 |
 |
 |
| |
Boosted Saquinavir Does Not Affect Methadone
|
| |
| |
Reported By Jules Levin
8th Intl Congress on Drug Therapy in HIV Infection, Glasgow, UK
"EFFECT OF BOOSTED SAQUINAVIR ON THE PHARMACOKINETICS AND PHARMACODYNAMICS OF RACEMIC METHADONE IN OPIATE-DEPENDENT PATIENTS ON STABLE METHADONE MAINTENANCE THERAPY"
C. Jamois1, M. Riek1, A. Patel2, P.F. Smith3, R. Morrison4 and C. Schmitt1
1F. Hoffmann-La Roche Ltd, Basel, Switzerland; 2F. Hoffmann-La Roche Ltd, Welwyn, United Kingdom; 3University of Buffalo, Buffalo, USA;
4Northwest Kinetics, Tacoma, USA
INTRODUCTION: HIV infection is highly prevalent among injection drug users. The Methadone Maintenance Treatment (MMT), in place since 1964 as a medical response to the post-World War II heroin epidemic in New York City, is considered an effective treatment for opioid dependence.1 Methadone reduces and/or eliminates the use of heroin, reduces the death rates and crime associated with heroin use and allows patients to improve their health and productive life (employment, education). Methadone is metabolized by CYP3A4 and to a lesser extent CYP2D6. Saquinavir (SQV) and ritonavir (RTV) are CYP3A4 inhibitors. RTV also shows inhibition-associated induction of CYP3A4 and is a strong inhibitor of CYP2D6. This study was performed to more accurately define the interactions between ritonavir-boosted saquinavir (SQV/r) and methadone.
AUTHOR CONCLUSION: There was no clinically significant interaction between ritonavir-boosted saquinavir and standard methadone maintenance treatment. Therefore, boosted saquinavir may be a practical HIV treatment option for patients on methadone maintenance therapy, as no dosage adjustment is required either for saquinavir, ritonavir or methadone. (Previous work has shown that an increase in the AUC of methadone up to 35% and a decrease in methadone AUC up to 30% have not been associated with clinical symptoms of toxicity or methadone withdrawal.3,4)
SUMMARY & DISCUSSION:
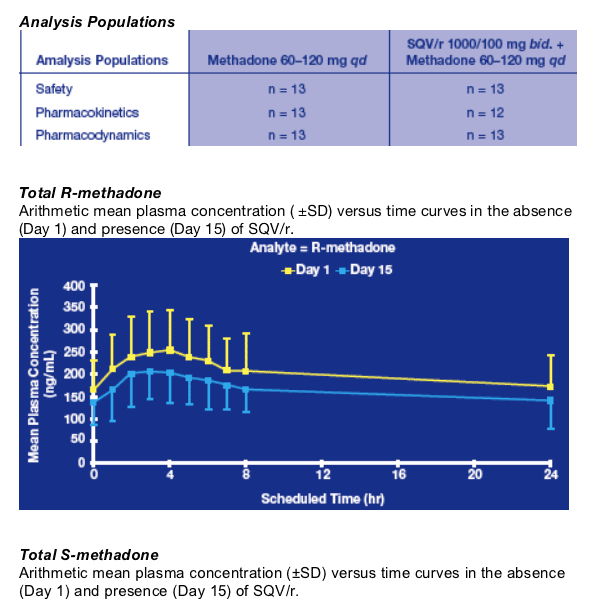
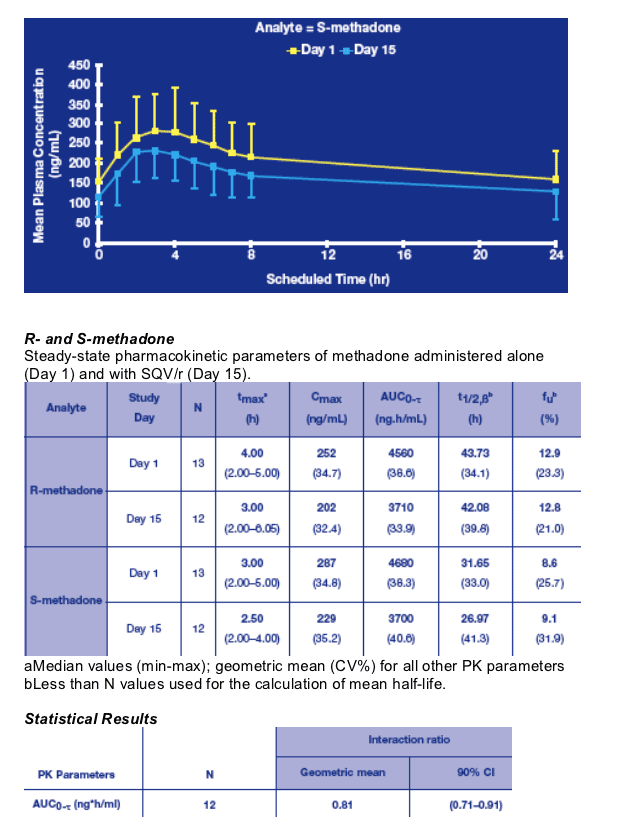
Following 2 weeks of pretreatment with SQV/r, the AUC0-_ of total R-methadone was reduced by 19%, but none of the patients suffered from methadone withdrawal symptoms during the study. Based on the predefined no effect boundaries this decrease was not clinically relevant.
Cytochrome P450 3A4 and to a lesser extent CYP2D6 and CYP1A2 appear to be the main isoforms involved in methadone metabolism.7 Because RTV is a potent inhibitor of CYP3A4 and a moderate inhibitor of CYP2D6, and SQV a moderate inhibitor of CYP3A4, it could have been hypothesized that co-administration of SQV/r with methadone would increase methadone concentrations. However, these data show the opposite effect and corroborate similar results obtained with methadone and RTV in a previous study.8
RTV, which is also an inducer of CYP3A4, may have produced stronger CYP3A4
induction than inhibition. However, the importance of CYP3A4 in methadone
metabolism might be questioned knowing that indinavir, a pure CYP3A4 inhibitor, and rifabutin, a CYP3A4 inducer, have no effect on methadone pharmacokinetics.9,10
Recently, some in vitro data in hepatic microsomes from cell systems expressing
specific human cytochromes have shown that CYP2B6 and CYP2C19 were involved in the N-demethylation of methadone.11 Thus methadone exposure was significantly decreased by concurrent use of efavirenz or nevirapine, resulting in opiate withdrawal symptoms.12-14 Because these two drugs are inducers of CYP2B6,15,16 CYP2B6 may represent an important CYP isoenzyme in metabolism of methadone.
In human hepatocytes, RTV strongly induced CYP2B617 whereas its in vivo effect on drugs metabolized by CYP2B6 or CYP2C19 has not been well characterized. However, RTV may induce CYP2C19, as revealed by the increased exposure of active nelfinavir metabolite, M8.18
The decrease of methadone exposure following co-administration with SQV/r might be due to an induction of CYP2B6 and CYP2C19, which might have been strong enough to overcome the CYP3A4 inhibition.
The _1-acid glycoprotein (AAG) levels were not measured during the study, as no changes were expected in this population of HIV-negative patients stable under MMT, and without chronic diseases other than hepatitis C.19,20 Only the unbound fractions of R- and S-methadone were measured and remained unaffected by pretreatment with SQV/r, suggesting that there is no relevant protein displacement of methadone.
The R enantiomer is responsible for the drug opioid activity and only the unbound
methadone is capable of diffusion within the central nervous system. However, in this study, the moderate diminution of the R-methadone free plasma concentration following co-administration with SQV/r did not lead to development of methadone withdrawal symptoms.21,22
Whether given alone or in combination, all 3 treatments in this study were generally well tolerated. Hyperhidrosis was the most frequently reported adverse event, which is a known adverse event associated with methadone therapy. The majority of adverse events were mild in intensity and most of them were considered by the Investigators to be unrelated to the study drugs. No other clinically relevant changes in laboratory parameters, ECGs or vital signs were noted.
METHODS
Study Design
Two-center, open-label, one-sequence, crossover, multiple-dose study with sole
administration of methadone followed by concomitant administration of methadone and SQV/r study in 13 male and female HIV-negative patients stable on MMT.
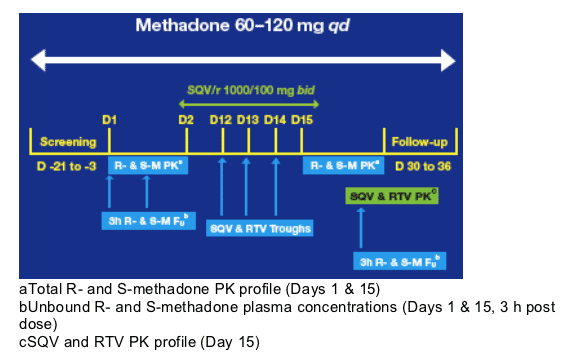
aTotal R- and S-methadone PK profile (Days 1 & 15)
bUnbound R- and S-methadone plasma concentrations (Days 1 & 15, 3 h post dose)
cSQV and RTV PK profile (Day 15)
Scheduled Assessments:
Patients continued to receive their racemic methadone (60-120 mg qd) for the duration of the study and received SQV/r 1000/100 mg bid with food from Day 2 to Day 15.
The pharmacokinetics (PK) of Total R- (the more biologically active) and S-methadone were evaluated at baseline (Day 1) and after 2 weeks of SQV/r co-administration (Day 15). Steady-state SQV and RTV PK were evaluated on Day 15.
For all samples, total plasma concentrations were measured using a validated HPLCMS/ MS method. The lower limit of quantification in plasma was 5 ng/mL for R- and Smethadone and 10 ng/mL for SQV and RTV. In addition, protein binding was determined for both R- and S-methadone using equilibrium dialysis.
Patients were confined to the study center from Day -2 to Day 16. During study
conduct, patients' vital signs were recorded daily at pre-dose; in addition a 12 lead-ECG was recorded at pre-dose on Days 1, 7 and 15. Patients were observed for adverse events and standard safety laboratory assessments were made during the study. Patients were evaluated daily for changes in methadone pharmacodynamics by the Short Opiate Withdrawal Scale and pupillary diameter observations.2 A medical followup was done on Days 30-36.
Pharmacokinetic (PK) Evaluation:
Saquinavir/ritonavir plasma concentrations:
- Blood samples for steady-state assessment were collected at pre-dose on Days 12, 13, 14, and 15.
- Blood samples for full profiles were collected on Day 15 at pre-dose, and at 0.5, 1, 2, 3, 4, 6, 8, 10, 12 and 24 hours post dose.
- PK parameters: Ctrough, AUC0-_, Cmax, tmax, C12h, t1/2β. The PK evaluation was performed using standard non-compartmental methods. AUC0-_ was calculated applying the linear trapezoidal rule.
Total R- and S-methadone:
- Blood samples for full profiles were collected on Day 1 and Day 15 at pre-dose and at 1, 2, 3, 4, 5, 6, 7, 8 and 24 hours post dose.
- PK parameters: AUC0-_, Cmax, tmax, t1/2β.
Unbound R- and S-methadone:
- Samples were collected 3 hours post dose on Day 1 and Day 15.
- PK parameters: fu.
Statistical Evaluation:
The primary study variable was the PK parameter area under the plasma concentration-time curve over one dosing interval _, AUC0-_, of total R-methadone assessed on Day 1 and Day 15.
Individual ratios of Day 15 to Day 1 of the primary study variable AUC0-_ of total
R-methadone was assumed to be independently and identically log normally distributed with parameters _* = exp(_) and _* = exp(_), where _ denotes the expected value and _2 the variance of the natural log transformed individual ratios.
The geometric population mean of the individual ratios, _*, was estimated and reported together with the two sided 90% confidence interval. The coefficient of variation for the individual ratios will be calculated using the relationship CV = exp(_2)-1.
For the primary study variable AUC0-_ of total R-methadone the following hypotheses were tested (_ = 0.05):
- H0: the geometric population mean of the individual ratios of Day 15 to Day
1 lies outside the range defined by the no effect boundaries 0.70 and 1.35
(i.e., ¨_* ≦ 0.70 or _* ≦ 1.35)
versus
- H1: the geometric population mean of the individual ratios of Day 15 to Day
1 lies within the range defined by the no effect boundaries 0.70 and 1.35
(i.e., 0.70 ≦ _* ≦ 1.35)
Previous work has shown that an increase in the AUC of methadone up to 35% and a decrease in methadone AUC up to 30% have not been associated with clinical symptoms of toxicity or methadone withdrawal.3,4 Therefore, 0.70 and 1.35 were selected as clinically acceptable no effect boundaries for methadone AUC.
According to the hypotheses tested, absence of a clinically significant effect of a
1000/100 mg bid combination regimen of SQV/r on a standard methadone maintenance therapy was concluded if a) the 90% confidence interval for _* of the individual ratios of AUC0-_ of total R-methadone is entirely included within the range defined by the no effect boundaries of 0.70 and 1.35; and b) no other information emerging from other collected PK, PD and safety data points towards a potentially clinically significant effect. The decision rule based on the two-sided 90% confidence interval was equivalent to the decision of the two one-sided tests procedure.5
RESULTS
Disposition and Demographics of Study Subjects
13 Caucasian HIV negative patients (5 female) were enrolled into this study.
11 completed the study. Patients ranged in age from 24 to 54 years and their BMIs ranged from 19.1 to 33.5 kg/m2. All patients had negative test results for hepatitis B whereas 12 were tested positive for hepatitis C.
Patient #105, a 46 year old Caucasian male, was withdrawn prematurely and replaced after he became uncooperative and verbally abusive to staff and other participants.
Patient #103, a 28 year old Caucasian male, failed to return to the follow-up visit.
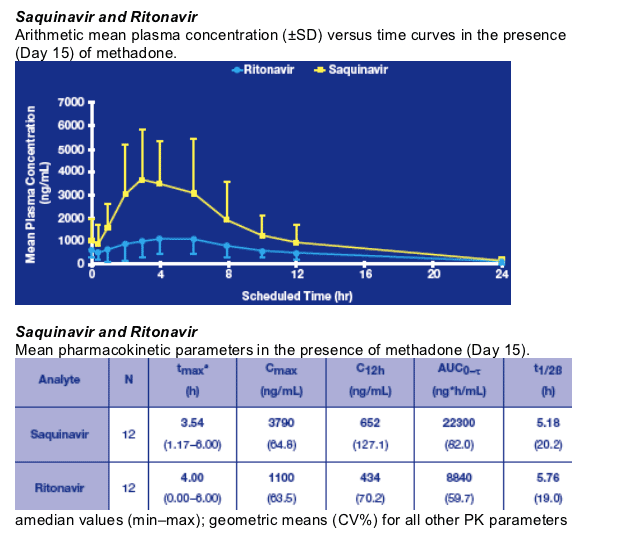
Saquinavir and Ritonavir:
Visual inspection of individual trough concentration data indicated that all patients were at steady state for both SQV and RTV after 2 weeks' treatment. Arithmetic mean trough plasma concentrations for SQV and RTV were approximately 1000 ng/mL and 500 ng/mL, respectively.
The steady-state PK parameters of SQV and RTV were comparable to those observed in previous studies.6
Pharmacodynamics
None of the patients developed methadone withdrawal symptoms during the study.
References
1. Joseph H, Appel P. Historical perspectives and public health issues. In: State Methadone Treatment Guidelines. Treatment Improvement Protocol (TIP) Series #1. 1993, 11-24;
2. Gossop M. Additive Behaviors 1990, 15: 487-490; 3. Gerber JG, et al. J AIDS 2001; 27: 153-160; 4. Cobb MN, et al. Clinical Pharmacology and Therapeutics 1998; 63: 655-662;
5. Schuirman DJ. J Pharm Biopharm 1987; 15: 657 680; 6. Boffito M, et al. 7th International Workshop on Clinical Pharmacology of HIV Therapy April 2006; Lisbon, Portugal; 7. Eap CB, et al.Clinical Pharmacokinetics 2002; 41 (14): 1153-1193; 8. Hsu A, et al. 5th CROI 1998; Chicago, Illinois, USA. #342; 9. Cantilena L, et al. Clin Pharmacol Ther 1999; 65: 135;
10. Brown LS, et al. Drug Alcohol Depend 1996; 43:71-77; 11. Gerber JG, et al. Chirality 2004; 16: 36-44; 12. Clarke S, et al. 7th European Conference on Clinical Aspects and Treatment of HIV-Infection; 1999; Lisbon, Portugal; 13. Altice FL, et al. AIDS 1999;
13: 957-62; 14. Otero MJ, et al. AIDS 1999; 13:1004-5; 15. Riska P, et al. Drug Metabolism Disp 1999; 27: 895-901; 16. Lamson M et al. Clin Pharmacol Ther 1999; 65: 137;
17. Faucette SR, et al. Drug Met.& Disp. 2003; 32: 348-357; 18. Flexner C, et al. XII World Conference on AIDS; 1998; Geneva, Switzerland; #42265; 19. Eap CB, et al. Clin Pharmacol Ther 1990; 47:338-46; 20. Zhang XC et al. J Infect Dis 1999; 180: 1833-7; 21. Jolliet-Riant P, et al. Life Sci 1998; 62: PL219-26;
22.Garrido MJ, et al. J Pharm Pharmacol 1996; 48: 281-4.
|
| |
|
 |
 |
|
|