosis, hepatitis B virus infection, or treatment with ddI. Investigators also advised study participants to avoid AZT because it may compound the hematologic toxicity of ribavirin. All study participants had elevated aminotransferases and a CD4 count above 300. The trial enrolled 389 people, 74% of them men, and 90% of them former injecting drug users. While 49% had HCV-1, 38% had HCV-3, 12% HCV-4, and 1% HCV-2. Two thirds had an HCV load above 500,000 IU/mL, and 28% had F3 or F4 fibrosis. Median CD4 count stood at 546, 74% were taking antiretrovirals, and 72% had an HIV load below 50 copies.
Overall SVR stood at 49.6% after a 67.3% end-of-therapy response. SVR proved strikingly better among people with genotype 2 or 3 (Table).
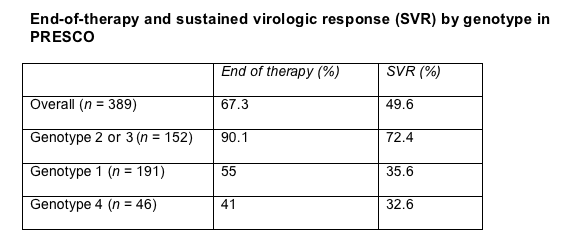
Extended therapy significantly improved SVR among people with genotype 1 or 4 (from 31% to 53%, P = 0.004) and among those with genotype 2 or 3 (from 67% to 82%, P = 0.04). But an 80% voluntary withdrawal rate (36 of 45 people) among HCV-1/4 patients in the extended-therapy arm limits the robustness of the comparison with the short-duration arm. Nine of 56 people (16%) with HCV-2 or -3 quit the extended-therapy arm, compared with 4 of 96 (4%) in the short-duration arm.
Overall, 174 people (44.7%) prematurely discontinued therapy, 66 because of virologic failure, 64 because of voluntary withdrawal, 32 because of serious adverse events, and 12 because of loss to follow-up (failing to return for study visits). While 15.4% had to lower their interferon dose, 21.6% cut their ribavirin dose.
Three factors independently predicted SVR--infection with HCV-2 or -3, lower pretreatment HCV RNA, and HCV RNA below 50 IU/mL at week 12.
Soriano concluded that both the higher ribavirin dose and extended therapy contributed to higher SVRs in this study than in previous trials of people coinfected with HIV. Although relatively few people (8.2%) had to leave the trial because of serious side effects or other setbacks, 29.8% had to trim their dose of either ribavirin or interferon. Soriano recommended that future trials weighing potential benefits of extended therapy should use a 4-week cutoff for undetectable HCV RNA to permit continued therapy. (note from Jules Levin: since the 12-week cutoff is more reliable in predicting SVR, using a 4-week cutoff would I think eliminate some patients who may still mount a viral response between weeks 4 and 12, particularly since coinfected patients may have a delayed viral response).
Reference
1. Nunez M, Miralles C, Berdun MA, et al. The PRESCO trial: role of extended duration of therapy with pegylated interferon alfa-2 plus weight-based ribavirin dose in 389 HCV/HIV co-infected patients. 8th International Congress on Drug Therapy in HIV Infection, November 12-16, 2006, Glasgow. Abstract PL9.5.
|
| |
|
 |
 |
|
|