 |
 |
 |
| |
SQV 500mg (1000/100) in Exp/Naives
|
| |
| |
Reported by Jules Levin
8th Intl Congress on Drug therapy in HIV, Glasgow, UK, Nov 12-16, 2006
"The RAINBOW - COHORT: successful initiation with or switch to the new saquinavir 500 mg formulation - first results from Germany"
1Knechten H, 2Lutz T, 3Knecht G, 4Haberl A, 5Carganico A, 6Glaessel F, 7Jaeger H, 8Tappe A, 9Wellmann E, 10Steinmueller J
1PZB Aachen, Germany; 2HIV-treating physician, Frankfurt; 3HIV-treating physician, Frankfurt;
4Klinikum Joh. Wolfg. Goethe Universität Frankfurt; 5HIV-treating physician, Berlin; 6HIV-treating physician, München;
7HIV Research and Clinical Care Center, Munich; 8-10Roche Pharma AG, Germany
Introduction
Invirase (saquinavir, SQV) boosted by low-dose ritonavir (r) is recommended for initial PI-based antiretroviral therapy (ART) regimens by international HIV treatment guidelines.1-3
SQV is now available in an Invirase 500 mg film-coated tablet formulation that offers a reduction in pill burden versus Invirase 200 mg capsules and a simple dosing schedule of two tablets of Invirase twice daily when given at the approved dosage of SQV 1000 mg bid with ritonavir 100 mg bid (SQV/r).
The aim of the RAINBOW - COHORT is to assess the efficacy and tolerability
of initiating treatment with, or switching treatment to the new Invirase 500 mg
film-coated tablet formulation.
We present an interim 24-week analysis of data from patients in Germany
who are participating in the RAINBOW - COHORT.
AUTHOR CONCLUSIONS
The results of this observational cohort of treatment with the new Invirase 500 mg tablet formulation of SQV are consistent with high efficacy and tolerability results seen in controlled studies of SQV/r.4-6
Favorable virological and immunological responses were seen following initiation of HAART containing SQV/r using the new Invirase 500 mg tablets. HIV RNA levels were either maintained at or suppressed to low levels and CD4 cell counts increased over 24 weeks of treatment in all patient subgroups, including PI-naive patients and PI-experienced patients.
The greatest improvements in disease status were obtained in PI-naive patients
(median increase in CD4 counts + 136 cells/mm3, reduction of median viral load of -2.87 log10 HIV RNA over 24 weeks of treatment and 73 % of patients with a viral load below 50 copies/mL at 24 weeks).
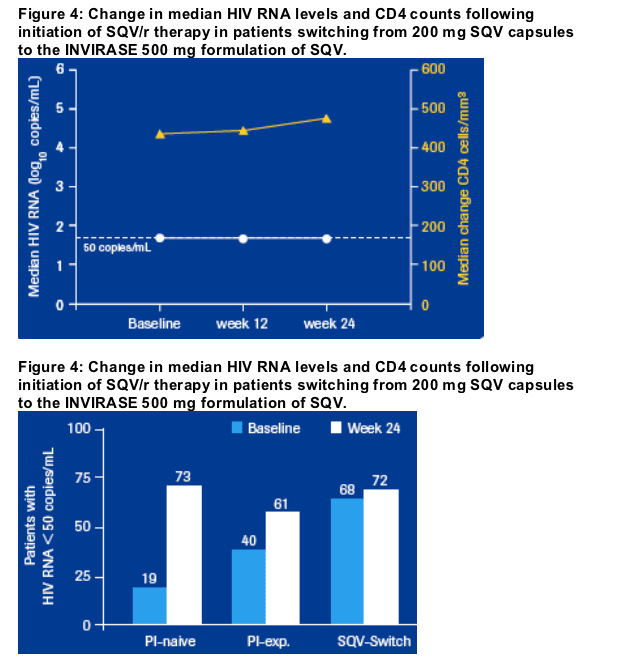
Viral suppression was well maintained in patients who switched from the previous 200 mg formulations of SQV to the 500 mg Invirase tablet; in addition these patients experienced a median increase in CD4 count of 37 cells/mm3 at week 24.
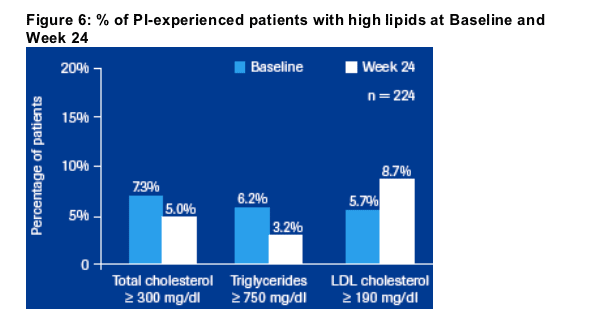
Minor lipid changes were seen and of note, the lipidprofile of PI-experienced patients was substantially improved with decreases in high triglycerides and
high total cholesterol from 6.2% and 7.3% at baseline to 3.2% and 5.0% at week 24, respectively.
Collectively these data confirm that an SQV/r regimen is effective and well tolerated in both PI-naive and PI-experienced patient populations in the realworld clinical setting.
The ongoing Gemini-study - an international, randomized, controlled clinical
trial comparing SQV/r-based therapy with lopinavir/r-based therapy in ARTnaive
patients - will provide further insight into the efficacy, safety and
tolerability of the Invirase 500 mg tablet formulation. (Interim Week 24 results from Gemini reported at Glasgow Nov 2006 and NATAP report is posted on NATAP website).
METHODS
Design
The RAINBOW - COHORT is an international, prospective, noninterventional,
observational cohort of patients who are being treated with HAART containing SQV/r using the new Invirase 500 tablets.
The RAINBOW - COHORT is intended to reflect the real-world clinical practice.
Patients received SQV/r in combination with any other antiretroviral agents deemed necessary to construct a suitable ART regimen.
This analysis primarily assessed the efficacy of the SQV-containing regimen, as determined by reduction in viral load (HIV RNA copies/mL) and increase in CD4 cell count at week 24.
Tolerability assessments included changes in serum lipid levels and the requirement of concomitant medications, including antidiarrheal and lipidlowering
agents, as available.
Patients
The eligible patient population is defined as chronically HIV-1 infected adults who are initiating SQV/r therapy with the Invirase 500 mg film-coated tablet in combination with other ART chosen as appropriate treatment by the treating physician.
Eligible patients include ART-naive patients about to start treatment and ART-experienced patients who are deemed by their physician to need a switch of
therapy.
RESULTS
The German cohort of RAINBOW enrolled 1235 patients between June 2005
and October 2006. Until week 24 data from 706 patients are available. Patient
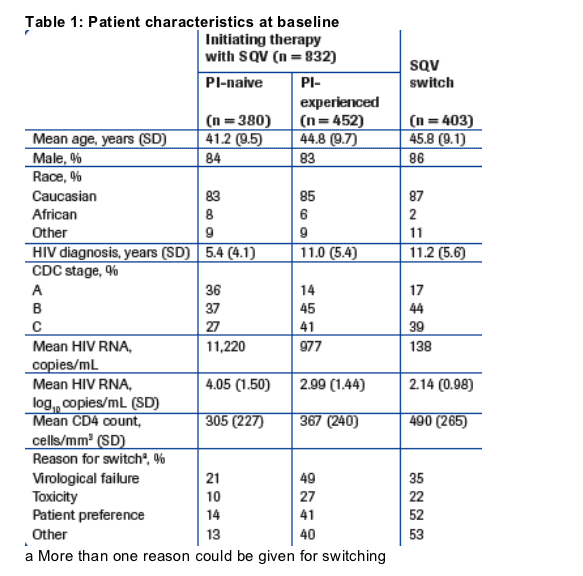
characteristics at baseline are shown in Table 1.
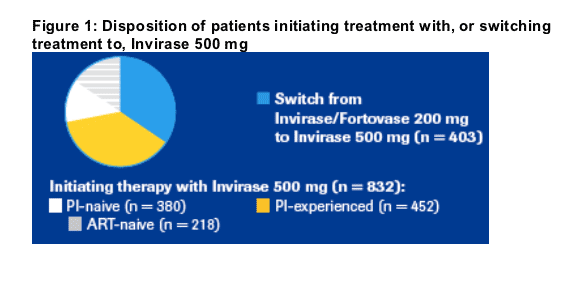
832 patients (67.4%) initiated treatment with Invirase 500 mg:
_ 452 (54.3%) were PI-experienced, and
_ 380 (45.7%) were PI-naive
- Of these 218 (57.4%) were absolutely ART-naive.
403 patients (32.6%) switched from 200 mg SQV capsules to the Invirase
500 mg tablet formulation of SQV ("SQV switch patients") (Figure 1).
Efficacy
Only 23 patients (1.9%) withdrew from the cohort during the first 24 weeks of
treatment.
Patients initiating treatment with SQV/r
PI-naive patients who initiated treatment with SQV/r experienced median
increases in CD4 cell counts from baseline of + 136 cells/mm3 at week 24
(Figure 2). This patient group experienced substantial reductions in viral load
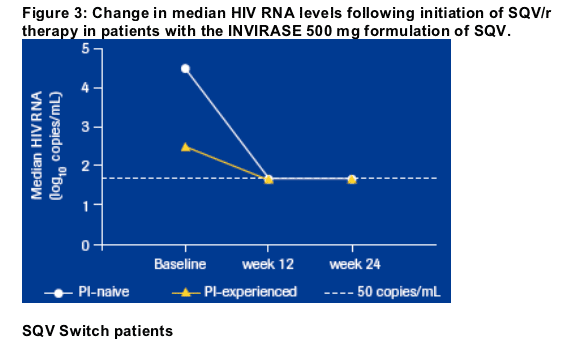
with a median viral load of < 50 copies/mL at 24 weeks (baseline 4.56 log10
copies/mL; Figure 3). The virological suppression rate (< 50 copies/mL) at
24 weeks was 73% compared with 19% at baseline (Figure 5).
Within the PI-naive patients the subgroup of the absolutely ART-naive patients
also experienced a median viral load of < 50 copies/mL at week 24 and a
comparable increase in CD4 cell counts (+ 137 cells/mm3), but startet both
with a higher median viral load of 5.01 log10 copies/mL and a lower baseline
CD4 cell count of 202 cells/mm3.
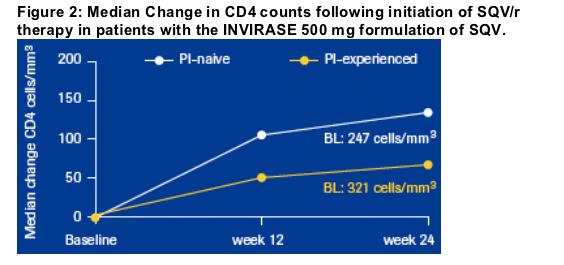
At week 24, PI-experienced patients taking SQV/r for the first time had median increases in CD4 cell counts from baseline of + 60 cells/mm3 (Figure 2) and reductions in viral load, with a median viral load of < 50 copies/mL at week 24 (Baseline 2.48 log10 copies/mL; Figure 3). Between baseline and week 24, the proportion of patients with undetectable viral load increased from 40% to 61% (Figure 5).
Between baseline and week 24, the median viral load remained well controlled
in those patients who switched from SQV capsules to the Invirase tablet formulation of SQV, while there was an increase in the median CD4 cell
count (+ 37 cells/mm3) (Figure 4). The proportion of SQV switch patients with
undetectable viral load (< 50 copies/mL at 24 weeks) was 72%, compared
with 68% at baseline (Figure 5).
Tolerability and Lipids
SQV/r based ART was generally well tolerated across all patient subgroups. The cohort is ongoing and still recruiting, therefore incomplete lipid data are available for the patient population at week 24 and only patients with available lipid data at baseline and at week 24 were considered for analysis. Given the incomplete data, no statistical analyses were performed.
PI-naive patients: Data for PI-naive patients initiating SQV/r therapy indicate
small increases in serum triglyceride levels, total and LDL cholesterol levels
(median increase from baseline to week 24: 22 mg/dL, 23 mg/dL and 12 mg/dL,
respectively). These increases resulted in 3.5% of patients with elevated total
cholesterol (≥ 300 mg/dL) over 1.3% at baseline, with 4.7% of patients with elevated triglycerides (≥ 750 mg/dL) over 1.3% at baseline and with 3.2% of patients with elevated LDL cholesterol (≥ 190 mg/dL) over 1.8% at baseline.
PI-experienced patients: A substantial proportion of PI-experienced patients
initiating SQV/r therapy had high lipid parameters at baseline. The proportion of patients with high triglycerides (≥ 750 mg/dL) and high total cholesterol
(≥ 300 mg/dL) at baseline decreased from 6.2% to 3.2% and from 7.3% to 5.0%, respectively, proportion of patients with elevated LDL cholesterol (≥ 190 mg/dL) increased from 5.7 % to 8.7 % (Figure 6).
Switch patients: For patients switching from former saquinavir formulations there were no substantial changes in any lipid parameters between baseline and week 24 (serum triglycerides, total cholesterol, HDL and LDL cholesterol). The percentage of patients using concomitant medications, including antidiarrheal medicines and lipid-lowering agents, decreased across all patient subgroups between baseline and week 24.
|
| |
|
 |
 |
|
|